Gamma Knife Radiosurgery for the Treatment of Acoustic Neuromas
Andrew G. Shetter, MD
Division of Neurological Surgery, Barrow Neurological Institute, Mercy Healthcare Arizona, Phoenix, Arizona
Abstract
The Gamma Knife employs stereotactic techniques and 201 collimated Cobalt 60 sources to deliver a high dose of ionizing radiation to a small, precisely defined volume of tissue. There is increasing interest in utilizing this method as an alternative to microsurgical resection for the treatment of small to medium-sized acoustic neuromas. This article summarizes the published experience with Gamma Knife radiosurgery for acoustic neuromas, contrasting it with the natural history of the disease and the outcomes associated with open surgery. The author’s conclusions
regarding the current indications for Gamma Knife radiosurgery in acoustic neuroma patients are discussed.
Key Words : acoustic neuroma, Gamma Knife, stereotactic radiosurgery, vestibular schwannoma
The Gamma Knife was first used to treat an acoustic neuroma (vestibular schwannoma) in 1969.11 Since then this method of radiosurgery has been employed with increasing frequency, particularly within the last 5 to 10 years. By July 1996, approximately 5500 acoustic neuromas had been treated with Gamma Knife radiosurgery.9 An additional, but unknown, number have been treated with linear accelerator-based radiosurgical systems.
During this same period, major advances occurred in the microsurgical treatment of these relatively common neoplasms. Thirty years ago, the surgical removal of acoustic neuromas was often incomplete. Death was not rare and postoperative cranial nerve palsies were routine. Experienced teams of neurosurgeons and otologic surgeons now take justifiable pride in their ability to remove even large tumors completely, with a mortality rate of almost zero, a high likelihood of preserving facial nerve function, and some possibility of preserving residual hearing. Both neurosurgeons and radiosurgeons have benefited from dramatic improvements in neuroimaging techniques, which allow safe and easy diagnosis of these lesions when they are still small and not yet associated with major neurological deficits.
An intense debate is currently underway regarding the relative merits of radiosurgery and microsurgery in the treatment of acoustic neuromas and the future role of these competing technologies. This article reviews the results that have been reported with Gamma Knife radiosurgery to date and contrasts these outcomes to the natural history of acoustic neuromas and to the outcomes associated with their microsurgical treatment.
Gamma Knife Radiosurgery
Technique
Leksell10 originally defined stereotactic radiosurgery as the delivery of a high dose of ionizing radiation in a single fraction to a small, precisely defined volume of tissue. This approach differs from conventional radiotherapy, which uses a wide field of radiation administered in a low dosage and multiple fractions. Radiosurgery attempts to kill or alter all cells within the defined volume of the lesion and distributes the entrance and exit doses so that tissues outside the lesion are irradiated minimally. Radiotherapy delivers radiation equally to both normal and abnormal tissue and depends on a differential biological sensitivity to radiation for its therapeutic effect. The concepts behind these two methods are quite different, and they should not be confused.
In August 1987 the first 201-source 60Co gamma unit in the United States became operational at the University of Pittsburgh.15 This group has had extensive experience with acoustic neuromas, and they have been extremely diligent in publishing their results in peer-reviewed journals. Their initial technique, used to treat 26 patients, involved visualization of the tumor with high-resolution CT.12 A mean of 1.7 isocenters or shots per patient was used with collimators that were 4, 8, 14, and 18 mm in diameter. The mean marginal tumor dose was 19.75 Gy, usually delivered to the 50% isodose line.
Since then this group’s approach has evolved significantly, both in response to new technology and in an attempt to lower cranial nerve morbidity rates.16 They currently utilize multiplanar contrast-enhanced MR imaging to visualize a tumor, including volume acquisition with 1-mm thick slices in the axial plane.16 The mean number of isocenters is now six per patient with a preference for the smaller diameter collimators. Differential weighting of individual shots and the use of partial beam-blocking patterns further enhance the ability to shape the 50% isodose line to the true volume of the lesion. This more complex conformal planning is feasible because treatment-planning software and high-speed computers have improved. The marginal tumor dose also has been decreased from their initial 20 Gy to 13 to 15 Gy. These modifications in technique must be kept in mind when outcomes are evaluated because more recent reports by the Pittsburgh group are better than their earlier efforts.
Treatment Morbidity
Norén and colleagues19 at the Karolinska Institute in Sweden were the first to treat acoustic neuromas using the Gamma Knife. In 1992, they summarized their 23 years of experience with 325 acoustic neuroma patients.20 Adequate data with follow-up intervals of at least 12 months were available for 227 tumors. Facial weakness was noted 4 to 15 months after radiosurgery in 16% of patients, although it was said to be temporary in all instances. Slight trigeminal numbness was noted in 12% of the cases and more pronounced numbness in 8% of treated individuals. There was “slight” worsening in hearing in 53%, “severe” worsening or hearing loss in 23%, and stable or improved hearing in 24%. In their initial patients, the radiation dose delivered to the tumor periphery was 25 to 35 Gy. In later years, however, the dose was gradually decreased to 15 to 25 Gy.
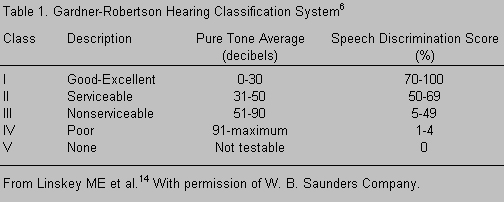
Working with numerous collaborators at the University of Pittsburgh, Drs. Lunsford, Kondziolka and Flickinger have acquired the largest contemporary experience with Gamma Knife radiosurgery for acoustic neuromas. Their ongoing efforts have been detailed in a variety of publications.4,12-14,16,22,23,25 As of 1995,16 they had treated 260 patients with tumors smaller than 30 mm in average dimensions. Larger lesions and those producing signs or symptoms of brain stem compression were excluded. After treatment with the improved techniques discussed above, 90% of their recently treated patients retained facial nerve function at a preoperative level. By the Gardner-Robertson Classification System (Table 1),6 Class I or II hearing was retained in 45% of patients who had serviceable hearing at the time of diagnosis, and hearing was preserved to some degree in 70% of the patients. Trigeminal neuropathy, frequently mild, occurred in fewer than 20% of the treated individuals.
The Pittsburgh group also analyzed their morbidity rates associated with treatment in subsets of patients with smaller tumors. From 1987 through 1990, they identified 20 patients with useful preoperative hearing (Gardner-Robertson Class I or II) from 115 patients with acoustic neuromas who underwent Gamma Knife radiosurgery.22 Immediately after treatment, serviceable hearing was preserved in 100% of the cases, in 50% at 6 months, and in 45% at both 1 and 2 years after treatment. If it did so, hearing deteriorated within 1 week to 1 year of treatment, and no further progression was observed after 2 years. The incidence of hearing preservation was greatest in patients with purely intracanicular tumors. Facial nerve function was normal in 85% of the patients 2 years after treatment, while 15% had mild or moderate weakness by the House-Brackmann7 facial nerve grading system (Table 2). Mild deficits of the sensory portion of the trigeminal nerve were seen in 25% of the patients 2 years after radiosurgery, but none developed a deafferentation pain syndrome.
This same group of investigators reviewed the results of treatment in 31 patients with tumors 10 mm or less in diameter.23 These patients were those considered to have the best chance of retaining cranial nerve function. At a 2-year follow-up examination, preoperative levels of facial nerve function had been achieved in all but two patients. Fifty percent of those with Class I or II Gardner-Robertson preoperative hearing function maintained useful hearing 2 years later. Two individuals reported subjective numbness or paresthesia of the trigeminal nerve.
Other radiosurgical teams have begun to report their experience with radiosurgery for acoustic neuromas, and their morbidity rates seem comparable to those reported by the University of Pittsburgh group. Yamasoba et al.27 evaluated 22 patients with some measurable hearing (Gardner-Robertson Classes I-V), including 13 patients who had useful hearing (Classes I-II) before undergoing Gamma Knife radiosurgery for their tumors. At a 2-year follow-up examination, 62% had retained useful hearing and 86% had measurable hearing function. From May 1991 through January 1994, Fukuoka et al.5 treated 43 patients with acoustic neuromas using a mean marginal tumor dose of 13.4 Gy delivered with the Gamma Knife. Useful hearing was preserved in 80% of the patients and the incidence of facial paresis was 2.3%.
Using a linear accelerator-based radiosurgery system rather than the Gamma Knife, the University of Florida radiosurgery team18 has treated 56 acoustic neuromas. Their patients were limited to those who were considered to be poor candidates for craniotomy because of a medical infirmity, tumor recurrence after prior surgery, or age greater than 65 years. Fifteen complications (23%) developed after treatment, including new onset of facial and/or trigeminal palsy (seven patients), worsening of preexisting facial and/or trigeminal deficits (five patients), and hydrocephalus requiring shunting (three patients). Auditory function was not assessed since none of their patients had serviceable preoperative hearing in the affected ear. The likelihood of complications was related to the volume of the tumor treated and the radiation dose.
Tumor Control Rates
Unlike microsurgery, which mechanically removes tumor tissue leaving behind a void, radiosurgery does not “cure” vestibular schwannomas. The tumor cells that remain after treatment may or may not have the capacity to grow at a later date. It is therefore more accurate to speak of tumor control rates over a specified period of time when considering the outcomes associated with radiosurgery. An imaging abnormality persists despite treatment,13 and serial MR images taken over an extended period are required to determine whether an individual tumor has the potential for mitotic activity.
Norén et al.21 have accumulated the longest follow-up data on acoustic neuromas treated with the Gamma Knife. In an abstract published in 1993, they described the outcomes of 40 patients treated between 1975 and 1980. Four patients were believed to have received a suboptimal dose of radiation and their tumors were subsequently removed surgically. Of the remaining cases, tumor control was achieved in 91.7% over a follow-up interval that averaged 123 months.
During the past 9 years, the University of Pittsburgh group has carefully documented their follow-up experience with acoustic neuromas. In 1991, they reported their findings on posttreatment MR images and CT scans in 88 patients.13 At a mean follow-up of 14.6 months, the size of 73% of the tumors was unchanged, 22% were smaller, and 4% had grown. Contrast enhancement of the central portion of the tumor was lost in 79%. A similar analysis was performed on 47 tumors treated with Gamma Knife radiosurgery during 1990 and 1991.25 The mean marginal tumor dose delivered during treatment was 16.3 Gy. The size of the lesion was unchanged in 60%, smaller in 34%, and larger in 6% at a median follow-up of 24 months (range, 3 to 46 months). None of the patients whose tumors had increased required further surgery. These investigators believe that their current tumor control rate after Gamma Knife radiosurgery is 92%.16 Almost all patients whose tumors progressed significantly did so within the first 2 years of treatment.
It must be remembered, however, that the marginal tumor doses delivered by Norén and by the Pittsburgh group during their early treatment years were substantially greater than the doses presently employed by most centers. Ten-year or longer follow-up data on a large number of patients with acoustic neuromas treated with contemporary radiosurgical techniques are not yet available.
Microsurgical Treatment
A full review of the many excellent papers discussing the microsurgical removal of acoustic neuromas is beyond the scope of this article. Some comparative data, however, are required to place the role of stereotactic radiosurgery in perspective. Translabyrinthine, retrosigmoid, and middle fossa surgical approaches all have been used with good success, depending upon the size of the lesion, the presence or absence of hearing function, and the individual preference of the surgeon. In experienced hands, the likelihood of a surgical cure using any of these three approaches is more than 90% for tumors less than 3 cm in diameter. This high cure rate constitutes the clearest advantage of microsurgery over radiosurgery. Conversely, radiosurgery has a clear advantage over microsurgery in avoiding the risks inherent to an open operative procedure. These include complications related to general anesthesia, infection, the formation of postoperative hematomas, cerebrospinal fluid (CSF) leak, and prolonged incisional pain. Death and brain stem infarction are rare using contemporary microsurgical techniques but they can occur.
The relative advantages of microsurgery and radiosurgery with respect to morbidity rates affecting the function of the trigeminal, facial, and cochlear nerves are less straightforward. Published microsurgical series3,8,24,26 have reported normal facial function in 60 to 90% of operated patients, depending primarily upon the preoperative size of their lesions. The rate of hearing preservation averaged 33% in a literature review performed by Gardner and Robertson in 1988,6 although individual series have achieved better results.26 Hearing outcome again varied with tumor size and the specific definition of useful hearing. The data on radiosurgery from the University of Pittsburgh group are summarized in Table 3. A rigorously fair comparison of these cranial nerve outcomes and those reported by microsurgical teams is impossible because of differences in selection criteria, a bias toward larger tumors in the open surgical series, and the lack of uniform criteria for evaluation.
Pollock et al.25 attempted to address this issue by analyzing the outcomes of 40 patients treated with microsurgery and 47 patients treated with radiosurgery at the University of Pittsburgh during 1990 and 1991. All tumors had a diameter of less than 3 cm, and the patients’ preoperative characteristics and the size of the tumors were considered similar in the two groups. The patients were not randomly selected, however, and the data were analyzed retrospectively. The long-term (median follow-up, 36 months; range, 25 to 48 months) likelihood of achieving normal facial function was 83% in the radiosurgery group and 63% in the microsurgery group. Serviceable hearing (Gardner-Robertson Class I or II) was present preoperatively in 21 of the patients who underwent microsurgery and in 8 of the patients who underwent radiosurgery. The long-term preservation rates of serviceable hearing were 14 and 75%, respectively, in the microsurgery and radiosurgery groups. The advantage of radiosurgery over microsurgery for facial paresis and hearing preservation was statistically significant in both instances. Permanent postoperative trigeminal nerve symptoms occurred in 11% of the microsurgery patients and in 14% of the radiosurgery patients, a difference that did not reach statistical significance. The direct hospital and professional charges for the two groups were 53% less in patients treated with radiosurgery. These figures, however, do not reflect the additional long-term costs that may accrue to patients treated with radiosurgery because of the need for more frequent follow-up MR imaging and the possibility of tumor progression that could require further treatment at a later date.
An individual operative series that does permit a comparative analysis is that of Ojemann.24 This respected and highly experienced surgeon removed 410 unilateral acoustic neuromas between 1979 and 1992. In 249 cases the maximum diameter of the tumors in the posterior fossa was less than 3 cm. In this subgroup, facial nerve function was normal in 70% and House-Brackmann Grade III or better in 96%. Patients with preoperative speech discrimination scores of at least 35% (Gardner-Robertson Class I to III) had a 60% chance of maintaining this level of function if the tumor was intracanalicular or if it extended less than 5 mm into the posterior fossa. If the tumor was 0.5 to 1.5 cm in diameter, the chance of preserving useful hearing was 35%. Hearing was rarely preserved in patients with tumors greater than 2.0 cm. Incomplete tumor removal was accepted in 4.4% of patients, usually because of their age or because the tumor adhered to the brain stem or facial nerve.
Natural History
The true natural history of acoustic neuromas is unknown. There is ample evidence, however, that these neoplasms grow slowly and that their growth may be arrested for years or perhaps indefinitely in a certain segment of patients. From 526 patients who presented with unilateral acoustic neuromas between 1983 and 1992, Deen et al.2 selected 68 patients for conservative treatment and follow-up with serial CT or MR imaging. At a mean follow-up of 3.4 years, 71% of the tumors demonstrated no growth and 29% had enlarged. There were no instances of tumor regression. In this highly select group of patients, the mean rate of tumor growth was 0.55 mm at the 2-year follow-up.
Bederson et al.1 treated 70 acoustic neuroma patients conservatively and followed them every 2 months with either CT or MR imaging for a mean of 26 months. The tumors grew in 53%, stayed the same in 40%, and decreased in 6%. The overall tumor growth rate was less than 2 mm/year.
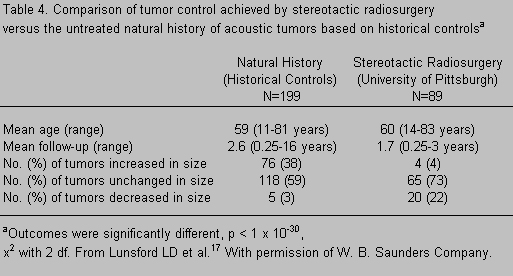
Because of such reports and their implications for assessing the outcome of Gamma Knife treatment, Linskey et al.13,17 reviewed the literature on the growth rates of untreated acoustic neuromas. They were able to identify 199 patients followed conservatively and described in 10 publications. The “natural history” of these untreated patients is contrasted with the tumor control rates seen in 89 patients who underwent radiosurgery for their tumors in Table 4. Although there are methodological problems in interpreting comparative studies of this type, it seems likely that stereotactic radiosurgery has a positive effect on rates of tumor control that exceed the natural history of the disease. A randomized prospective investigation that compares no treatment to radiosurgical treatment for small tumors with minimal symptoms has not yet been performed but would be of considerable interest.
Conclusion
Gamma Knife radiosurgery is an increasingly attractive alternative to microsurgery for the treatment of small to medium-sized acoustic neuromas. Improved dosimetry software, the use of high-resolution MR imaging for targeting the tumor, and a better understanding of the optimal radiation dose for a given tumor volume have all contributed to this trend.
The University of Pittsburgh group’s outcome statistics as of 1994 for preserving the function of the facial and cochlear nerves16 are equal, and possibly superior, to those reported by the very best microsurgical series (Table 3). Complications such as CSF leaks, infections, death, and general medical problems are avoided altogether. The short-term or perioperative costs for radiosurgery are considerably less than those associated with microsurgery, both in terms of direct expenses and the number of lost work days. As with any new technique, it will be necessary for other centers to confirm the excellent results of this pioneering group before such outcomes can be accepted unconditionally. There is no reason at present to believe this will not be the case.
The principle unknown regarding radiosurgery is the true incidence of tumor recurrence. The 92% tumor control rate observed by the University of Pittsburgh group refers primarily to patients followed for 5 years or less. Although they and Norén have rarely seen tumor growth beyond 2 years after treatment, we do not have representative data on large numbers of patients followed for many decades. Until this information becomes available, there will be legitimate differences of opinion about the respective indications for these two techniques.
Given our present state of knowledge, when should Gamma Knife radiosurgery be recommended for patients with acoustic neuromas? There is a clear consensus that tumors larger than 25 to 30 mm in diameter or those producing signs or symptoms of brain stem compression are not candidates for radiosurgery and should continue to be treated with microsurgical resection. For smaller, less symptomatic lesions, both radiosurgery and microsurgery should be considered.
Although some centers favor radiosurgery in all instances, I believe more restricted indications are in order until better long-term follow-up data are available. These indications include (1) patients older than 65 to 70 years with an enlarging tumor (elderly patients with stable symptoms and no evidence of tumor growth on serial MR imaging may be managed conservatively); (2) patients with major medical problems that significantly increase the risk of a general anesthetic; (3) patients with residual tumor after microsurgical resection by an experienced team or those whose tumors recur after having undergone a prior resection; and (4) patients who opt for radiosurgery instead of microsurgery after a comprehensive discussion with their physician about the advantages and disadvantages of both options. If tumor control rates remain greater than 90% in radiosurgically treated patients followed for 10 to 20 years, the indications for Gamma Knife radiosurgery will continue to expand.
References
- Bederson JB, von Ammon K, Wichmann WW, et al: Conservative treatment of patients with acoustic tumors. Neurosurgery 28:646-651, 1991
- Deen HG, Ebersold MJ, Harner SG, et al: Conservative management of acoustic neuroma: An outcome study. Neurosurgery 39:260-266, 1996
- Ebersold MJ, Harner SG, Beatty CW, et al: Current results of the retrosigmoid approach to acoustic neurinoma. J Neurosurg 76:901-909, 1992
- Flickinger JC, Lunsford LD, Linskey ME, et al: Gamma Knife radiosurgery for acoustic tumors: Multivariate analysis of four year results. Radiother Oncol 27:91-98, 1993
- Fukuoka S, Seo Y, Takanashi M, et al: Dizziness or imbalance occurring after Gamma Knife radiosurgery in cases of acoustic neurinomas. Brain Trust 1:12-13, 1996
- Gardner G, Robertson JH: Hearing preservation in unilateral acoustic neuroma surgery. Ann Otol Rhinol Laryngol 97:55-66, 1988
- House JW, Brackmann DE: Facial nerve grading system. Otolaryngol Head Neck Surg 93:146-147, 1985
- House WF, Hitselberger WE: The neuro-otologist’s view of the surgical management of acoustic neuromas. Clin Neurosurg 32:214-222, 1985
- Leksell Gamma Knife Society: Patients treated with the Leksell Gamma Knife Survey, June 30, 1996 (Abstract)
- Leksell L: Stereotaxis and Radiosurgery: An Operative System. Springfield, IL: Charles C Thomas, 1971
- Leksell L: A note on the treatment of acoustic tumours. Acta Chir Scand 137:763-765, 1971
- Linskey ME, Lunsford LD, Flickinger JC: Radiosurgery for acoustic neuromas: Early experience. Neurosurgery 26:736-745, 1990
- Linskey ME, Lunsford LD, Flickinger JC: Neuroimaging of acoustic nerve sheath tumors after stereotaxic radiosurgery. AJNR 12:1165-1175, 1991
- Linskey ME, Lunsford LD, Flickinger JC, et al: Stereotactic radiosurgery for acoustic tumors. Neurosurg Clin North Am 3:191-205, 1992
- Lunsford LD, Flickinger J, Lindner G, et al: Stereotactic radiosurgery of the brain using the first United States 201 cobalt-60 source Gamma Knife. Neurosurgery 24:151-159, 1989
- Lunsford LD, Kondziolka D, Pollock BE, et al: Gamma Knife stereotactic radiosurgery for acoustic tumors: What we have learned.Neurosurgeons 14:164-169, 1995
- Lunsford LD, Linskey ME: Stereotactic radiosurgery in the treatment of patients with acoustic tumors. Otolaryngol Head Neck Surg 25:471-491, 1992
- Mendenhall WM, Friedman WA, Buatti JM, et al: Preliminary results of linear accelerator radiosurgery for acoustic schwannomas. J Neurosurg 85:1013-1019, 1996
- Norén G, Arndt J, Hindmarsh T: Stereotactic radiosurgery in cases of acoustic neurinoma: Further experiences. Neurosurgery 13:12-22, 1983
- Norén G, Greitz D, Hirsch A, et al: Gamma Knife radiosurgery in acoustic neurinoma, in Steiner L (ed): Radiosurgery: Baseline and Trends. New York: Raven, 1992, pp 141-148
- Norén NG, Hirsch A, Mosskin M: Long-term efficacy of Gamma Knife radiosurgery in vestibular schwannomas (abstract). Acta Neurochir (Wien) 122:164, 1993
- Ogunrinde OK, Lunsford LD, Flickinger JC, et al: Stereotactic radiosurgery for acoustic nerve tumors in patients with useful preoperative hearing: Results at 2-year follow-up examination. J Neurosurg 80:1011-1017, 1994
- Ogunrinde OK, Lunsford LD, Flickinger JC, et al: Cranial nerve preservation after stereotactic radiosurgery for small acoustic tumors. Arch Neurol 52:73-79, 1995
- Ojemann RG: Management of acoustic neuromas (vestibular schwannomas). Clin Neurosurg 40:498-535, 1993
- Pollock BE, Lunsford LD, Kondziolka D, et al: Outcome analysis of acoustic neuroma management. A comparison of microsurgery and stereotactic radiosurgery. Neurosurgery 36:215-229, 1995
- Post KD, Eisenberg MB, Catalano PJ: Hearing preservation in vestibular schwannoma surgery: What factors influence outcome. J Neurosurg 83:191, 1995
- Yamasoba T, Kurita H, Ito K, et al: Auditory findings after stereotactic radiosurgery in acoustic neurinoma. Skull Base Surg 6:163-167, 1996