
Intraoperative Death at Time of Endoscopic Shunt Placement in Newborn with Myelomeningocele: Case Report
Authors
Trimurti D. Nadkarni, MD†
Harold L. Rekate, MD
Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona †Current Address Department of Neurosurgery, K.E.M. Hospital and Seth G.S. Medical College Parel, Mumbai, India
Abstract
An infant born with massive hydrocephalus and a very small sacral myelomeningocele underwent shunt placement followed by repair of the myelomeningocele during the same surgical procedure. Some cerebrospinal fluid (CSF) was lost during posterior parietal placement of the ventricular catheter, and the fontanel was sunken. The child was placed prone and the myelomeningocele was repaired in the usual fashion. As the baby was transferred to the isolette, CSF suddenly gushed from the burr hole site and the baby suffered cardiorespiratory arrest. Resuscitative efforts, including transfusion and reinfusion of Ringer’s lactate into the ventricular system, were unsuccessful and the infant died. Shunting patients with massive hydrocephalus is often fraught with complications, but the order of the procedures in this case may have increased the infant’s risk. Presumably, if the myelomeningocele had been repaired first the child would have required less handling and the marked shift of brain tissue may have been avoided.
Key Words: cerebrospinal fluid shunt, hydrocephalus, myelomeningocele, neuroendoscopy, shunt complication
In neurosurgery a consensus is growing that surgery to repair myelomeningoceles and to place ventriculoperitoneal (VP) shunts should be performed during the same operative setting.[1-5,7,8,10] Two separate operative procedures with double the attendant anesthetic risk are thereby avoided. Stress on the family is also minimized. Furthermore, the economic impact of a single operation instead of two is compelling. The incidence of cerebrospinal fluid (CSF) leaks from the repair site may be lessened. The risks of shunt infection and malfunction are not increased by performing these procedures simultaneously compared to patients undergoing staged procedures.[8] These advantages are significant. However, little evidence is available to determine which procedure should be performed first.
We would like to draw attention to the fact that repair of the myelomeningocele followed by shunt insertion minimizes the need to move an infant into different positions in the operative field. Such movements may be critical in infants with massive hydrocephalus. The coronal burr hole for endoscopic placement of ventricular catheter appears to be the best option.
Case Report
A 1-day-old female was delivered at 39 weeks gestation by cesarean section because of fetal distress. The mother had received no prenatal care. The fetal heart rate had dropped to about 80 beats/minute. As noted on rupture of the membranes, the amniotic fluid was clear. The birth weight was 2860 g. The infant exhibited evidence of intrauterine growth retardation. A small cleft was present in the soft palate. The umbilical cord had two vessels. Both the 1- and 5-minute Apgar scores were 7. The infant was slightly tachypneic with a respiratory rate of 50 breaths/minute but otherwise was stable.
At birth, the infant had a large head and midsacral meningomyelocele. The neural placode was about 0.5 cm long. There was a clear discharge at the site of the meningomyelocele. The occipitofrontal circumference was 46 cm. The anterior and posterior fontanelles were large, tense, and bulging. The scalp vessels were engorged and en larged. Spontaneous movement in all extremities were hypotonic. The limbs were symmetrical. The infant’s reflexes were positive for grasp, root, and suck.
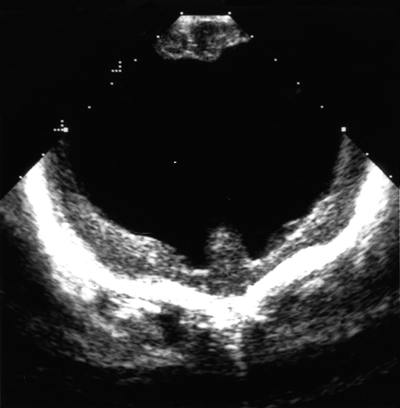
Ultrasonographic examination of the brain showed a very thinned cortical mantle with massive hydrocephalus (Fig. 1). A two-dimensional echocardiogram with color-flow and Doppler analysis obtained before surgery confirmed multiple ventricular septal defects. The overall amount of left-to-right shunting was small. Other anomalies detected were a stretched foramen ovale with a small atrial-level shunt, a small-to-moderate patent ductus arteriosus, and a morphologically abnormal aortic valve with no evidence of stenosis.
The infant was operated upon the day after birth. General anesthesia was induced uneventfully. She was placed supine with her head turned to the left to maximize exposure of the right parietal area. The right head, neck, thorax, and abdomen were prepared and draped in the usual sterile fashion for the insertion of a right VP shunt. The ventricular catheter was introduced under endoscopic guidance through the right parietal burr hole. Fine debris floating in the ventricular cavity suggested that the infant had an intraventricular hemorrhage in utero. The foramen of Monro, septal vein, and choroid plexus were identified. The septum pellucidum was incompetent from massive hydro cephalus, and both frontal horns could be entered. The ventricular catheter was guided into the right frontal horn, and the endoscope was withdrawn leaving the catheter in position. After egress of CSF and valve function were confirmed, the distal portion of the shunt was tunneled from the right parietal incision to the abdominal incision and placed into the peritoneum. All incisions were closed in the routine manner.
At the end of the procedure, the infant’s cranial size had decreased appreciably. The sutures were overriding, and the surrounding skin was sunken. During the intraventricular exploration for shunt placement, a significant amount of CSF was apparently lost through the catheter.
The infant was turned prone. Her lower back was prepared and draped. The small neural placode demonstrated the usual neural elements, including a median raphe, which was dissected free circumferentially. The edges of the isolated neural placode were approximated to create a neural tube. Primary dural and skin closure was achieved.
The blood loss for both these procedures was minimal. The amount of intravenous fluids administered for both procedures was 34 ml. The infant was turned to the supine position and moved to the isolette. A blood-stained CSF discharge was noted at the site of the burr hole.
At this stage, the infant’s respiratory rate began to decrease. Her skin color had become mottled and pale. Atro pine and normal serum albumin were infused. The fontanelle was lax. Despite cardiorespiratory resuscitative measures, the infant could not be revived. A coroner’s examination was requested, but the death was ruled by natural causes and a postmortem was not performed.
Discussion
One-stage meningomyelocele closure and VP shunt placement are well-established practices.[1-5,7,8,10] Parent and McMillan[8] and Reigel and Rotenstein[10]perform the shunting procedure as the first of the two procedures and have reported no immediate complications. Neither McLone,[7] Bell et al.,[1] nor Dias and McLone[4] have commented specifically on the order of shunt placement and back repair. Hubballah and Hoffman[5] and Chadduck and Reding[2] place the child prone, with the head turned to the left and the neck flexed, to permit both procedures to be performed in one position. The shunt is thus placed dorsally and close to a possible relaxing incision.
Fiber optic ventriculoscopy may be used to assist in placing the ventricular catheter in an optimal position.[6] The site of ventricular catheterization tends to reflect the surgeon’s personal preference. Neuroendoscopy for placement of ventricular catheters typically is safe, but endoscopic surgery has an overall complication rate of 7% and a steep learning curve.[11] If, however, the catheter is placed through the occipital route, the burr hole site is below the top of the column of CSF in a supine position. This situation can lead to overdrainage of CSF. The coronal burr hole remains nondependent throughout the procedure. Such an inadvertent CSF leak could be critical if massive hydrocephalus is present.
We elected to place the shunt first, and this choice was likely an error in this specific case. We strongly believe that this infant died from brain collapse precipitated by CSF drainage at the time of ventriculoscopy as well as by pericatheter CSF leakage through the parietal burr hole site. The lowered intracranial pressure (ICP) was aggravated by changing the infant’s position on several occasions within the operating field (i.e., supine, prone, supine). If the myelomeningocele had been repaired first and shunt placement had followed, the infant would have required less repositioning on the operating table and isolette (prone, supine).
The infant’s hydrocephalus was so massive that she would have remained at high risk for a long time. The frequent change in positions following the decrease in ICP from CSF drainage likely precipitated brain collapse and intracranial hemorrhage and led to the infant’s death. Normal variations in intracranial, intraventricular, and subarachnoid pressure with alterations in position are well documented.[9] Intracranial hemorrhage is a complication of lowered ICP in infants,[12] and subdural hematomas result from CSF overdrainage.[9]
Although the exact cause of death remains speculative in the absence of a coroner’s report, the lessons we learned from this tragedy are as follows. The risk of endoscopic shunt placement increases markedly when massive ventriculomegaly is present. CSF leakage might be decreased by placing a coronal burr hole for introducing the endoscope. Manipulation of these infants after shunt placement should be minimized. Hence, the myelomeningocele should be re paired first, especially if massive hydrocephalus is present.
References
- Bell WO, Arbit E, Fraser RAR: One-stage meningomyelocele closure and ventriculoperitoneal shunt placement. Surg Neurol 27:233-236, 1987
- Chadduck WM, Reding DL: Experience with simultaneous ventriculo-peritoneal shunt placement and myelomeningocele repair. J Pediatr Neurosurg 23:913-916, 1988
- Dias MS, McLone DG: Hydrocephalus in the child with dysraphism. Neurosurg Clin N Am 4:715-726, 1993
- Epstein NE, Rosenthal AD, Zito J, et al: Shunt placement and myelomeningocele repair: Simultaneous vs sequential shunting. Review of 12 cases. Childs Nerv Syst 1:145-147, 1985
- Hubballah MY, Hoffman HJ: Early repair of myelomeningocele and simultaneous insertion of ventriculoperitoneal shunt: Technique and results. Neurosurgery 20:21-23, 1987
- Kanev PM, Park TS: The treatment of hydrocephalus. Neurosurg Clin N Am 4:611-619, 1993
- McLone DG: Care of the neonate with a myelomeningocele. Neurosurg Clin N Am 9:111-120, 1998
- Parent AD, McMillan T: Contemporaneous shunting with repair of myelomeningocele. Pediatr Neurosurg 22:132-136, 1995
- Pudenz RH, Foltz EL: Hydrocephalus: Over drainage by ventricular shunts. A review and recommendations. Surg Neurol 35:200-212, 1991
- Reigel DH, Rotenstein D: Spina bifida, in Cheek WR, Marlin AE, McLone DG, et al (eds): Pediatric Neurosurgery. Surgery of the Developing Nervous System. Philadelphia: W.B. Saunders, 1994, pp 51-76
- Teo C, Rahman S, Boop FA, et al: Complications of endoscopic neurosurgery. Childs Nerv Syst 12:248- 253, 1996
- Welch K: The intracranial pressure in infants. J Neurosurg 52:693-699, 1980
