
Endocrinologic Issues in Children with Hypothalamic Hamartomas
Donna C. Wallace, RN, MS, cPNP
William L. White, MD
Roger Johnsonbaugh, MD*
Harold L. Rekate, MD
Divisions of Neurological Surgery and *Pediatric Endocrinology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
The preoperative endocrinologic status of patients seeking treatment for intractable seizures associated with hypothalamic hamartomas was reviewed retrospectively. Hypothalamic hamartomas manifest with precocious puberty or seizures and behavioral disturbances. The epileptic component tends to be resistant to pharmacologic treatment and has also resisted management by surgical procedures that do not address resection of the hamartoma itself. We reviewed the preoperative endocrinological status of 22 patients who underwent resection of their hypothalamic hamartoma in an attempt to cure or ameliorate their medically intractable seizure disorder. Of these 22 patients, 13 (59%) had presented with signs and hormonal studies consistent with precocious puberty and had previously had their condition treated medically with gonadotrophinreleasing hormone (GnRH) analogue. From these results we conclude that patients with hypothalamic hamartomas associated with intractable epilepsy also frequently exhibit a syndrome of precocious puberty that can be managed well medically. The coexistence of these two conditions related to a hypothalamic hamartoma does not affect the decision to manage these patients surgically. Previous studies have found that precocious puberty can be managed successfully by resecting a hamartoma through a pterional or modified orbitozygomatic approach. However, it is too early to assess the need for hormonal treatment after resection through a transcallosal interforniceal approach.
Key Words: endocrinology, hamartoma, hypothalamus
As the name implies, hypothalamic hamartomas are abnormal collections of tissue containing disorganized neuronal tissue that occurs as a birth defect within the hypothalamus. In some children, these lesions manifest with precocious puberty and a characteristic syndrome involving epilepsy and behavioral abnormalities. Children with hypothalamic hamartomas may have both conditions.
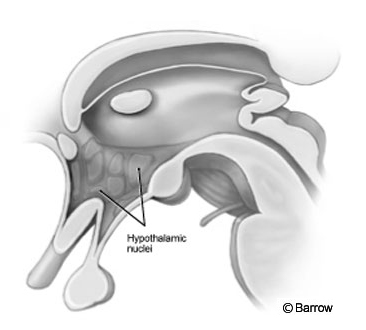
The size of these masses varies greatly as does their relationship to the various nuclei of the hypothalamus. The hypothalamus surrounds the third ventricle, and most of the nuclei of the hypothalamus contact the cerebrospinal fluid and ventricular wall (Fig. 1). We suspected that some patients would incur damage to various nuclei of the hypothalamus after surgery through the transcallosal interforniceal route and would exhibit a significant panhypopituitary state after surgery. This report discusses the preoperative endocrinological problems associated with these children; which, if any, endocrinological studies are needed preoperatively; and the initial endocrinological outcomes of the first 22 patients treated by The Hypothalamic Hamartoma Center at our institution.
Children with Precocious Puberty
As of this writing, 22 patients have undergone surgery for hypothalamic hamartomas through the Barrow Hypothalamic Hamartoma Program. All patients had intractable epilepsy and were treated surgically in the hope of alleviating their seizures and behavioral disturbances. Along with a routine neurosurgical evaluation, all patients underwent thorough preoperative testing, which included growth and development parameters and endocrinological evaluation. In a review of patients with precocious puberty, hypothalamic hamartomas were the cause of the disorder in 20% of the patients.[2] That rate was 18% in a study conducted by the National Institutes of Health.[10] As reported by their referring centers, more than half of our patients had precocious puberty as a comorbid finding. Patients’ histories, radiographic findings, and postoperative courses were reviewed, and recommendations for future endocrinological evaluations were made. The relationship between precocious puberty and hypothalamic hamartomas is well established.[1,2,7-9] Either surgical resection or administration of gonadotropin-releasing hormone (GnRH) antagonist has been the treatment for precocious puberty.
Pathophysiology
Normal puberty begins with the pulsatile release of GnRH by the hypothalamus. GnRH stimulates the release of luteinizing hormone (LH) and folliclestimulating hormone (FSH). In women FSH causes the ovarian follicles to develop, and LH acts on the follicles to cause their maturation and the secretion of estrogen. In men FSH promotes spermatogenesis, and LH stimulates androgen production by the testes. [4,11]
For girls puberty normally begins between the ages of 7 and 13 years. Secondary sexual characteristics become apparent at a mean age of 10 years (Tanner Stage 1 to 2). Female secondary characteristics include breast development, pubic hair, and an increase in height. These changes occur over the next few years after the onset of puberty and include the onset of menses. Puberty for boys begins between the ages of 9.5 and 13.5 years. Their secondary sexual characteristics include increasing size of the testes (Tanner Stage 1), the appearance of pubic hair, and growth of the penis. Complete maturation requires about 3 years.
Precocious, or early puberty, is defined as the onset of secondary sexual characteristics before the age of 8 years in girls and before the age of 9 to 9.5 years in males.[1,3,6] Precocious puberty also increases height, muscle mass, hunger, and personality changes. Precocious puberty is caused by peripheral, central, or mixed causes. Peripheral causes include congenital adrenal hyperplasia; familial male precocious puberty; McCune-Albright syndrome; or neoplasms of the ovary, testes, or adrenal glands. Central causes include disorders such as hydrocephalus, arachnoid cysts, septo-optic dysplasia, trauma, irradiation, neurofibromatosis, tuberous sclerosis, infections, or granulomas. Tumors that may also be included in the central category, include hypothalamic hamartomas, hypothalamic gliomas, germinomas, chiasmal gliomas, and craniopharyngiomas.[5,10]
Patient Population
The 22 patients included 15 boys and 7 girls (male-to-female ratio, 2:1). Their mean age was 10.2 years (age range,2-23 years). Eight of the 15 males and 4 of the 7 females became symptomatic with some sign of precocious puberty; the youngest in this group was 4.5 years. Symptoms included obesity, short stature (Pallister-Hall), and early secondary sexual characteristics.
Children were diagnosed with precocious puberty by referring centers because of deficiencies in growth hormone or unusual growth parameters. Three children were on a GnRH antagonist. One child was taking thyroid hormone. These children were taken off the medications by their referring center at the appropriate age to begin puberty.
Twenty transcallosal interforniceal approaches and two pterional orbitozygomatic approaches were performed. Surgical treatment of pedunculated hamartomas associated with precocious puberty has normalized endocrine function in a number of patients.[1,2]
Postoperative Endocrine and Hypothalamic Dysfunction
For 2 to 4 days after surgery, patients underwent serial serum sodium determinations every 2 to 6 hours. The levels of other hormones were checked 14 days after surgery. Six children had elevated sodium levels after surgery, three of whom were briefly given desmopressin acetate. Because these incidents were transient, they did not meet the criteria for diabetes insipidus. Only one patient had persistent diabetes insipidis. None of the other children are on new hormone-replacement therapy. The children are being followed for endocrinological assessment by their referring centers. In all patients thyroid function after surgery was within normal limits. When patients returned home to their own medical system, it was suggested that they undergo routine evaluations of their hormonal levels late in their course. Their serum sodium levels should be elevated in 1 to 2 months. We will also follow these patients when their laboratory and growth parameter results are made available.
Satiety, obesity, and thirst were addressed in our patients. Patients undergoing transcallosal interforniceal approaches often gain weight after surgery. The pattern, however, varies, and the weight gain usually stabilizes in the first 6 weeks after surgery. Two patients who continued to gain some weight beyond that point are being followed by their endocrinologists and pediatricians at home. During stressful times, children can have high sodium levels. Originally, this finding was attributed to diabetes insipidus. Now, however, it seems likely that derangement of the thirst mechanism must play some role in this phenomenon.
Conclusion
Fifty-eight percent of our patients with a hypothalamic hamartoma and intractable epilepsy also had precocious puberty. Surgical removal of the hamartoma normalizes the regulation of sexual development. The likelihood of residual hormonal dysfunction associated with this surgery tends to be low in patients followed for 6 months.
Preoperative testing is unlikely to lead to the discovery of unrecognized preoperative endocrinopathies that would affect the decision to pursue surgery. We therefore do not recommend extensive preoperative endocrinological testing. We do, however, recommend a careful, compulsive postoperative search for endocrinopathies such as diabetes insipidus, hypothyroidism, and hypocortisolemia. It is also important to be aware that after surgery these patients tend to have increased appetite, some weight gain, and a decreased sense of thirst. These changes may lead to hyper- or hyponatremia.
References
- Albright AL, Lee PA: Hypothalamic hamartomas and sexual precocity. Pediatr Neurosurg 18: 315-319, 1992
- Albright AL, Lee PA: Neurosurgical treatment of hypothalamic hamartomas causing precocious puberty. J Neurosurg 78:77-82, 1993
- Bates B, Bickley LS, Hoekelman RA: A Guide to Physical Examination and History Taking. Philadelphia: J.B. Lippincott, 1995
- Brownstein MJ: Neuropeptides, in Siegel GJ, Agranoff BW, Albers RW, et al (eds): Basic Neurochemistry. New York: Raven Press, 1994, pp 341-366
- DeGroot LJ: Endocrinology. Philadelphia: W.B. Saunders, 1995
- Gunn VL, Nechyba C: The Harriet Lane Handbook. A Manual for Pediatric House Officers. Philadelphia: Mosby, 2002
- Johnson DL, Jones B: Hemispheric malformations, in Albright AL, Pollack IF, Adelson PD (eds): Principles and Practice of Pediatric Neurosurgery. New York: Thieme, 1999, pp 143-164
- Luo S, Li C, Ma Z, et al: Microsurgical treatment for hypothalamic hamartoma in children with precocious puberty. Surg Neurol 57:356-362, 2002
- Lustig RH: The neuroendocrinology of childhood obesity. Pediatr Clin North Am 48:909-930, 2001
- Pescovitz OH, Comite F, Hench K, et al: The NIH experience with precocious puberty: Diagnostic subgroups and response to short-term luteinizing hormone releasing hormone analogue therapy. J Pediatr 108:47-54, 1986
- Tortora GS, Anagnotakos NP: Principles of Anatomy and Physiology. New York: Harper & Roe, 1984
