
Giant Pituitary Adenoma with Extension to the Nasopharynx, Sphenoid, and Ethmoid Sinuses: Case Report
Fernando L. Gonzalez, MD
Randall W. Porter, MD
Paul W. Detwiler, MS, MD†
Jacque L. Reiff, RN, BS
Robert F. Spetzler, MD
† Current Address: Tyler Neurosurgical Group, Tyler, Texas
Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
Giant pituitary tumors, defined as larger than 4 cm, present unique diagnostic and therapeutic challenges. This article describes one of the largest adenomas reported in the literature to date and proposes a staged plan for the treatment and management of related complications. Giant pituitary adenomas can be managed effectively with aggressive surgical and medical treatment. A staged surgical technique avoids excessive retraction of the frontal lobes. Endocrinologically, giant lesions are often functionless, eliminating the possibility for medical treatment with dopamine agonists.
Key Words: giant pituitary adenoma, skull base approach
Pituitary adenomas constitute approximately 10% of all intracranial tumors. Grossly, they are classified as microadenomas if their diameter is less than 10 mm or as macroadenomas if it is greater than 1 cm. In 1979 Symon etal.[16] introduced the term giant for pituitary adenomas larger than 4 cm in any direction.[2,12]
Histologically, micro- and macro adenomas are identical.[14,15] Not surprisingly, however, larger lesions have a worse prognosis than smaller ones. In one series, only 3 of 19 patients with suprasellar and lateral extensions were alive a mean of 12 years after surgery.[5] Pituitary adenomas, however, can also be present for many years without causing any symptoms and be discovered only at autopsy.[1] The following case represents one of the largest pituitary adenomas yet reported in the literature.
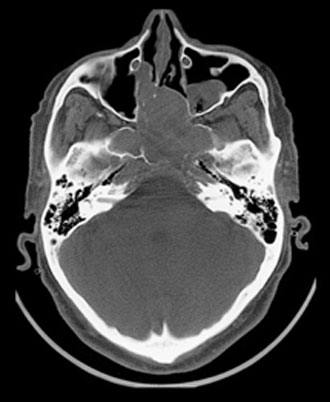
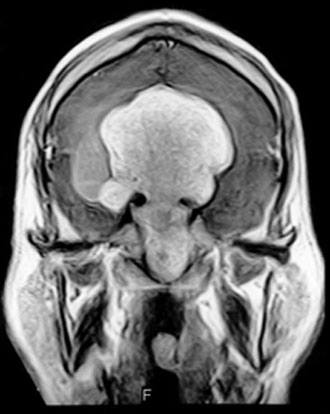
Case Report
A 57-year-old male patient presented with a 3-year history of seizures and cognitive changes. He had been anosmic for 4 years. The patient and his family reported no symptoms attributable to hormonal deficiency. His family had noted that his thinking and concentration were impaired and becoming worse. His medical history was positive for atherosclerotic heart disease and a myocardial infarction. A coronary stent was placed 2 years before admission. A physical examination confirmed complete anosmia. His visual fields were normal to confrontation; otherwise, he was neurologically intact.
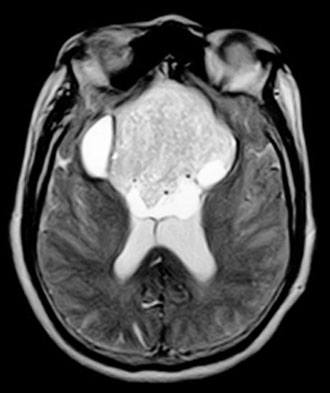
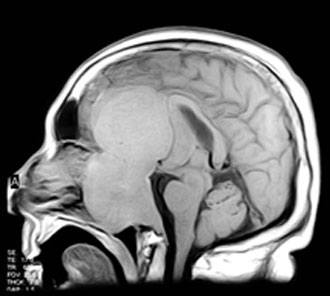
Computed tomography and magnetic resonance (MR) imaging demonstrated a giant, bifrontal, enhancing mass in the anterior cranial fossa. It originated from the sella and invaded the sphenoid and ethmoid sinuses and nasopharynx, displacing the corpus callosum posteriorly (Figs. 1-4). A transnasal biopsy was performed at another facility, and the pathological findings were interpreted as a pituitary adenoma. No preoperative endocrinological evaluation was available. The overall height of the mass was about 11 cm, and the intracranial component was about 8 x 7 cm. The inferior component involving the sphenoid and ethmoid sinuses and posterior nasopharynx measured about 6.3 x 4.6 cm at its greatest dimension.
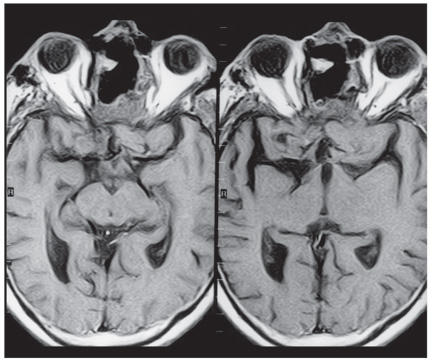
The patient underwent a planned two-staged procedure. First, a bifrontal craniotomy was performed, and the intracranial portion of the tumor was debulked aggressively. There was no evidence of intracranial communication with the sinuses. The dura was closed with a pericranial graft. Tumor was left in the fronto-ethmoid region. In the second stage, the patient underwent a level III transfacial approach (bifrontal craniotomy, frontonaso – orbital osteotomy).[8] The anterior cranial fossa was reconstructed with a split cranial bone graft and a vascularized pericranial flap. The frontal sinuses were obliterated with a temporalis muscle graft.
As expected, postoperative imaging revealed a small amount of residual tumor (Fig. 5). The residual was mainly along the floor of the anterior cranial fossa to maintain separation between the ethmoid and the subarachnoid space. Histological analysis revealed a nonsecreting pituitary adenoma (Fig. 6).
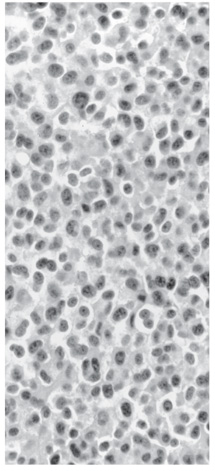
Figure 6. Histological plate showing monotonous cells with round and uniform nuclei and well-defined cell borders. The cytoplasm is eosinophilic. Neoplastic cells were not reactive for prolactin, growth hormone, adrenocorticotrophic hormone, or luteinizing hormone. They were immunoreactive for both keratin AE1/AE3 and synaptophysin, confirming the diagnosis of a pituitary adenoma (hematoxylin and eosin).
Microscopic analysis revealed a glial neoplasm with atypical cells and a high nuclear-cytoplasm ratio, nuclear pleomorphism, and clumped chromatin. Developing rosettes, focal giant cells, and bone invasion were also present. Immunocytochemistry was negative for prolactin and for growth, adrenocorticotrophic, and luteinizing hormones. It was positive for keratin and synaptophysin, confirming that the lesion was a pituitary tumor.
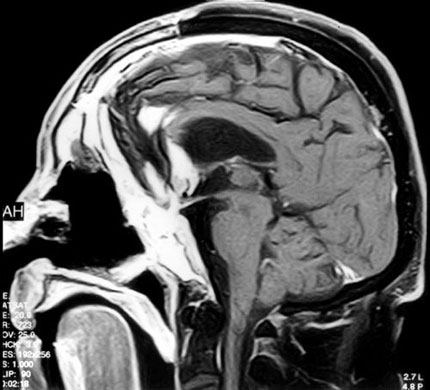
After the second surgery, the patient developed a cerebrospinal fluid leak (CSF) and a gram-negative ventriculitis resistant to traditional treatment. Two weeks after surgery the patient developed a fever. MR imaging showed enhancement consistent with a subdural empyema (Fig. 7), which was evacuated in a third surgery. The right frontal sinus was repacked with a free temporalis muscle flap, and a “feed-me drain-me” antibiotic irrigation system was placed.[9] A tracheostomy also was placed to reduce oral airway pressure in the frontal sinus. The CSF leak resolved 3 days after tracheostomy.
At the patient’s 3-month follow-up examination, the tracheostomy was removed. The residual lesion was treated with adjuvant radiotherapy (total dose, 4500 cGy in 30 daily fractions). At the patient’s 12-month follow-up examination, he had a left temporal visual field cut and anosmia. Otherwise, he was neurologically intact. He received no hormone replacement therapy. He undergoes periodic endoscopic debridement, and he irrigates his sinuses with saline 6 to 10 times daily.
Discussion
Invasive adenomas may correspond to a form of pituitary tumor that is intermediate between the typical benign type and a metastasizing carcinoma.[14] Invasive lesions are cellular and pleomorphic and may show more mitotic activity than noninvasive ones.[14] Invasiveness, however, cannot be assessed histologically, even when mitotic figures and pleomorphism are present.[12,17] Rather, distant metastasis must be demonstrated via the blood stream.[11,14,17] Pituitary carcinomas with or without metastasis are extremely rare.
Pituitary tumors can expand in any direction. They can extend superiorly into the hypothalamus, diaphragm sella, and chiasm; laterally into the middle fossa and cavernous sinus; posteriorly into the posterior fossa; and inferiorly into the sphenoid sinus and clivus. Anteriorly, the ethmoid sinus can be invaded. In rare instances, the tumor can even extend into the nasopharyngeal region,[10] causing symptoms related to nasal obstruction, epistaxis, or recurrent nasal discharge. Recently, a giant pituitary tumor with a nasopharyngeal extension (change in voice and nasal speech) and a significant intracranial component reaching the third ventricle has been reported.[13] No surgical outcome, however, was available because the patient died from a myocardial infarction immediately after surgery. Pituitary adenomas with a suprasellar extension may show a statistically higher proliferation rate as determined by Ki-67.[7,12] The frequency of invasion may range between 5 and 20%, but the incidence is uncertain because the accuracy of histology for detecting invasion is poor.[14]
Diagnosis
The clinical features of giant pituitary adenomas typically reflect the preponderance of mass-related symptoms more than endocrinological features. The latter are most common among women[10,17] while symptoms related to mass effect are most common among men. Our patient had a typical adenoma based on pathological analysis. His symptoms corresponded to the superior and subfrontal extension of the lesion, which compromised the olfactory nerves (complete anosmia) and the frontal cortex producing cognitive changes.
The differential diagnosis of these lesions includes olfactory groove meningiomas. In our patient, however, the tumor extended into the ethmoid and sphenoid sinuses and into the sella, which is an unusual feature for a meningioma. Juvenile angiofibromas grow from the pterygomaxillary fossa[4] and are seldom associated with the superior (subfrontal), symmetrical, and midline extension exhibited by this patient’s tumor. Furthermore, they occur almost exclusively in adolescent males. Esthesioneuroblastomas, another possibility, grow from olfactory neuroepithelium close to the cribriform plate[3] and rarely invade the clivus, as in this case. Nasopharyngeal carcinomas can extend into the anterior and middle fossae,[16] but they seldom extend superiorly as in this patient. Chordomas also must be considered in the differential diagnosis, but to attain the size of this patient’s tumor their clinical manifestation would be far more aggressive and they barely extend in the subfrontal region. The dumbbell shape of our lesion also made chordoma a less likely diagnosis.
The shape of the lesion on MR imaging resembled a girdle (creating a “figure 8”), reflecting the tumor’s growth against a competent sellar diaphragm. This finding is typical of pituitary adenomas. Hypointense-to-isointense lesions are a common finding on T1-weighted MR images; iso-to-hyperintense images are typical of T2-weighted MR images. Cysts may recur subclinically. Symptomatic hemorrhage inside the tumor is also common.
Treatment
Giant pituitary adenomas can rarely be removed completely.[2] Thus, some centers recommend preoperative treatment with dopaminergic agents such as bromocriptine if the lesion is a prolactin secretor.[18] Endocrinologically, giant lesions are often functionless as reported in 35 of 41 (87%) patients with giant pituitary tumors.[6] In this case, histological studies demonstrated a nonsecreting pituitary tumor.
Our patient’s endocrinological status was unavailable. It is important to mention that a normal prolactin level in the presence of a giant pituitary lesion does not eliminate the possibility of a prolactinoma. The “hook effect” is a well-known false-negative (low prolactin level) caused by excessive hormone in the serum sample oversaturating the antibodies and going underdetected, creating a false low-prolactin level. Progressive serum dilution should be performed in the presence of a giant pituitary tumor associated with normal levels of serum prolactin.[2]
A number of approaches have been advocated for the resection of giant adenomas. The transsphenoidal approach is useful for lesions with no lateral extension (cavernous sinus or temporal lobe). A frontal craniotomy is the preferred route for decompressing the optic pathway, internal carotid artery, and middle fossa.[6] The risk of postoperative epilepsy after the subfrontal approach is 15%.[6] The skull base approach used in this case permitted the lesion to be removed from the ethmoid sinus, clival area, and subfrontal area.
A multistaged approach to this kind of lesion is recommended in order to avoid frontal lobe damage. Staging the operation allowed the frontal lobe to expand slowly into the empty space. In our experience, attempts to resect massive tumors of the anterior cranial fossa in one stage uniformly result in severe bifrontal damage. When the anterior cranial fossa is explored, removing the supraorbital bar8 offers a better angle for visualizing the lesion while minimizing retraction over the frontal lobe. In these approaches, reconstructing the floor of the anterior fossa is key. We used a flap of pericranium to create a vascularized barrier to prevent a CSF leak and provided osseous support by suturing the frontal bone to reconstruct the ethmoid plate. No attempt was made to preserve the olfactory nerves. The tracheostomy was made to reduce the pressure on the upper respiratory airway, and the leak resolved definitively. This technique is useful when the respiratory airway is in communication with the healing tissue.
Conclusion
We report the history and radiographic findings of a large pituitary adenoma with an excellent outcome. The primary treatment of this lesion was a staged surgical approach followed by radiation. Complications such as CSF leak, ventriculitis, and empyema are expected. Extensive preoperative informed consent will lead to realistic expectations for the family.
Acknowledgment
We thank Dr. Ameet Patel, MD, Neuroradiology Fellow, for interpreting the radiographs.
References
- Auer RN, Alakija P, Sutherland GR: Asymptomatic large pituitary adenomas discovered at autopsy. Surg Neurol 46:28-31, 1996
- Barkan AL, Chandler WF: Giant pituitary prolactinoma with falsely low serum prolactin: The pitfall of the “high-dose hook effect”: Case report. Neurosurgery 42:913-916, 1998
- Cantrell RW: Esthesioneuroblastoma, in Sekhar LN, Janecka IP (eds): Surgery of Cranial Base Tumors. New York: Raven, 1993, pp 471-476
- Draf W: Juvenile angiofibroma, in Sekhar LN, Janecka IP (eds): Surgery of Cranial Base Tumors. New York: Raven, 1993, pp 485-496
- Hashimoto N, Handa H, Yamashita J, et al: Long-term follow-up of large or invasive pituitary adenomas. Surg Neurol 25:49-54, 1986
- Jeffreys RV: The surgical treatment of large pituitary adenomas. Br J Neurosurg 3:147-152, 1989
- Knosp E, Kitz K, Steiner E, et al: Pituitary adenomas with parasellar invasion. Acta Neurochir Suppl (Wien) 53:65-71, 1991
- Lawton MT, Beals SP, Joganic EF, et al: The transfacial approaches to midline skull base lesions: A classification scheme. Operative Techniques in Neurosurgery 2:201-217, 1999
- Levi AD, Dickman CA, Sonntag VK: Management of postoperative infections after spinal instrumentation. J Neurosurg 86:975-980, 1997
- Levy RA, Quint DJ: Giant pituitary adenoma with unusual orbital and skull base extension. AJR Am J Roentgenol 170:194-196, 1998
- Martins AN, Hayes GJ, Kempe LG: Invasive pituitary adenomas. J Neurosurg 22:268-276, 1965
- Murphy FY, Vesely DL, Jordan RM, et al: Giant invasive prolactinomas. Am J Med 83:995-1002, 1987
- Ravichandran TP, Bakshi R, Heffner RR, et al: Aggressive giant pituitary adenoma presenting as a nasopharyngeal mass: Magnetic resonance imaging and pathologic findings. J Neurooncol 41:71-75, 1999
- Scheithauer BW, Kovacs KT, Laws ER, Jr., et al: Pathology of invasive pituitary tumors with special reference to functional classification. J Neurosurg 65:733-744, 1986
- Srivastava VK, Narayanaswamy KS, Rao TV: Giant pituitary adenoma. Surg Neurol 20:379-382, 1983
- Symon L, Jakubowski J, Kendall B: Surgical treatment of giant pituitary adenomas. J Neurol Neurosurg Psychiatry 42:973-982, 1979
- Tindall GT, Barrow DL: Tumors of the sellar and parasellar area in adults, in Youmans JR (ed): Neurological Surgery. A Comprehensive Reference Guide to the Diagnosis and Management of Neurosurgical Problems. Philadelphia: W.B. Saunders, 1996, pp 2935-2969
- van der Mey AG, van Seters AP, van Krieken JH, et al: Large pituitary adenomas with extension into the nasopharynx. Report of three cases with a review of the literature. Ann Otol Rhinol Laryngol 98:618-624, 1989
