
Diagnosis and Management of Blunt Traumatic Vascular Injuries to the Cervical Carotid and Vertebral Arteries
Authors
Patrick P. Han, MD
David Fiorella, MD
Iman Feiz-Erfan, MD
Felipe C. Albuquerque, MD
Cameron G. McDougall, MD
Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
As awareness increases and radiological testing improves, blunt injury to the carotid artery or vertebral artery is increasingly diagnosed. Although computed tomographic angiography and magnetic resonance angiography are helpful, conventional angiography remains the gold standard for diagnosis. For most blunt carotid and vertebral injuries, heparin therapy is the preferred treatment unless contraindicated by associated injuries. Close angiographic follow-up is warranted to identify healing or progression of arterial dissections or pseudoaneurysms. Pseudoaneurysms are less likely to heal than dissections. Indications for surgical or endovascular revascularization include vascular injuries that cannot be anticoagulated because of associated injuries and injuries that remain symptomatic despite anticoagulation, pseudoaneurysms, asymptomatic occlusions, flow-limiting dissections, or pseudoaneurysms associated with poor cerebrovascular reserve. Long-term follow-up is needed to clarify these indications, but endovascular stent placement appears promising for the treatment of these lesions.
Key Words: blunt trauma, carotid artery, vertebral artery
Carotid and vertebral artery trauma can result from nonpenetrating, penetrating, or iatrogenic injury. The traumatic injury is often diagnosed after the patient experiences a devastating stroke.[5,10,12] Thus, identification of a vascular injury before a stroke occurs is crucial. Once such injuries are found, their treatment can be complicated by a coexistent head or systemic injury, or both. Treatment options include antiplatelet or anticoagulation medical therapy,[5,10,12] surgical reconstruction,[11] and, more recently, endovascular treatments.[6,8]
Illustrative Cases
Case 1
Six months earlier, a 23-year-old female had suffered multiple injuries, including orbital fractures, cardiac and pulmonary contusions, a pneumothorax, and a traumatic brain injury in a skiing accident. During her hospitalization magnetic resonance (MR) imaging revealed several small ischemic infarcts within the territories of the left carotid and left vertebral arteries. Conventional cerebral angiography performed soon after injury revealed a large pseudoaneurysm involving the left internal carotid artery (ICA) at the skull base and a small pseudoaneurysm involving the proximal left vertebral artery. The patient underwent anticoagulation therapy and had no further ischemic symptoms. Follow-up cerebral angiography obtained about 2 months after injury revealed a slight enlargement of the carotid pseudoaneurysm, which measured 21 x 15 mm, and a much smaller pseudoaneurysm (4 x 2 mm) on the left proximal vertebral artery.
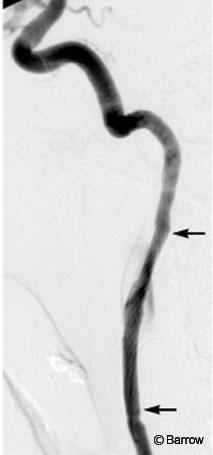
Six months after injury the patient was alert, oriented, and followed commands. She exhibited mild postoperative ptosis of the left eye and intact extraocular movements. Her pupils were equal and reactive. Her face was symmetric. She was mildly hoarse from paralysis of the left vocal cord sustained in her initial injury. Strength in her upper and lower extremities was normal. Cerebral angiography demonstrated slight enlargement of the pseudoaneurysm on the left ICA (Fig. 1). The pseudoaneurysm on the left vertebral artery appeared smaller.
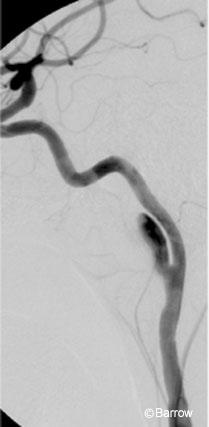
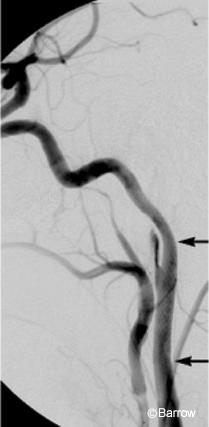
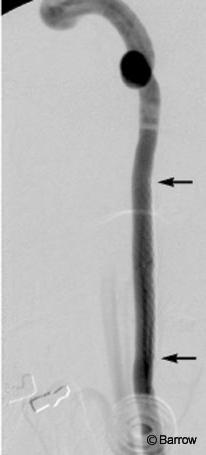
Using standard endovascular techniques with local anesthesia, a 6-mm x 20-mm wall stent was placed across the pseudoaneurysm on the left ICA. Angiography obtained immediately after the stent was placed (Fig. 2) showed reduced blood flow into the pseudoaneurysm. After the procedure, the patient remained at her neurological baseline and was transferred to the intensive care unit. She was discharged home the next day with dual antiplatelet therapy for 3 weeks and was then maintained only on aspirin. She remained neurologically stable. Follow-up cerebral angiography (Fig. 3) obtained 4 months later revealed complete resolution of both pseudoaneurysms.
Case 2
A 45-year-old male driving a motorcycle collided with a pickup truck. He sustained a severe fracture of the left femur and a fracture of the right tibia-fibula. He also developed aphasia and right-sided weakness. He was placed on heparin and transferred to our facility after stabilization at an outside hospital, where his orthopedic injuries were treated and bilateral ICA dissections were diagnosed.
After transfer to our facility, cerebral angiography confirmed the bilateral ICA dissections and showed a left direct carotid- cavernous fistula. The patient had already suffered an infarction of the left hemisphere and developed a retroperitoneal hematoma after heparin therapy.
The heparin therapy was discontinued, and a superficial temporal artery onlay bypass was performed to revascularize the left hemisphere. Eventually, the heparin was resumed and he was converted to warfarin. During his 1-month hospitalization, his aphasia and weakness gradually improved. He was transferred to a rehabilitation center for further recovery.
At his 6-month follow-up examination, his neurological condition had improved. He was awake, alert, followed commands, and had some mild residual aphasia. His cranial nerve function appeared intact. He had minimal right-sided pronator drift and still required wheelchair assistance due to the severe fractures involving his lower extremity. He was still on the warfarin.
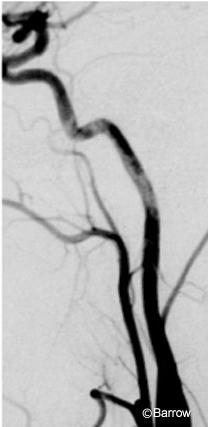
The patient was placed under local anesthesia with intravenous sedation. Angiography demonstrated occlusion of the left ICA; therefore, the left carotid- cavernous fistula no longer filled. Compared to previous studies, the pseudoaneurysm on the right ICA had enlarged (Fig. 4). Severe stenosis caused by the dissection was treated with a self-expanding 6-mm x 31-mm Magic Wallstent (Schneider, Bulach, Switzerland).
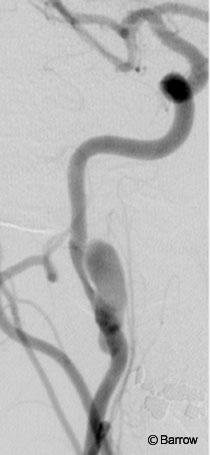
Immediate angiography showed improvement in the stenotic vessel with slow filling of the pseudoaneurysm (Fig. 5). After the procedure, the patient was neurologically stable and was discharged home the next day. He was maintained on dual antiplatelet therapy for 3 weeks and then maintained on aspirin alone.
Follow-up cerebral angiography performed 5 months after stent placement confirmed excellent patency of the stented vessel with almost complete resolution of the previous pseudoaneurysm (Fig. 6).
Diagnosis of Blunt Cervical Carotid and Vertebral Injuries
Figure 4. Right common carotid artery angiogram shows a pseudoaneurysm involving the internal carotid artery.
Traditionally, blunt injuries of the carotid have been considered rare with an incidence on the order of 0.1%.[10] In recent studies of aggressive screening of patients with blunt head and neck trauma for blunt cerebrovascular injury, however, that incidence increased to 1.0%.[9] Patients with blunt cerebrovascular trauma were screened prospectively to compare the accuracy of computed tomographic (CT) angiography and MR angiography to that of conventional angiography.[9]
The screened population included all patients with cervical spine fractures, LeFort II or III facial fractures, Horner’s syndrome, skull base fractures involving the foramen lacerum, soft-tissue injuries of the neck, or neurological abnormalities unexplained by intracranial injuries. Patients underwent screening with four-vessel cerebral angiography. During the first half of the study helical CT angiography was performed and selected patients underwent MR angiography. At diagnosis anticoagulant or antiplatelet therapy was begun unless clinically contraindicated.
Of 216 patients screened in this study over 2 years, angiography identified a carotid artery injury in 24 patients and a vertebral artery injury in 43 patients.[9] The overall screening yield in this selected population was 29% or a 1% incidence of blunt cerebrovascular injury for the entire population with blunt trauma. Compared to a previous study performed at this institution,[7] the stroke rates were similar for carotid artery injury but were lower for vertebral artery injury (0% compared to 14% in the previous study). Based on 143 patients the sensitivity of CT angiography for carotid injury was 47% and the sensitivity of MR angiography was 50%. For vertebral artery injuries, the sensitivity of CT angiography was 53% and that of MR angiography was 47%. The authors concluded that early identification and treatment of blunt cerebrovascular injury may reduce stroke rates and that less invasive imaging modalities such as CT angiography and MR angiography were inadequate for screening.
Recently, evidence-based guidelines based on a comprehensive computerized search of the National Library of Medicine for the management of vertebral artery injuries after nonpenetrating cervical trauma have been published.[2] The evidence was insufficient to support diagnostic standards or guidelines. Conventional angiography or MR angiography was the option for the diagnosis of vertebral artery injury after nonpenetrating cervical trauma in patients with complete cervical spinal cord injuries, fractures through the foramen transversarium, facet dislocations, and/or vertebral subluxation. Evidence was insufficient to support treatment standards or guidelines. Treatment options include anticoagulation therapy with intravenous heparin for patients with a vertebral artery injury who have evidence of a posterior circulation stroke. Either observation or treatment with anticoagulation is recommended for patients with vertebral artery injury and evidence of posterior circulation ischemia. Observation is recommended for patients with a vertebral artery injury and no evidence of posterior circulation ischemia.
Biffl et al.[3] have reported the largest prospective study using conventional angiography. Patients were chosen from 7,205 blunt trauma patients using clinical and radiographic criteria. Patients underwent angiography if they had bleeding from the mouth, nose or ears; a cervical bruit; an expanding cervical hematoma or a cerebral infarction on CT; or a lateralizing neurological deficit. Asymptomatic patients were selected for angiography if they had a cervical hyperextension-rotation injury, a hyperflexion injury, a closed head injury associated with diffuse axonal injury, soft-tissue injuries to the neck, basilar skull fractures extending into the carotid canal, and fractures or distractions of the cervical vertebral body, or if they had experienced near hanging. Thirty-eight patients with a vertebral artery injury were identified. The incidence of posterior circulation stroke was 24%, and the mortality rate related to blunt vertebral artery injury was 8%.
Various pathologies can result from blunt trauma to the cervical carotid and vertebral arteries: (1) occlusion and thrombosis, (2) intimal flap or dissection, (3) complete transection, (4) pseudoaneurysms, (5) or a combination of injuries.[8] Complications and death from these injuries can be caused by embolic stroke, hemodynamic compromise resulting in ischemia and stroke, localized mass effect, injury compromising the airway, and Horner’s syndrome.
Symptoms from a blunt injury to the carotid artery or vertebral artery may develop hours to weeks after the initial insult. Because the initial injury is often asymptomatic, the diagnosis of blunt carotid artery injury or vertebral artery injury may be delayed. Physical examination may reveal a localized hematoma or pulsatile mass, unilateral Horner’s syndrome, bruit, or palpable thrill. These findings should motivate further radiographic imaging to identify possible vascular injury. However, as many as 50% of patients with blunt trauma and blunt carotid injury have no external signs of neck trauma. Thus, identifying blunt cerebrovascular injury in asymptomatic trauma patients requires a high index of suspicion and an aggressive diagnostic approach.
Treatment of Blunt Cervical Carotid and Vertebral Injuries
The management of blunt traumatic injuries involving the carotid and vertebral arteries depends on the type of injury and its location, the patient’s neurological status, and the presence of associated injuries. The goal of treatment is to reduce the risk of embolic or ischemic stroke from occlusion or severe stenosis.
Occluded arteries are often well tolerated when the circle of Willis is competent and provides collateral circulation. Cerebral angiography can identify the latter. A cerebral blood flow study with CT or MR perfusion imaging can help provide additional data on the cerebrovascular reserve of the affected hemisphere. If the patient cannot tolerate a traumatically occluded artery, a surgical bypass procedure can be performed for revascularization. If a traumatic dissection has caused severe stenosis and ischemia, surgical bypass, direct surgical repair, or endovascular revascularization can be performed.
Endovascular stent or stent-grafts can be used to provide urgent revascularization during angiography. Multiple case reports and small series have been published regarding endovascular revascularization, but no substantial long-term data on the outcomes of endovascular treatment of traumatic vascular injury are available. Endovascular treatment, however, offers many advantages: (1) the ability to treat the lesion immediately at the time of diagnostic angiography, (2) the potential to preserve the parent artery, (3) the potential use of local anesthesia, (4) rapid recovery times, (5) a small likelihood of injuring related structures in the neck and cervical column, and (6) access to areas potentially difficult to access surgically (i.e., zone I or III injuries). The disadvantages of the technique include the need for antiplatelet medication after stent placement and the lack of long-term follow-up data on patency rates and definitive repair of stented and stent-grafted injuries.
If the patient is able to tolerate the occluded artery, conservative medical management is used to prevent future thromboembolic events, including anticoagulation or antiplatelet medications if not contraindicated by the presence of other injuries. The rationale for anticoagulation or antiplatelet medication is to minimize clot formation at the injury site to allow the internal fibrinolytic system to lyse the clot and to prevent embolization of the clot.[9]
A dissection with or without formation of a pseudoaneurysm often follows a blunt injury to the carotid or vertebral artery. In multiple case reports and small series, dissections with or without a pseudoaneurysm have been treated by observation, anticoagulation therapy, surgical reconstruction, or endovascular stent placement. Fabian et al.[7] reported 67 patients with 87 blunt carotid injuries: 54 dissections, 11 pseudoaneurysms (all had associated dissections), 17 thromboses, 4 carotid-cavernous fistulas, and 1 transected ICA. Five injuries involved the common carotid artery and 82 injuries involved the ICA. The four patients with a carotid-cavernous fistula were treated with balloon embolization. The transected ICA was ligated. Of the other 62 patients, 47 (76%) received heparin at diagnosis, 8 received no therapy, 6 received aspirin, and 1 underwent surgery.
In this study,[7] a significantly better outcome was associated with heparin therapy compared to no heparin (p<0.01). Six complications were associated with heparin therapy: one gastrointestinal hemorrhage, one hemorrhage from a recently repaired liver laceration, one tracheal hemorrhage, two subdural hematomas requiring evacuation, and one worsening intraventricular hemorrhage. Despite these complications, heparin therapy was associated with independent survival and improved neurologic outcomes. Of the 46 survivors, 39 underwent angiographic follow-up at a mean of 172 days. Of those with a dissection, 62% appeared normal on angiography but 29% had developed pseudoaneurysms. None of the latter reverted to normal.
These findings argue for close angiographic follow-up of blunt cerebrovascular injuries. Many dissections heal, allowing anticoagulant therapy to be discontinued. Some dissections progress to pseudoaneurysm formation, prompting continuation of anticoagulation and possibly endovascular stent treatment. A recent study by Biffl et al.[4] supports these findings. They identified 114 patients with 157 blunt carotid artery injuries and 79 patients with 97 blunt vertebral artery injuries. A grading system was devised: I, intimal irregularity; II, dissection/flap/thrombus; III, pseudoaneurysm; IV, occlusion; and V, transection. The distribution of injury by grade was as follows: 137 grade I, 52 grade II, 32 grade III, 25 grade IV, and 8 grade V injuries. Seven to 10 days after injury, 114 (73%) carotid and 65 (67%) vertebral arteries were restudied with angiography: 82% of the grade IV and 93% of the grade III injuries were unchanged. However, 57% of the grade I and 8% of the grade II injuries healed, allowing the cessation of therapy. Eight percent of grade I and 43% of grade II lesions worsened to pseudoaneurysm formation and prompted interventional treatment. Thus, routine follow-up angiography changed therapy in 61% of the grade I and II patients.
Coldwell et al.[6] reported the endovascular stent treatment of 14 patients with 16 extracranial posttraumatic ICA pseudoaneurysms. Initially, these patients were part of a cohort of patients diagnosed with traumatic dissections who were placed on heparin anticoagulation. If an angiogram obtained 7 days after injury revealed a pseudoaneurysm, a stent was placed across the neck of the patient’s pseudoaneurysm and the patient was converted to warfarin. If the pseudoaneurysm had resolved 2 months later, anticoagulation therapy was discontinued and the patient was switched to aspirin. If the pseudoaneurysm persisted, the patient continued the warfarin and was re-evaluated every 2 months until the pseudoaneurysm resolved. In 12 patients the pseudoaneurysm resolved after stent placement. The pseudoaneurysm had resolved incompletely on the 2-month angiogram in two patients who required two more months of warfarin. At their 4-four month follow-up, angiography showed that their pseudoaneurysms had resolved. One patient exhibited mild (10%) initial hyperplasia. No complications or neurological symptoms were reported attributable to stent placement at a mean follow-up of 16 months.
Albuquerque et al.[1] reported 12 patients who underwent 13 carotid dissections. Angioplasty and stenting were performed successfully in 11 of the 13 procedures, and follow up demonstrated excellent patency in nine vessels. Two required further angioplasty and stenting for stenosis outside the previously treated arterial segments. Six patients had pseudoaneurysms, four of which were treated with stenting across the lesion alone. All pseudoaneurysms treated in this fashion resolved.
Conclusion
Although long-term follow-up studies are needed to clarify the indications, the placement of endovascular stents appears promising for the treatment of blunt traumatic injuries of the cervical carotid and vertebral arteries.
References
- Albuquerque FC, Han PP, Spetzler RF, et al: Carotid dissection: Technical factors affecting endovascular therapy. Can J Neurol Sci 29:54-60, 2002
- Joint Section on Disorders of the Spine and Peripheral Nerves, American Association of Neurological Surgeons/Congress of Neurological Surgeons: Management of vertebral artery injuries after nonpenetrating cervical trauma. In Guidelines for Management of Acute Cervical Spinal Injuries. Supplement to Neurosurgery, vol. 50, 2002, pp. S173-S178
- Biffl WL, Moore EE, Elliott JP, et al: The devastating potential of blunt vertebral arterial injuries. Ann Surg 231:672-681, 2000
- Biffl WL, Ray CE, Jr., Moore EE, et al: Treatment-related outcomes from blunt cerebrovascular injuries: Importance of routine follow-up arteriography. Ann Surg 235:699-706, 2002
- Cogbill TH, Moore EE, Meissner M, et al: The spectrum of blunt injury to the carotid artery: A multicenter perspective. J Trauma 37:473-479, 1994
- Coldwell DM, Novak Z, Ryu RK, et al: Treatment of posttraumatic internal carotid arterial pseudoaneurysms with endovascular stents. J Trauma 48:470-472, 2000
- Fabian TC, Patton JH, Jr., Martin A, et al: Blunt carotid injury. Importance of early diagnosis and anticoagulant therapy. Ann Surg 233:513-525, 1996
- Larsen DW: Traumatic vascular injuries and their management. Neuroimag Clin N Am 12:249-269, 2002
- Miller PR, Fabian TC, Croce MA, et al: Prospective screening for blunt cerebrovascular injuries: Analysis of diagnostic modalities and outcomes. Ann Surg 236:386-395, 2002
- Parikh AA, Luchette FA, Valente JF, et al: Blunt carotid artery injuries. J Am Coll Surg 185:80-86, 1997
- Schievink WI, Piepgras DG, McCaffrey TV, et al: Surgical treatment of extracranial internal carotid artery dissecting aneurysms. Neurosurgery 35:809-816, 1994
- Watridge CB, Muhlbauer MS, Lowery RD: Traumatic carotid artery dissection: Diagnosis and treatment. J Neurosurg 71:854-857, 1989
