
Disorders of Behavior and Self-Awareness
George P. Prigatano, PhD
Division of Neurology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
Disorders of self-awareness frequently follow frontal lobe damage. Patients with bilateral lesions of the premotor cortex often have poor self-awareness and finger tap slowly. Patients with orbitofrontal lesions also may have impaired self-awareness, but their speed of finger tapping is normal. Two such patients are described, and the implications for a theory of disordered self-awareness after brain injury are considered.
Key Words: anosognosia, denial, frontal lobes, impaired awareness, neuropsychology, traumatic brain injury
Numerous behavioral disorders have been associated with injuries to the anterior regions of the brain, which are referred to as the frontal lobes.17 The structures anterior to the central sulcus occupy as much as a third of the brain’s mass in adult humans and is anything but homogeneous in terms of its cytoarchitectonics and patterns of neural connections.1 Although large regions of the brain appear to contribute to the phenomenon of humanconsciousness, the frontal region may play a special role. It is related to our capacity for intention and attention and important for the perception of events in time and verbal control of motor functions. It may be especially important for our sense of what is familiar and therefore real.
This article offers a broad definition of human consciousness and describes two indirect methods of measuring impairments in self-awareness associated with brain damage. It discusses how speed of finger tapping may serve as a behavioral marker for impaired self-awareness in some patients. Examples of how damage to different regions of the frontal lobe can produce different behavioral patterns associated with impaired self-awareness are provided. Finally, the model of human self-awareness presented argues that this emergent brain function integrates thinking with feeling and therefore is not a pure cognitive function.
Human Consciousness: Broad Definition and Indirect Measurements
Human consciousness appears to be an emergent brain function that permits an individual to experience a subjective sense of reality. By its nature, consciousness conveys a sense of what is real now as well as what was real in the past. It provides a sense of contiguity for the self and thereby mediates how individuals perceive and deal with the external world.
Because human consciousness and associated disorders of self-awareness represent a phenomenological state in which individuals experience themselves in the here and now, it can only be measured indirectly. Clinically, we have used two methods to study self-awareness. Both methods have limitations, but they provide useful information.
In the course of neuropsychological rehabilitation, many postacute traumatic brain injury (TBI) patients appear to have limited insight into their behavioral disorders.12 This phenomenon is not a direct function of their degree of dementia or their impairment in abstract reasoning skills. In fact, some patients with significant frontal lobe pathology perform well on standardized neuropsychological measures but do not recognize their inappropriate social behavior.6
We constructed a simple scale of 30 behavioral items that asks patients to rate how easy or difficult it is for them to perform a series of behavioral tasks. Independently, relatives or significant others who know the patient well are also asked to rate the patient on each of the same behavioral items. The difference between the relatives’ and patient’s perceptions is considered an indirect measure of impaired self-awareness. The scale, called the Patient Competency Rating Scale (PCRS), was developed primarily to supplement information obtained during neuropsychological interviews.12
Numerous studies have been conducted using the PCRS, some across cultures. Overall, the findings have been consistent. Patients with moderate to severe TBI often overestimate certain behavioral competencies compared to their relatives’ reports of how well the patient can perform the activity. The degree of overestimation relates to the severity of brain injury and to the severity of disturbed consciousness at the time of injury.9-11,14,15 This method, however, has a serious limitation. Depending on the relative who rates a given patient’s behavioral competencies, the resulting evaluation can be fairly objective or quite biased. That some family members make objective reports while others do not prompted the development of a second method for assessing impaired awareness.
The traditional neurological approach is to observe patients and to ask them direct but simple questions about their ability to function. For example, Starkstein and coworkers16 developed an anosognosia questionnaire that included questions such as, Why are you here? Is anything wrong with your arm or leg? Is your limb weak, paralyzed, or numb? Depending on the patient’s answers to these and other questions, clinicians rated whether anosognosia was present.
Frequently, disturbances of self-awareness can exist in the absence of a frank neurological condition (e.g., the absence of aphasia, hemiplegia). Nevertheless, by carefully observing a patient’s behavior, clinicians can make a clinical judgment about whether the patient shows evidence of impaired awareness. Recently, a Clinician’s Rating Scale (CRS) has been developed to evaluate impaired self-awareness and denial of disability after brain injury.13 In Jacksonian terminology, anosognosia or a lack of awareness after brain injury is a direct effect of brain dysfunction and therefore should be viewed as a negative symptom. In contrast, denial phenomena after brain injury are an indirect effect of disturbed functioning and therefore a positive symptom. In this situation, individuals have partial knowledge of their disability or impairments but “deny” their disability, apparently for psychological reasons.
Although the formulation is incomplete,8 this dichotomy suggests that several characteristics are associated with impaired self-awareness. First, patients with impaired self-awareness lack information about themselves. Second, they experience cognitive “perplexity” when they receive feedback about their behavioral limitations. Finally, they exhibit a cautious willingness or indifference when asked to consider new information about their level of functioning. In contrast, patients who deny their disability exhibit an implicit knowledge of impaired functioning. They actively resist information about their impairments and disabilities and exhibit some form of active (versus passive) response to the “new information” about themselves. Based on these broad criteria, an impaired self-awareness scale and a denial of disability scale were developed.
Speed of Finger Tapping, Impaired Self-Awareness, and Denial of Disability
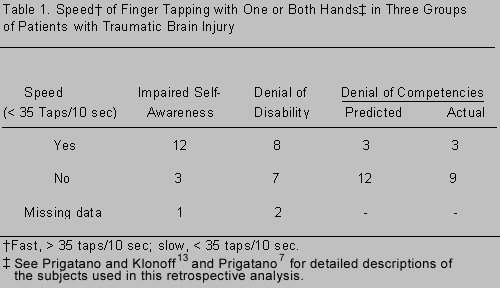 Speed of finger tapping is slower in TBI patients who show limited self-awareness.9,15 The speed of finger tapping in patients later classified as showing primarily impaired self-awareness rather than denial of disability13 was evaluated when they first entered a neuropsychological rehabilitation program. A mean speed of finger tapping of 35 taps/10 seconds or less in one or both hands on the Halstead Finger Oscillation Test was classified as slow. Retrospective analysis revealed that 12 of 15 (80%) subjects (one subject’s data were missing) with impaired self-awareness showed this pattern in contrast to 8 of 15 subjects (53.3%; two subjects’ data were missing) in the denial of disability group. These findings suggested that brain dysfunctional patients who showed behavioral indications of impaired self-awareness tended to tap their fingers slowly.
Speed of finger tapping is slower in TBI patients who show limited self-awareness.9,15 The speed of finger tapping in patients later classified as showing primarily impaired self-awareness rather than denial of disability13 was evaluated when they first entered a neuropsychological rehabilitation program. A mean speed of finger tapping of 35 taps/10 seconds or less in one or both hands on the Halstead Finger Oscillation Test was classified as slow. Retrospective analysis revealed that 12 of 15 (80%) subjects (one subject’s data were missing) with impaired self-awareness showed this pattern in contrast to 8 of 15 subjects (53.3%; two subjects’ data were missing) in the denial of disability group. These findings suggested that brain dysfunctional patients who showed behavioral indications of impaired self-awareness tended to tap their fingers slowly.
Another analysis was conducted to explore this relationship further. Finger-tapping scores from a group of patients referred for a clinical neuropsychological evaluation but later considered to be free of a primary brain disorder were analyzed retrospectively (these patients were control subjects in another study).7 The distribution of the speed of finger tapping scores was predicted to be opposite that of the group with impaired self-awareness. These patients, who believed that they had significant brain damage but who were considered not to, were referred to as a denial of competency group. The differences among the three groups were highly significant (Table 1). Thus, speed of finger tapping may be a neurobehavioral marker associated with impaired self-awareness after brain injury.
Disorders of Impaired Awareness After Frontal Lobe Injury
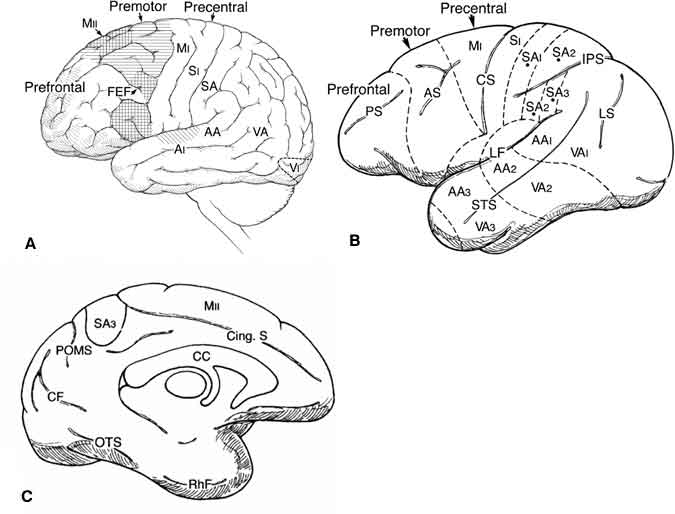
The anatomy of the brain can be studied from different perspectives, and the perspective influences how the relationship between certain regional neuropathological disturbances and disturbances in human consciousness is regarded. The frontal lobes have been studied from the perspective of evolution, their morphological characteristics (i.e., cytoarchitectonics), and their complex matrix of neural connections with other regions of the brain.3
Traditionally, neuroscientists have identified at least three and possibly four distinct regions of the frontal lobes. The three major subdivisions are the precentral, premotor, and prefrontal regions (Fig. 1). The cingulate sulcus on the mesial undersurface of the brain includes large areas of the prefrontal and promotor regions. A fourth division, the orbitofrontal region, has inputs to and from the limbic system. The orbitofrontal region involves the ventral portion of the prefrontal region and the anterior cingulate gyrus and may play a unique role in psychological processing. Pribram5 has suggested that this area is crucial to our ability to experience a “feeling of familiarity” when processing information (p. 19). Moreover, the prefrontal, orbitofrontal, and what Pribram refers to as the “far frontal” regions of the human frontal lobe may be involved in both attention and intention. Depending on which region is injured, individuals might exhibit not only different changes in behavior but subtle differences in disturbances in consciousness. Two patients with frontal lobe damage offer clinical insights into patterns of response after TBI.
Clinical Manifestations of Impaired Self-Awareness
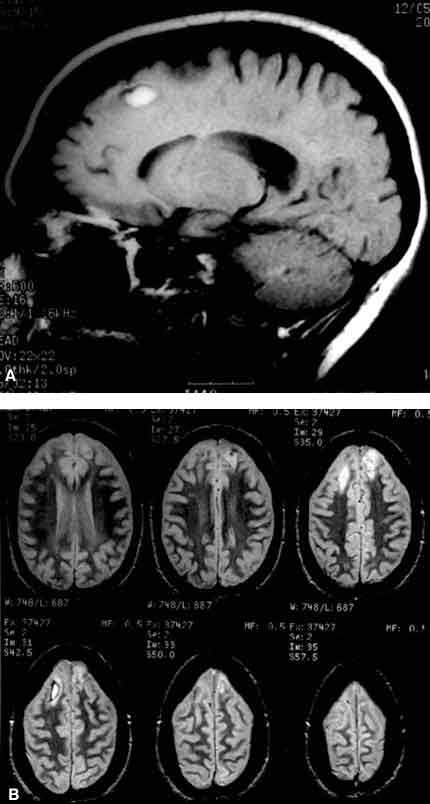
A 31-year-old, right-handed female suffered a severe TBI (admitting score on the Glasgow Coma Scale = 7) in November 1997. A magnetic resonance imaging study revealed bilateral injuries of the premotor (including supplementary motor area) and prefrontal regions (Fig. 2), which project directly to the basal ganglia. She was evaluated 7 days after brain injury and followed serially for the next 4 months.
Initially, she had notable difficulty performing the Halstead Finger Tapping Test. When given the typical instruction: “ready, set, go,” several seconds would elapse before she could initiate the first tapping motion. Her index finger would start to show a slight downward movement, but full extension of the finger took 2 to 3 seconds. A few weeks after injury, her rate of finger tapping was markedly slow compared to normal (Table 2). Within 4 months of injury, however, her rate of tapping approached normal.
The patient also demonstrated behavioral characteristics often associated with bilateral injuries to the premotor and prefrontal cortex. She was highly perseverative and exhibited hemi-inattention for the right side of space. She never verbalized a want or desire. Her husband noted that she was sexually disinhibited but could be easily distracted. Her face revealed no evidence of emotion, and she behaved in a “robot-like” manner. When asked if she could remember the accident, she stated that she could not but showed no distress that she was in the hospital or had significant neurobehavioral deficits. She expressed no desire to stay in or leave the hospital. She was aspontaneous and apathetic. When asked whether she had any significant impairments, she said no.
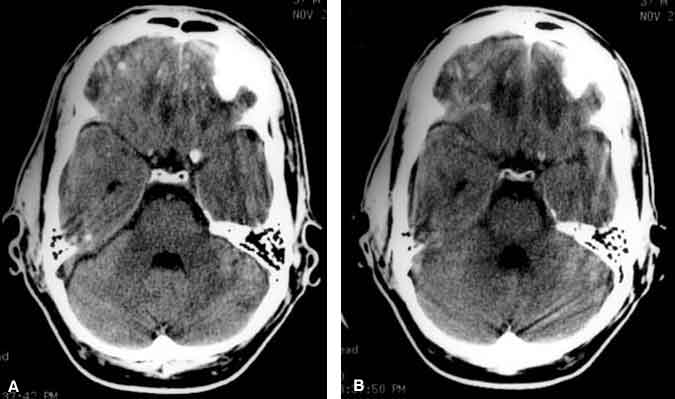
The second patient, a 34-year-old male who fell while working, exhibited a strikingly different pattern of behavior. The length of time that he was unconsciousness was undetermined. When he was discovered, he was combative, argumentative, and disoriented but insisted that he was perfectly “fine.” He begrudgingly consented to go to the hospital. Upon arrival, he told the doctors that he thought he was there because he had a “flu.” He soon became belligerent and insisted on leaving the neurosurgical ward against medical advice. He felt that nothing was being done for him and that he had absolutely no disturbance that would require hospitalization. In contrast with the first patient, he had definite wants and desires. He underwent computed tomography (CT), which revealed an orbitofrontal injury (Fig. 3).
This patient, who was followed for more than a year, repeatedly insisted that he could not believe that he had sustained a TBI. Nothing in his experience led him to believe that he had a brain injury. When his medical records and diagnostic films were reviewed with him about a year after his injury (and in the context of psychotherapy), he reacted intensely to his CT scan. He broke into tears, stating that his injury, for the first time, seemed real to him. Overall, he seemed to lack any sense of familiarity with his brain injury (as suggested by Pribram5). Perhaps individuals with a significant orbitofrontal injury lose the sense of recognizing or feeling familiar with their injury. If so, they might become belligerent or use other premorbid behavioral strategies to cope with an environment that they do not fully understand.
Within 1 month of injury, this patient’s bilateral speed of finger tapping was normal (56.4 taps/10 seconds with the right hand and 50.8 taps/second with the left hand) and has remained so. Some examiners might have described this patient as denying his disability, but his case cautions therapists to judge patients slowly before invoking the term denial. This patient’s impaired self-awareness and associated behavioral abnormalities suggest that different patterns of impaired self-awareness may be associated with damage to different areas of the frontal lobes. Furthermore, speed of finger tapping parallels impaired self-awareness in some patients but not in others.
Disorders of Impaired Awareness over Time
One of the more perplexing issues is how to study impaired self-awareness, particularly given that the disorder can change rapidly over time. Anosognosia for hemiplegia is a striking example. Within the first few days of a cerebrovascular accident, about a third of hemiplegic patients may deny their hemiplegic side.16 Within the first couple of weeks, however, they often begin to recognize that the side has been affected. These patients may appear apathetic or euphoric. What mechanisms underlie the recognition of their hemiparetic limb and concomitant indifference to an event that most people would perceive as a major loss?
Months or years can elapse without some patients (such as those with severe TBI) fully recognizing the neuropsychological consequences of their injury. Many of these individuals appear surprised when their cognitive deficits are demonstrated to them. They do not seem to experience the deficits or the implications of the deficits as do people around them. A comprehensive model of impaired self-awareness is needed to improve understanding of this class of disorders.
Model for Conceptualizing Disorders of Self-Awareness After Brain Injury
Human consciousness is considered an emergent brain function that depends heavily on the functional integrity of the area of the brain that Mesulam2 has referred to as heteromodal cortex. Heteromodal cortex integrates information from sensorimotor and unimodal regions with the limbic system. In other words, these regions integrate thinking and feeling. Thus, damage to these regions produces both cognitive and affective disturbances. Therefore, disorders of self-awareness are associated with specific cognitive and affective “markers.”
After an acute unilateral lesion, cerebral dysfunction is often bilateral.4 The degree to which the heteromodal cortex is disrupted bilaterally may determine whether frank anosognosia is present. If dysfunction of heteromodal cortical regions is widespread and bilateral, individuals may exhibit a severe impairment of self-awareness months or years after injury. As regions of heteromodal cortex begin to regain their functional integrity with time, rehabilitation, or both, the clinical picture changes. This model implies that permanent anosognosia would exist only in individuals with extensive bilateral damage to the heteromodal cortex.
Theoretically, the return of normal physiological activity to one side of the brain would be associated with a partial impairment of self-awareness. Such individuals would likely attempt to use some method to cope with the changes in their ability to function. Clinicians often observe two methods of coping in brain-injured patients. The first is a defensive strategy for which the defense mechanism of denial may be an apt description. In the second instance, the method of coping is not denial. Rather, patients may use a nondefensive strategy that they used to cope with problems before their brain injury. In other words, individuals may simply rely on previous methods of functioning to make sense of their environment and to get their needs met. This behavior does not reflect denial as the term is typically used in psychiatry.8
The model also suggests that four general syndromes may be associated with disorders of self-awareness. Each syndrome would be associated with heteromodal damage to frontal, parietal, temporal, and occipital areas, respectively. The behaviors associated with these proposed heteromodal syndromes of impaired awareness are described elsewhere.8
Conclusions
Damage to the frontal lobe is associated with disturbances in both behavior and consciousness. Within the context of the behavior observed in patients after TBI, two manifestations are possible. The first is associated with damage to premotor and prefrontal regions that involve the supplementary motor area. Individuals with such injuries often lack initiation and planning, are aspontaneous, and exhibit a flat affect. Bilaterally, their speed of finger tapping is often slow. The other manifestation may be associated with an orbitofrontal injury. Such individuals often finger tap at normal speeds but have substantial difficulties in sustaining their attention and in controlling their emotional reactions. Unlike patients with injuries of the dorsolateral premotor and prefrontal regions, patients with orbitofrontal injuries exhibit clear intentions but may have a difficult time controlling those intentions. Some of these individuals report a loss of their sense of smell, providing further indirect evidence of damage to the orbital surface of the frontal lobe. Such patients often complain that they “cannot believe” that they have suffered a brain injury because nothing in their experience allows them to believe that a brain injury has actually occurred. Using Pribram’s terminology, these patients have lost the sense of familiarity. Consequently, their experience of reality is altered.
Adapted from Prigatano GP: Disorders of behavior and self-awareness. In Azouzi P, Bussel B (eds): Prefrontal Dysfunctions: Assessment and Rehabilitation, Paris: Arnette Publishers, 1998, pp. 77-84. With permission from Arnette Publishers.
References
- Fuster JM: The Prefrontal Cortex, Anatomy, Physiology, and Neuropsychology of the Frontal Lobe. Philadelphia: Lippincott-Raven, 1997
- Mesulam MM: Principles of Behavioral Neurology. Philadelphia: F.A. Davis, 1985
- Pandya DN, Barnes CL: Architecture and connections of the frontal lobe, in Perecman E (ed): The Frontal Lobes Revisited. New York: IRBN, 1987, pp 41-72
- Perani D, Vallar G, Paulesu E, et al: Left and right hemisphere contribution to recovery from neglect after right hemisphere damage—an [18F] FDG PET study of two cases. Neuropsychologia 31:115-125, 1993
- Pribram KH: The subdivision of the frontal cortex revisited, in Perecman E (ed): The Frontal Lobes Revisited. New York: IRBN, 1987, pp 11-39
- Prigatano GP: Disturbances of self-awareness of deficit after traumatic brain injury, in Prigatano GP, Schacter DL (eds): Awareness of Deficit After Brain Injury. Clinical and Theoretical Issues. New York: Oxford University, 1991, pp 111-126
- Prigatano GP: Behavioral limitations TBI patients tend to underestimate: A replication and extension to patients with lateralized cerebral dysfunction. The Clinical Neuropsychologist 10:191-201, 1996
- Prigatano GP: Principles of Neuropsychological Rehabilitation. New York: Oxford University, 1999
- Prigatano GP, Altman IM: Impaired awareness of behavioral limitations after traumatic brain injury. Arch Phys Med Rehabil 71:1058-1064, 1990
- Prigatano GP, Altman IM, O’Brien KP: Behavioral limitations that traumatic-brain-injured patients tend to underestimate. The Clinical Neuropsychologist 4:163-176, 1990
- Prigatano GP, Bruna O, Mataro M, et al: Initial disturbances of consciousness and resultant impaired awareness in Spanish patients with traumatic brain injury. J Head Trauma Rehabil 13:29-38, 1998
- Prigatano GP, Fordyce DJ, Zeiner HK, et al: Neuropsychological Rehabilitation After Brain Injury. Baltimore: Johns Hopkins University, 1986
- Prigatano GP, Klonoff PS: A clinician’s rating scale for evaluating impaired self-awareness and denial of disability after brain injury. The Clinical Neuropsychologist 12:56-57, 1998
- Prigatano GP, Leathem JM: Awareness of behavioral limitations after traumatic brain injury: A cross-cultural study of New Zealand Maoris and non-Maoris. The Clinical Neuropsychologist 7:123-135, 1993
- Prigatano GP, Ogano M, Amakusa B: A cross-cultural study on impaired self-awareness in Japanese patients with brain dysfunction. Neuropsychiatry Neuropsychol Behav Neurol 10:135-143, 1997
- Starkstein SE, Fedoroff JP, Price TR, et al: Anosognosia in patients with cerebrovascular lesions. A study of causative factors. Stroke 23:1446-1453, 1992
- Stuss DT, Benson DF: The Frontal Lobes. New York: Raven, 1986

