
Petroclival Meningiomas
Carlos A. David, MD*
Robert F. Spetzler, MD*
Division of Neurosurgery, University of Missouri, Columbia, Missouri
Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
Petroclival meningiomas are formidable surgical challenges. Surgical outcomes, however, have improved as neuroimaging and surgical approaches have advanced. The surgical anatomy associated with petroclival meningiomas and the skull base approaches best suited for their resection are reviewed.
Key Words: clivus, meningiomas, petrosectomy, surgical approaches, tumors
Meningiomas of the clivus and petrous apex remain formidable surgical challenges. Characterized by slow, relentless growth, these tumors can become enormous before they become apparent clinically. The involvement of the brain stem, cranial nerves, and critical vascular structures invariably makes these tumors fatal. Before the last decade, attempts at extirpation were associated with a mortality rate greater than 50%,[4,6,10,30] leading many surgeons to conclude that these tumors were incurable.
Advances in neuroimaging with concurrent refinements in surgical technique and the development of skull base surgery have improved surgical outcomes. These tumors, however, remain a substantial surgical challenge, and significant morbidity is still associated with their surgical removal. The most important surgical advance enabling acceptable morbidity rates has been the development of approaches that maximize surgical exposure while minimizing the extent of brain retraction. Several surgical approaches, which can be used by themselves or in combination, have been devised to maximize exposure from different vantage points. The selection of a particular approach or combination is based on careful consideration of the size, location, and extent of the lesion as well as on the patient’s neurological status. This article reviews the various approaches and their appropriate selection as well as the complications, management, and outcomes associated with the treatment of petroclival meningiomas.
Surgical Anatomy
The clivus is the segment of the skull base that adjoins the foramen magnum to the sphenoid bone. Otherwise known as the basilar part of the occipital bone (basiocciput), this thick plate of bone extends rostrally at an angle of 45° from the foramen magnum to the dorsum sellae. Early in life an articulation exists between these two regions known as the sphenoid occipital synchondrosis; this articulation usually disappears by adulthood. Laterally, the clivus meets the petrous portion of the temporal bone at the petro-occipital fissure. Together, these three bones form the skull base and middle fossa floor. The entire region is lined with a double layer of dura consisting of the endosteal and periosteal layers. Between these two layers are found various venous sinuses and plexi.
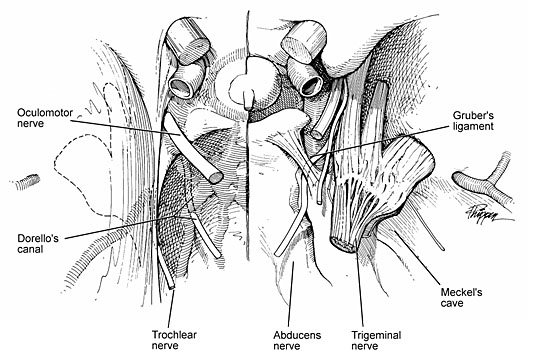
The concave intracranial surface of the clivus is lined by a thick double layer of dura that contains the basilar venous plexus. This venous plexus forms a venous confluence with the posterior cavernous sinus and the inferior petrosal sinus, which contains an osteofibrous compartment known as Dorello’s canal.[29] This canal is found at the region of the petrous apex between the two dural leaves. The abducens nerve lies within this canal, coursing toward the cavernous sinus below a strong fibrous trabecula known as the petrosphenoidal ligament or Gruber’s ligament (Fig. 1). Slightly lateral to this region, along the petrous apex, is the trigeminal groove or impression. With its dural lining, this region forms Meckel’s cave, the entrance of the trigeminal nerve. The trigeminal nerve, surrounded not only by a dural sheath but also by a well-developed cistern,[24] enters this region from the posterior fossa. As the trigeminal nerve travels anteriorly, it leaves the cistern, and the individual divisions of the nerve become surrounded by dural sheaths as they travel along the middle fossa floor to their respective exiting foramina.
The petrous portion of the temporal bone houses the carotid artery as it ascends in its vertical portion before its anteromedial bend. In addition, the sigmoid sinus, jugular bulb, vestibulocochlear apparatus, and facial nerve are encased within the mastoid bone.
Classification
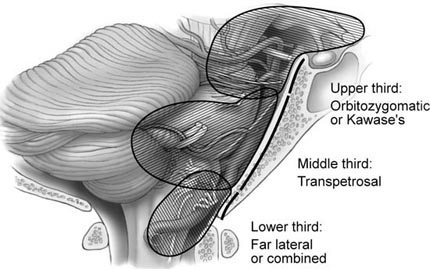
No ideal scheme exists for classifying petroclival meningiomas. An overall classification scheme would encompass tumor location as well as assess technical difficulty and choice of approach. Sekhar et al. [22] have developed a useful scheme that is based on the tumor’s anatomical location along the clivus, mainly along the upper, middle, or lower clivus. A similar scheme based on both anatomical location and tumor extent permits more rational decision making regarding the optimal surgical approach. The tumors are divided into three areas (Fig. 2). Tumors of the upper third involving the petrous apex and posterior cavernous sinus are best approached via an orbitozygomatic or Kawase’s approach. Tumors of the middle clival level involving the caudal extent of the internal auditory meatus to the jugular foramen are best approached via a transpetrosal route. Tumors of the lower clivus down to the foramen magnum are best approached by the far-lateral approach or combination approaches.
Surgical Approaches
Because petroclival meningiomas occupy difficult locations, a number of surgical approaches have been proposed for their removal. [1,5,9,11,14,16,21,23,25] Understanding these lesions and their surgical anatomy and realizing the need to maximize exposure without brain retraction have been the driving forces behind the development of various skull base approaches. A number of excellent approaches for dealing with lesions in this location are now available to surgeons.
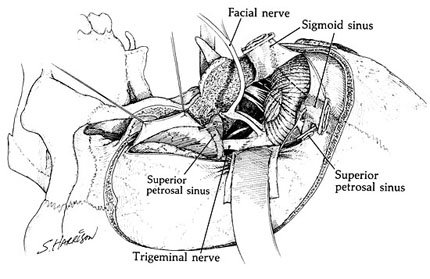
The selection of the approach primarily reflects the location of the meningioma in the petroclival region. The size of the tumor and its rostral-caudal extent along the clivus are factors that determine the surgical exposure needed (Fig. 3). The patient’s neurological status as well as the goals of surgery must be considered. The goals of surgery should be radical removal when possible, particularly during the first operation. However, if critical vascular structures are encased by tumor or if important venous structures or the brain stem is involved, a subtotal resection and less extensive approach may be necessary.
Traditionally, tumors in this location have been approached via a subtemporal route or a lateral suboccipital approach. In an effort to reach lesions with caudal extensions into the posterior fossa, the subtemporal-transtentorial route was developed.[20] Malis [15] proposed combining the subtemporal route with the suboccipital approach to gain access to the clivus and medial petrous origin. Modifications of this approach incorporating petrosectomy options have led to the development of the combined supra- and infratentorial transpetrosal approaches.[18,27,28] This approach, with its various options for petrous bone removal, can provide an extensive view of the entire petroclival region.
Small tumors located along the upper clivus with no significant infratentorial extension have been approached via frontotemporal-transsylvian routes.[8,17] A recent modification, which involves the addition of an orbitozygomatic osteotomy, has significantly enhanced the exposure of tumors in this region.[13]
Combined Approach
The combined supra- and infratentorial transpetrosal approach employs various degrees of petrous bone resection with a subtemporal-suboccipital craniotomy. It thereby exposes both the supra- and infratentorial compartments, which are then connected via division of the tentorium (Fig. 3). Because it allows extensive exposure of the petroclival region, this approach has become the main route for removing tumors.
The patient is positioned supine with the head turned opposite the side of the lesion. The head is maintained parallel to the floor with the vertex axis inclined downward a few degrees. The head is held in position using a Mayfield three-pin head holder. A gel roll is placed under the ipsilateral shoulder to provide support and to decrease neck tension that could impede venous outflow.
Beginning anterior to the ear within 1 cm of the tragus at the level of the zygoma, the skin is incised. The incision is carried around the ear and continues to just below the mastoid tip. The scalp flap is then retracted inferiorly with fishhooks attached to a Leyla bar.[26] The temporal muscle is retracted over the scalp flap, and the sternomastoid muscle is detached and reflected inferiorly. In this manner, the lateral temporal bone, the zygoma, external auditory meatus, mastoid, and suboccipital regions are exposed.
The otologist may perform the petrous bone resection at this point or after the neurosurgeon has turned the craniotomy flap. The mastoidectomy or posterior petrosectomy is performed using a high-speed drill, initially without magnification, but later under the operating microscope for more detailed bone removal near critical structures.
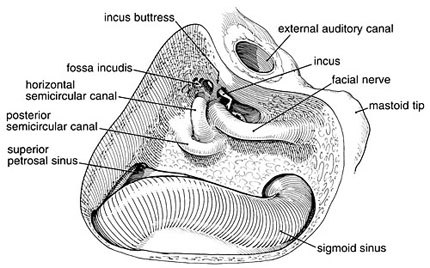
Initially, the cortical bone of the mastoid is removed, delineating a triangular region bounded by the linea temporalis superiorly, the mastoid tip inferiorly, and the posterior wall of the external auditory meatus anteriorly. The first step clearly defines the sigmoid plate and sinodural angle. Proceeding through the superior air cells along the posterior canal wall leads to the mastoid antrum. The medial floor of the antrum is the location of the lateral semicircular canal. This structure becomes evident as cortical bone within the antrum.
Further drilling along the inferior air cells locates the digastric ridge whose anterior limit ends at the stylomastoid foramen where the facial nerve exits. At this point, if needed, the semicircular canals can be better defined. The lateral canal, which has already been identified, is delineated along its posterior limit. In this region the posterior semicircular canal parallels the posterior fossa plate. Its superior end joins the superior semicircular canal at the common crus. The course of the facial nerve within the fallopian canal runs between the inferior edge of the lateral canal and the stylomastoid foramen parallel to the posterior auditory canal wall (Fig. 4).
Depending on the petrosectomy option employed, [28] these structures can be drilled further (see below). Once this drilling has been completed, the drilling of the sigmoid plate is completed and followed inferiorly to the jugular bulb. With the middle fossa plate, posterior fossa plate, and labyrinth exposed, drilling is completed along this presigmoid area (Trautmann’s triangle) following the petrous ridge medially, drilling off the amount needed, and completing the posterior petrosectomy.
At this juncture the craniotomy is completed. A subtemporal-suboccipital craniotomy is then fashioned exposing the transverse sinus and transverse-sigmoid junction. A three-limb dural incision is begun in the presigmoid region. The incision extends from the superior limit of the jugular bulb to the superior petrosal sinus. A second limb is made along the temporal base to the transverse-sigmoid junction. The final limb divides the superior petrosal sinus and extends along the tentorium anteriorly to a point just posterior to the trochlear nerve. If greater exposure is needed, the sigmoid sinus can be ligated and divided. This maneuver can be performed safely only after confirming that the sigmoid sinus communicates freely with the contralateral sigmoid sinus and superior sagittal sinus via the torcula.
Intraoperative pressure measurements can help determine the safety of sacrificing the sigmoid sinus. After the petrosal sinus has been ligated, a 25-gauge needle is used to measure sigmoid sinus pressure after temporary ligation. The sigmoid sinus can be divided only if the pressure does not increase more than 10 mm Hg.[28] Typically, ligation of the sigmoid sinus is unnecessary because a second incision placed behind the sigmoid sinus allows the surgeon to work around the sinus in the same area.
The closure requires meticulous attention because the risk of a cerebrospinal fluid (CSF) fistula is high. The dural opening should be closed when possible with 4-0 nylon suture. The defect created in the petrous bone should be filled with either muscle or abdominal fat in addition to fibrin glue. The eustachian canal should be packed with fat. Spinal drainage is used 3 to 5 days.
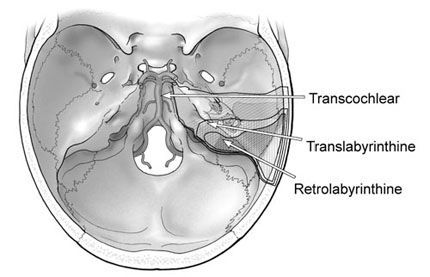
Petrosectomy Options
The petrosectomy options include a retrolabyrinthine resection, which preserves hearing but provides a very small avenue in which to work. The translabyrinthine technique sacrifices hearing and provides a somewhat larger working area. The transcochlear technique, which provides the largest working area via maximal petrous resection, sacrifices hearing and transposes the facial nerve (Fig. 5).
Retrolabyrinthine Petrosectomy. To preserve hearing the integrity of the labyrinth is maintained. In addition to skeletonizing the lateral semicircular canal as already described, the posterior and superior canals are also skeletonized. Drilling is taken as far anterior as possible, exposing the maximal area of dura. This route provides good access to the cerebellopontine angle and lower clivus but is limiting along the anterior brain stem and more rostral clivus.
Translabyrinthine Petrosectomy. The translabyrinthine approach, which sacrifices hearing, is accomplished via the same basic mastoidectomy already described, but the semicircular canals are also removed. The three canals are skeletonized and drilled away. Drilling then continues forward so that the posterior aspect of the internal auditory canal also can be skeletonized. The bone overlying the labyrinthine and mastoid segments of the facial nerve is also thinned. These maneuvers should be performed under continuous facial nerve monitoring.
Transcochlear Petrosectomy. This technique provides the maximal area of exposure by completely removing the petrous bone. The external auditory canal is transected and oversewn. The initial drilling is as described for the translabyrinthine technique. The facial nerve canal is completely skeletonized and opened to allow the removal of the facial nerve. The greater superficial petrosal nerve is divided to permit the posterior transposition of the facial nerve. This maneuver then allows the complete removal of the internal auditory canal and cochlear apparatus. Drilling is continued until the internal carotid artery has been skeletonized up to the siphon.
This technique provides extensive exposure of the petroclival region at the cost of sacrificing hearing and facial nerve paresis or paralysis. This route is also associated with a significant risk of CSF fistula.
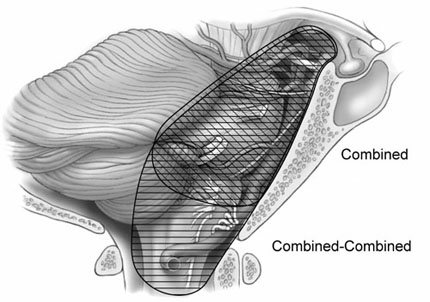
Combined-Combined Approach
The combined-combined approach is useful when lesions extend from the entire clivus to the foramen magnum making their removal via a single approach impossible. Visualization can be improved by combining the far-lateral approach with the combined supra- and infratentorial transpetrosal approach. [3] This combination is referred to as the “combined-combined” approach. These exposures can provide extremely wide views of the entire clival region down to the upper cervical region. Again, the choice of approach is based on the location and extent of the tumor (Fig. 6).
The combined-combined approach is performed as already described for the combined approach. The skin incision is modified to include the exposure needed for the far-lateral approach. Beginning anterior to the ear within 1 cm of the tragus at the level of the zygoma, the skin is incised. The incision continues around the ear, to the midline, and down to the level of C3-C4. A cuff of muscle and fascia is left along the superior nuchal line for later reapproximation of the muscle. The soft tissues are dissected to expose the lateral temporal bone, zygoma, mastoid region, and external auditory meatus.
The petrosectomy is then performed and is followed with the far-lateral exposure. The temporal craniotomy can be performed as part of the suboccipital craniotomy or as a separate section.
The dural incision is begun by opening over the temporal lobe parallel to the middle fossa floor and extending across the superior petrosal sinus to join a second incision made anterior to the sigmoid sinus. The petrosal sinus is clipped and cauterized before it is divided. The dural incision is continued through the tentorium behind the trochlear nerve to the tentorial incisura connecting the supra- and infratentorial spaces. The dural opening for the far-lateral approach is also performed. If the sigmoid sinus is to be sacrificed, the two dural incisions are joined providing a panoramic view of the entire clivus and anterior brain stem. Closure follows the same principles described for the other approaches.
Subtemporal Approach with Anterior Petrosectomy (Kawase’s)
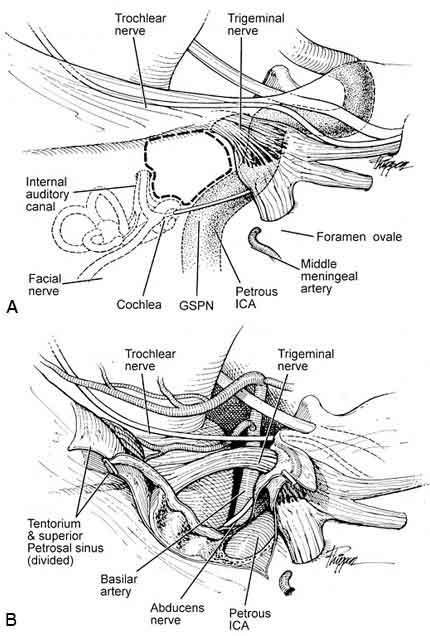
Lesions confined to the petrous apex and upper half of the clivus also can be approached via an anterior petrosectomy. [12,18] Furthermore, this approach is useful when lesions involve the posterior cavernous sinus. The approach is made through a subtemporal craniotomy. The initial approach is extradural with identification and ligation of the middle meningeal artery. This maneuver allows further extradural dissection exposing the greater and lesser petrosal nerves and the mandibular division of the trigeminal nerve as it travels to the foramen ovale (Fig. 7A). The greater superficial petrosal nerve is sacrificed to avoid traction on the facial nerve.
The petrous carotid artery is identified medial to the third division of the trigeminal nerve at the foramen ovale. It is often incompletely covered by dehiscent bone or cartilage. In addition to these landmarks, the arcuate eminence, which overlies the superior semicircular canal, must be identified. It lies perpendicular to the petrous ridge forming a 120° angle with the greater superficial petrosal nerve. [18] The internal auditory canal lies between these two structures at about a 60° angle to the superior semicircular canal.
Initially, drilling is begun to expose the carotid artery if it is not completely visible. Using a high-speed diamond drill, bone is removed along the medial border of the greater superficial petrosal nerve until the artery is visible. Drilling continues anterior to the arcuate eminence and proceeds posteriorly to identify the anterior border of the superior semicircular canal. Once this structure is identified, the drilling is performed along the midportion of the expected position of the internal auditory canal. Once the internal auditory canal has been identified, the drilling is expanded and carried medially to expose the canal and posterior fossa dura. The drilling proceeds laterally only to begin to expose the vertical crest (Bill’s bar). The cochlear apparatus lies in the angle between the internal auditory canal and the greater superficial petrosal nerve beneath the region of the geniculate ganglion. This landmark constitutes the posterolateral limit of the drilling to avoid exposing the cochlea and damaging hearing.
At this point, drilling can be continued in the rhomboid region delimited by the mandibular division of the trigeminal nerve and the internal carotid artery anteriorly, the greater superficial petrosal nerve laterally, the internal auditory canal and superior semicircular canal posteriorly, and the petrous ridge medially. This region, known as Kawase’s triangle, can be safely removed continuing inferiorly along the posterior fossa dura to the inferior petrosal sinus.
With the drilling completed, the dura along the inferior temporal lobe is opened. The tentorium is divided posterior to the trochlear nerve, and the superior petrosal sinus is ligated and divided extending into the posterior fossa. The exposure provides a good view of the petrous apex, middle fossa, and posterior fossa regions (Fig. 7B).
To widen this exposure for lesions that involve the posterior cavernous sinus extensively, Fukushima and colleagues [7] described a modification to this approach. In this modification, the foramina ovale and rotundum are drilled more aggressively. Consequently, the trigeminal nerve can be mobilized anteromedially; in turn, the petrous apex can be removed completely under direct vision.
Although these approaches provide good working room, the risks of CSF fistula, hearing loss, and injury to the carotid artery and cranial nerve are significant. We have found that lesions limited to the petrous apex can be approached via a frontotemporal orbitozygomatic approach with considerably fewer complications.
Frontotemporal-Orbitozygomatic Approach
First described by Pellerin et al. [19] and Hakuba et al., [8] the orbitozygomatic approach has gained wide popularity for approaching lesions located at the upper clivus, sella turcica, and petrous apex. When combined with a pterional craniotomy, this approach provides extensive exposure of deep-seated lesions without excessive brain retraction. It offers improved working angles and multidirectional routes of exposure with minimal morbidity.
The patient is positioned supine with the head rotated 30° to 60° contralaterally. The degree of rotation is dictated by the location of the lesion. For lesions located in the petroclival area, upper clivus, and posterior fossa, the head should be turned minimally. Greater degrees of turning are beneficial for lesions in the middle and anterior cranial fossae. The neck is extended until the malar eminence is the most superior point of the operative field. The head is then secured in a three-point fixation device.
The scalp incision begins within 1 cm of the tragus at the level of the inferior border of the zygoma. It extends superiorly and then curves forward terminating behind the hairline at the contralateral midpupillary line. Care must be exerted with the most inferior point of the incision because the facial nerve is located approximately 1.5 to 1.7 cm below the zygoma near the mandibular condyle. [2] The scalp is then elevated exposing the temporalis fascia anteriorly until the fat pad just begins to come into view. Further elevation can injure the frontalis branch of the facial nerve, which lies within this fat pad. [2]
The temporalis fascia is incised in the line of the skin incision to a point just inferior to the superior temporal line. The fascial incision is carried forward leaving a myofascial cuff attached to the bone for later reapproximation. [26] This fascia is then elevated from the underlying muscle and dissected anteriorly to expose the zygoma, malar eminence, and supraorbital rim. Next, the temporalis muscle is incised along the fascial edges and elevated from the underlying skull. With the scalp flap and temporalis muscles elevated and retracted inferiorly with fishhooks on a Leyla bar, the periorbita is dissected from the orbital walls. This dissection begins at the lateral edge of the orbital rim and proceeds medially while the periorbita is separated gently from the lateral and superior orbital walls. The medial limit of the dissection is the supraorbital nerve. If more medial exposure is needed, the supraorbital nerve can be freed from its bony canal. When this maneuver is completed, the entire orbital rim should be exposed to the malar eminence of the zygoma.
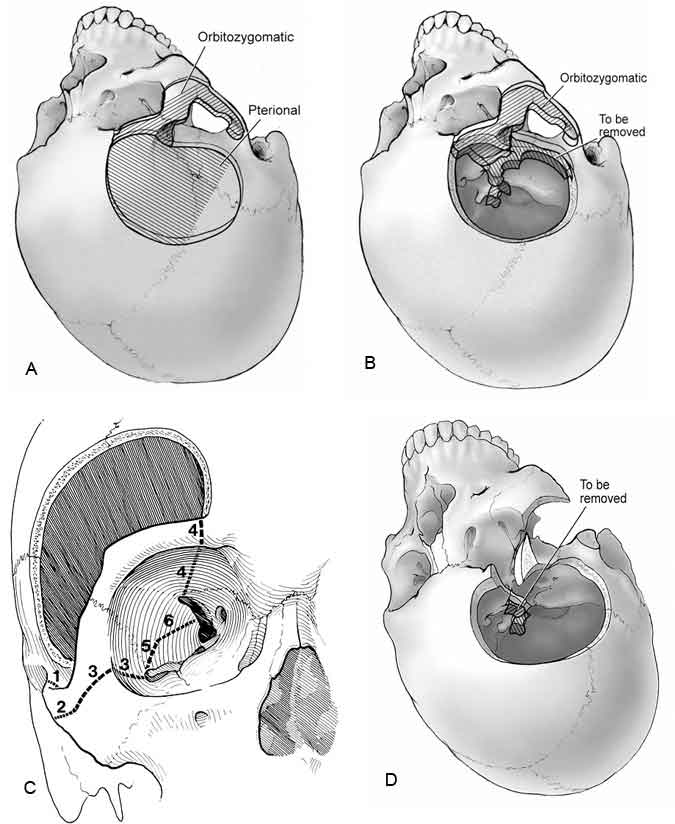
A standard pterional craniotomy is fashioned using standard drills (Fig. 8A). Once the pterional craniotomy has been completed, the temporalis muscle is brought back over the dura so that the zygoma can be visualized. A reciprocating saw is used to perform the orbitozygomatic osteotomy (Fig. 8B and C). This first cut is made at the base of the zygomatic arch on the temporal bone. The second cut begins at the inferolateral edge of the zygoma near the malar region and proceeds halfway along the malar eminence toward the lateral orbital rim. A third cut begins intraorbitally in the inferior orbital fissure and extends posterolaterally until it meets the second saw cut.
At this point, the dura is elevated from the orbital roof and anterior temporal fossa. The fourth cut begins intraorbitally 1 to 2 mm lateral to the supraorbital notch and proceeds posteriorly along the orbital roof about 3 to 4 cm and medially to the superior orbital fissure. A fifth cut is made from the inferior orbital fissure posteriorly to the temporal region. A final cut is made from the superior orbital fissure inferiorly to meet the former cut. With these maneuvers completed, the entire orbitozygomatic bone is free and can be removed. The remaining sphenoid wing is then thinned carefully and removed as in a standard pterional craniotomy.
The temporalis muscle is retracted inferolaterally to a greater degree than possible with the zygoma present. The dura is opened in the standard fashion and held in place inferiorly while the orbital contents are retracted to provide a wide, unrestricted view. If needed, the posterior clinoid as well as the medial petrous apex can be removed (Fig. 8D).
With the intradural portion of the operation completed, the standard closure is performed by fixating the bone flap and orbitozygomatic bone with miniplates. Anatomic alignment must be obtained to avoid cosmetic defects. The closure is completed in the usual manner.

Combined-Combined Orbitozygomatic Approach
Large lesions that extend from the petroclival region caudally to the lower clivus and rostrally into the parasellar region can pose a formidable challenge requiring multiple operations and approaches. To gain additional exposure for such tumors, we have combined the approaches described above (i.e., the supra- and infratentorial transpetrosal approach and frontotemporal-orbitozygomatic approach). All three cranial fossae are exposed, and multiple vantage points are available for tumor dissection and removal (Fig. 9). [13]
This approach is indicated for large extensive lesions involving the petroclival region, extending upward and contralaterally in the middle fossa and down into the lower third of the clivus. The approach is performed as described above for the individual techniques. A combined supra- and infratentorial transpetrosal approach is performed first, and the dura is exposed but not opened. The frontotemporal craniotomy and orbitozygomatic osteotomy follow next. The dura is then incised as for each separate approach joining in the region of the temporal dura to form the shape of a 3. The superior petrosal sinus is ligated and divided along with the tentorium as described previously.
Closure follows the standard closure for each separate approach. The exposure obtained with this approach is impressive, allowing visualization from the anterior fossa to the lower clivus. Although not needed routinely, this approach can be useful with the rare extensive skull base lesion.
Technique for Meningioma Removal
After the dura and arachnoid have been opened to permit CSF drainage and relaxation of the brain, the tumor is exposed. The surgeon must orient himself or herself because the normal appearance of the anatomy can be significantly distorted. Tumor removal proceeds according to general neurosurgical principles. Primarily, early devascularization is combined with central debulking. After the initial debulking, dissection proceeds along the tumor capsule. Dissection must remain within the arachnoid planes if the tumor is to be separated successfully from critical vascular and neural structures.
A combination of suction, bipolar coagulation, microscissors, and ultrasonic aspiration is used for the major debulking. During the debulking, the surgeon must maintain a clear mental picture of the expected anatomy and possible location of displaced structures. In particular, the abducens nerve and the basilar artery can be embedded within the tumor and difficult to identify. Tumor debulking proceeds from less critical superficial areas to deeper regions where critical structures may be found.
After generous debulking and relaxation, the tumor capsule is dissected free from the surrounding structures. Meticulous care must be taken to maintain the dissection within the arachnoid planes however difficult it may be to achieve this goal. Frequently, one must alternate between further debulking and capsule removal. When most of the tumor has been debulked, the thinned-out capsule is carefully dissected free of surrounding structures.
With large extensive tumors, the lower cranial nerves are often involved along the lower pole of the tumor. The facial and vestibulocochlear nerves are usually displaced dorsally and inferiorly. Dissection along these nerves can be enhanced with the use of monitoring and a facial nerve stimulator. The abducens nerve is typically within the tumor or stretched anteriorly. It is best to identify this nerve proximally and follow it distally into its dural entrance. Similarly, the trigeminal and trochlear nerves are identified. Occasionally, the dura over Meckel’s cave is opened to remove tumor that has grown into this region.
The basilar artery is usually within the tumor or displaced contralaterally. Utmost care must be exercised when dissecting this vessel from the tumor capsule, particularly its critical perforator vessels. It is often wiser to leave a remnant of firmly attached capsule to the basilar artery than risk neurological devastation from injuring the perforators or the basilar artery itself.
Complications
The surgical removal of petroclival meningiomas is associated with a significant risk of neurological morbidity. The confined space in association with the critical vascular and neural structures makes the potential for injury to these structures quite high. Specific complications are associated with each specific approach; however, the more important complications can be subdivided into vascular injuries, cranial nerve injuries, and general complications.
Vascular complications can occur during tumor debulking and dissection of the tumor capsule. As mentioned, perforators from the basilar artery can be quite difficult to identify and separate from the tumor, particularly when a large petroclival meningioma encases the basilar artery.
Cranial nerves are at significant risk during the removal of petroclival meningiomas. The trochlear nerve is often injured due to its size and fragility. The resulting defect, however, is relatively minor compared to those associated with other cranial nerve injuries. Although the trigeminal nerve is more robust than other cranial nerves, its injury can lead to complications such as keratitis from corneal anesthesia, facial pain, and weakness and atrophy of the temporalis and masseter muscles, which can lead to difficulty with chewing. The abducens nerve is frequently encased by tumor and therefore at high risk. Injury causes diplopia. The facial nerve is at risk during tumor removal but also may be affected by the particular approach employed. Transposition during a transcochlear approach causes facial paralysis, which is usually temporary. The vestibulocochlear nerve and lower cranial nerves can be injured by large tumors with significant caudal extension.
Other complications particularly related to the surgical approach include CSF leaks, hydrocephalus, and pseudomeningocele. CSF leakage with meningitis is a potential complication during resection of these tumors, particularly in the presence of preoperative hydrocephalus. It occurs more commonly with the translabyrinthine and transcochlear petrosal approaches. To lower the risk of CSF leakage, the dural and soft tissue closures must be meticulous. A good dural closure with fat grafting, if needed, and a tight fascial and skin closure should be ensured.
Between 1986 and 1993, 32 patients with petroclival meningiomas were treated at our institution. The rate of total tumor removal was 87%. The incidence of facial nerve paralysis was approximately 28%. CSF leaks occurred in 11% and abducens palsy in about 9%. The incidence of other general complications was 2 to 4%.
Conclusions
Meningiomas of the petroclival region continue to pose a difficult surgical challenge. The development of skull base approaches and their combinations has provided a safer, more effective venue for surgeons to deal with these lesions adequately. Despite these advances, the morbidity rate associated with total removal of these lesions remains high. The remaining challenge is to reduce this morbidity rate by more conservative applications of skull base approaches: attaining maximal tumor removal while preserving neurological function.
References
- Al-Mefty O, Fox JL, Smith RR: Petrosal approach for petroclival meningiomas. Neurosurgery 22:510-517, 1988
- Ammirati M, Spallone A, Ma J, et al: An anatomicosurgical study of the temporal branch of the facial nerve. Neurosurgery 33:1038-1044, 1993
- Baldwin HZ, Miller CG, van Loveran HR, et al: The far lateral/combined supra- and infratentorial approach. A human cadaveric prosection model for routes of access to the petroclival region and ventral brain stem. J Neurosurg 81:60-68, 1994
- Cherington M, Schneck SA: Clivus meningiomas. Neurology 16:86-92, 1966
- Crockard HA, Sen CN: The transoral approach for the management of intradural lesions at the craniovertebral junction: Review of 7 cases. Neurosurgery 28:88-98, 1991
- Cushing H, Eisendhardt L: Meningiomas: Their Classification, Regional Behaviour, Life History, and Surgical End Results . New York: Hafner, 1938
- Fukushima T, Day JD, Hirahara K: Extradural total petrous apex resection with trigeminal translocation for improved exposure of the posterior cavernous sinus and petroclival region. Skull Base Surgery 6:95-103, 1996
- Hakuba A, Liu S, Nishimura S: The orbitozygomatic infratemporal approach: A new surgical technique. Surg Neurol 26:271-276, 1986
- Hakuba A, Nishimura S, Jang BJ: A combined retroauricular and preauricular transpetrosal-transtentorial approach to clivus meningiomas. Surg Neurol 30:108-116, 1988
- Hakuba A, Nishimura S, Tanaka K, et al: Clivus meningiomas: Six cases of total removal. Neurol Med Chir 17:63-77, 1977
- House WF, Hitselberger WE: The transcochlear approach to the skull base. Arch Otolaryngol 102:334-342, 1976
- Kawase T, Shiobara R, Toya S: Anterior transpetrosal-transtentorial approach for sphenopetroclival meningiomas: Surgical methods and results in 10 patients. Neurosurgery 28:869-876, 1991
- Lawton MT, Daspit CP, Spetzler RF: The orbitozygomatic-combined supra- and infratentorial approach: Technical note. BNI Quarterly 12:4-9, 1996
- Long DM: Surgical approaches to tumors of skull base: An overview, in Wilkins RH, Rengachary SS (eds): Neurosurgery Update I. Diagnosis, Operative Techniques, and Neuro-Oncology . New York: McGraw-Hill, 1990, pp 266-276
- Malis LI: Surgical resection of tumors of the skull base, in Wilkins RH, Rengachary SS (eds): Neurosurgery. Vol. I. New York: McGraw-Hill, 1985, pp 1011-1021
- Mayberg MR, Symon L: Meningiomas of the clivus and apical petrous bone. Report of 35 cases. J Neurosurg 65:160-167, 1986
- McDermott MW, Durity FA, Rootman J, et al: Combined frontotemporal orbitozygomatic approach for tumors of the sphenoid wing and orbit. Neurosurgery 26:107-116, 1990
- Miller CG, van Loveren HR, Keller JT, et al: Transpetrosal approach: Surgical anatomy and technique. Neurosurgery 33:461-469, 1993
- Pellerin P, Lesoin F, Dhellemmes P, et al: Usefulness of the orbitofrontomalar approach associated with bone reconstruction for frontotemporosphenoid meningiomas. Neurosurgery 15:715-718, 1984
- Rosomoff HL: The subtemporal transtentorial approach to the cerebellopontine angle. Laryngoscope 81:1448-1454, 1971
- Samii M, Ammirati M, Mahran A, et al: Surgery of petroclival meningiomas: Report of 24 cases. Neurosurgery 24:12-17, 1989
- Sekhar LN, Jannetta PJ, Burkhart LE, et al: Meningiomas involving the clivus: A six-year experience with 41 patients. Neurosurgery 27:764-781, 1990
- Sekhar LN, Jannetta PJ, Maroon JC: Tentorial meningiomas: Surgical management and results. Neurosurgery 14:268-275, 1984
- Sen C, Chen CG, Post KD: Microsurgical Anatomy of the Skull Base and Approaches to the Cavernous Sinus . New York: Thieme: 1997
- Sen CN, Sekhar LN: An extreme lateral approach to intradural lesions of the cervical spine and foramen magnum. Neurosurgery 27:197-204, 1990
- Spetzer RF: Two technical notes for microsurgery. BNI Quarterly 4:38-39, 1988
- Spetzler RF, Daspit CP, Pappas CTE: The combined supra- and infratentorial approach for lesions of the petrous and clival regions: Experience with 46 cases. J Neurosurg 76:588-599, 1992
- Spetzler RF, Hamilton MG, Daspit CP: Petroclival lesions. Clin Neurosurg 41:62-82, 1994
- Umansky F, Elidan J, Valarezo A: Dorello’s canal: A microanatomical study. J Neurosurg 75:294-298, 1991
- Yasargil MG, Mortara RW, Curcie M: Meningiomas of the basal posterior cranial fossa, in Krayenbühl H (ed): Advances and Technical Standards in Neurosurgery . Vienna, Austria: Springer-Verlag, 1980, pp 3-11
