
Grading Scale for Cerebral Concussions
Roberto Masferrer, MD†
Mauricio Masferrer, MD‡
Virginia Prendergast, NP†
Timothy R. Harrington, MD
†Masferrer Neurosurgical, Colorado Springs, Colorado
‡Mental Health Center of East Central Kansas, Emporia, Kansas
Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
Traditionally, cerebral concussions have been defined as a transient loss of consciousness without neuroanatomical evidence of parenchymal injury. The concept, however, is no longer valid. When sustained in industrial accidents, motor vehicle collisions, and assaults, cerebral concussions can have forensic and medicolegal implications, and treating physicians must decide when is it safe for the injured patients to return to work or resume driving. Cerebral concussions also assume a significant role in sports, especially in high school and college. Medical personnel assigned to the sports arena must determine if athletes have been injured severely enough to preclude their continued participation in the event. Team doctors or coaches also must determine how soon injured athletes can participate in upcoming events. After a cerebral concussion, an athlete’s risk for a second injury increases, and second-impact syndromes can have devastating outcomes. A scientific consensus onhow to assess the severity of concussions has been lacking. In 1997 the American Academy of Neurology issued a practice parameter offering a grading scale to assess the severity of cerebral concussions sustained during athletic competitions. We concur with the need to grade these injuries, primarily to prevent the devastating sequelae associated with a second or repetitive injuries in sports. A grading scale could also be applied to concussions sustained in other circumstances and could be used for prognostic, medicolegal, and forensic determinations.
Key Words: cerebral concussion, closed head injury, head trauma, sport injuries, traumatic brain injury
In contemporary society cerebral concussions occur as a result of head trauma sustained in motor vehicle accidents, acts of violence, industrial accidents, falls, and sports. It is difficult to assess the incidence of cerebral concussions in the general population, but they are likely underreported. Each year 100,000 to 250,000 concussions may occur in sports alone.[4,16]
The classic definition of a cerebral concussion (commotio cerebri) implied a transient loss of consciousness but no underlying injury to the brain parenchyma. In contrast, the classic definition of a cerebral contusion (contusio cerebri) involved loss of consciousness with damage to the cerebral parenchyma. The hallmark of both injuries was loss of consciousness.[10] Based on these definitions, it was sometimes difficult to differentiate contusions from concussions or to determine the severity of injuries. Subjective and arbitrary values for the length of unconsciousness were used to determine whether a patient had suffered a concussion or a contusion. If consciousness was lost less than 10 minutes, it was considered a concussion; if the loss was longer, it was considered a contusion or a more serious intracranial injury.[1]
Computerized tomography (CT) and magnetic resonance (MR) imaging permit the degree of parenchymal damage to be quantified and graded, thereby improving the diagnosis of cerebral contusions. The degree of severity of a concussion, however, remains hard to assess based on imaging studies that are considered normal by definition. While a normal CT scan confirms the diagnosis, it provides no information about the severity of injury. The increased sensitivity of MR imaging, however, has revealed abnormal signal intensities in the brain parenchyma of some patients with cerebral concussions.[1,12,19,21] That the MR imaging studies of some patients with concussions are normal while others show abnormal signal intensities corroborates the concept that cerebral concussions have different levels of severity and supports the need for a universally accepted grading scale.
Historical Review
Loss of consciousness after head trauma has been known since Biblical times. In the classic story of David and Goliath, the Philistine giant lost consciousness after being struck in the head by a slingshot from the young shepherd. Whether Goliath suffered a concussion, a contusion, or a more serious brain injury is unclear as the young David did not allow the giant to recover from his posttraumatic slumber.
Millennia later in another epic saga (Historia Verdadera de la Conquista de la Nueva España), Bernal Diaz del Castillo described how Montezuma was struck in the temple by a rock. The Aztec leader immediately lost consciousness. He recovered transiently but became progressively ill and died within two days. This description probably represents the first report of an epidural hematoma in the New World. An Inca skull from the same period (housed at the Smithsonian Institute) has seven trephinations associated with skull fractures. Six of the trephinations show different stages of healing, indicating that preColumbian brain surgeons were successful and that repetitive head injuries were common among indigenous warring societies.
The first medical writings on head injuries date to the possible teachings of Imhotep, some 2,000 years before Christ. The Edwin Smith papyrus described 10 cases of head injury, one of which was a closed head injury with posttraumatic neurologic sequelae.[10,20]
The corpus Hippocraticum contains several references to both open and closed head injuries, including cerebral concussions, which were translated to Latin and then English as commotio cerebri. The following translation is from the book of Hippocratic aphorisms: “when commotion of the brain is caused by a blow, the victim loses his speech and cannot see or hear.”[28]
In his work De Arte Medica, Cornelius Celsus noticed that a “cerebral hemorrhage” could occur without a skull fracture. Some 700 years later in Persia, Rhazes recognized that concussive states could occur without skull injury.[10] Nothing new was added to these concepts until the 16th century. When Bernal Diaz del Castillo was recording the death of Montezuma in Tenochtitlán, Ambroise Paré in France re-embraced the concept of commotio cerebri, which he referred to as “concussion,” “commotion,” or “shaking of the brain.” In the 17th century, Jean Louis Petit’s surgical teachings included a classification of skull fractures and intracranial hematomas. He described contre-coup injuries and gave two reasons for loss of consciousness: “concussions” or “the accumulation of blood inside the skull.”[8]
In 1705 Littre re-affirmed the lack of an anatomical basis for concussion based on the autopsy findings of a patient with a head injury who died soon after ramming his head against a wall. He proposed the adjectives light, severe, and fatal to describe the severity of these injuries.[6] In 1839 Dupuytren described small cerebral hemorrhages as contusio cerebri. For the first time, he established the difference between loss of consciousness with (contusion) and without (concussion) parenchymal injury.[6]
Based on the teachings of John Hunter and John Hughlings Jackson, English authors emphasized that patients with concussions not only lost consciousness but developed posttraumatic amnesia. Gradually over the 19th and early 20th century, posttraumatic amnesia became a parameter for assessing the severity of a head injury.[6]
In 1941 Denny-Brown and Russell[7] showed experimentally that cerebral concussion is associated with a sudden, transient, traumatic disturbance of brain function but neither macroscopic nor microscopic lesions of the brain parenchyma. This experimental model established the scientific basis for the clinical descriptions used for years thereafter.
In 1970 Yarnell and Lynch[33] reported four football players who exhibited posttraumatic confusion after concussion but who were aware of immediate pretraumatic events. Only one of these patients lost consciousness transiently. Within 3 to 20 minutes of the impact, however, all four patients developed amnesia for the pretraumatic events. This report pointed out that recent memory may appear normal immediately after a traumatic event but that the information is not transferred to permanent memory.
Without data from CT or MR imaging, the terms concussion and contusion overlap. The adjectives mild, moderate, and severe have been added to establish a gradient for severity of injury. Despite these modifiers, the term concussion remains ambiguous and difficult to quantify for individuals outside the fields of neurology or neurosurgery. Before the Glasgow Coma Scale (GCS) was developed, the same ambiguity surrounded the terms lethargy, stupor, and coma. The ambiguity, however, disappeared after Teasdale and Jennett30 developed their system for rating the severity of lost consciousness.
The American Academy of Neurology (AAN) and the American Association of Neurological Surgeons (AANS) concur that a grading scale is essential for assessing the degree of injury in patients who sustain cerebral concussions as a result of sports injuries. This consensus was reached by reviewing the literature on the subject and creating panels of experts who presented their findings for review and feedback at two meetings. The first meeting, entitled “Sports-Related Concussion: Neurological and Neuropsychological Evaluation and Management,” was held in Pittsburgh on March 23, 1996. The second was convened at the annual meeting of the AANS in Minneapolis on April 28, 1996. As a result of these meetings, a grading scale was developed and the AAN published a practice parameter in March 1997.[2]
Pathophysiology
Before the 19th century, the concept of cerebral concussion was simplistic: The brain was “shaken” beyond its level of tolerance, transiently shutting down cerebral functions. In 1941 Denny-Brown and Russell7 defined cerebral concussion as an “essentially reversible syndrome without detectable pathologic changes in the cerebral parenchyma.” The underlying cause of the loss of consciousness was considered a physiologic disturbance of the brain stem.
In 1966, a neurosurgical ad hoc committee studying the nomenclature of head injury concurred that the definition of cerebral concussion was “a clinical syndrome characterized by immediate and transient impairment of neural function (e.g., alteration of consciousness, disturbance of vision, equilibrium) due to mechanical forces.”[11] The focus of the definition was the transient impairment of neural function caused by mechanical forces and based on the experimental work of Denny-Brown and Russell.[7] Earlier, Ward.[31] defined concussion as “the loss of consciousness and associated traumatic amnesia which occurs as the consequence of head trauma in the absence of physical damage to the brain.” By 1974 Ommaya and Gennarelli.[22] defined cerebral concussion as “a graded set of clinical syndromes following head injury wherein increasing severity of disturbance in level and content of consciousness is caused by mechanically induced strains affecting the brain in a centripetal sequence of disruptive effect on function and structure.”
These definitions are based on the basic pathophysiology of cerebral concussion. They all focus on the transient impairment of neural function or the disturbance in the level and content of consciousness. These two manifestations of cerebral concussion are the result of an underlying injury to the neural structures. The causes for these alterations have been attributed to both static and dynamic forces acting on the brain parenchyma.[22] Static forces are rare in trauma, occurring only in slow crush injuries. In contrast, the dynamic forces caused by acceleration or deceleration, in combination with rotation, are thought to be the primary cause of concussive injuries.[7,22]
In experimental models, nonimpact inertial loading has produced traumatic unconsciousness. Linear acceleration is not as important as rotation of the head. In deceleration injuries the inertia of the brain is the primary determinant of the severity of the injury, but a rotational component also tends to worsen the injury.[1,22] In clinical practice deceleration injuries are the most common and are associated with diffuse axonal injury. The inertia of the head, transmitted by sudden deceleration to the brain parenchyma, causes the injury.[1]
Cerebral concussions can be focal or diffuse. Focal concussions are restricted to an isolated, asymmetrical area of the cortex. Diffuse injuries, however, are thought to be the most common, and axonal disruption has been demonstrated on high-resolution light microscopy and electron microscopy.[5,22,23,25] As a result of these new findings, the concept of diffuse axonal injury was proposed as the pathophysiologic substrate of the clinical syndromes associated with cerebral concussion.[23]
From the surface of the brain to the brain stem, the distribution of the magnitude of the shear strains produced by an acceleration-deceleration-rotation injury decreases. At low levels of energy, the strains do not extend deeper than the cortex or its junction with the white matter. High-energy strain, however, injures deeper structures.[1,14,22,24] Unconsciousness may develop when the level of energy associated with the shear strain is large enough to reach the brain stem.[1,22] The level and distribution of these shear strains may explain the clinical differences manifested by patients after injury.[1,22]
Neuroimaging
The original definitions of commotio cerebri, contusio cerebri, and compresio cerebri reflected clinical observations at a time when radiology was unavailable. Even in the late 1970s when first-generation CT scanners became available, this new imaging modality was not used to evaluate cerebral concussions. By the mid- and late 1980s, however, CT was widely available and the sophistication of the software of the “newer” and “faster” fourth-generation units had increased. These improvements insured the widespread use of CT to evaluate head trauma, especially if associated with loss of consciousness. The consensus has been that CT images of cerebral concussions are normal.
In contrast, cerebral contusions are associated with abnormal CT images. Low-attenuation coefficients are thought to represent cerebral edema associated with an area of contusion. High-attenuation coefficients represent the hemorrhagic form of cerebral contusions. On serial studies some contusions can progress to frank parenchymal hemorrhages, which are referred to as delayed hematomas.
Cerebral hyperemia also has been postulated to underlie “diffuse cerebral swelling” on CT scanning, especially in children with head trauma. In such cases, the brain appears “swollen,” but the CT coefficients are higher than normal, suggesting hyperemia rather than true edema.[3] This appearance on CT is similar to that associated with normal perfusion pressure breakthrough (i.e., diffuse swelling of a lobe or hemisphere that can follow the resection of an arteriovenous malformation).[29] Reports of abnormal CT scans in patients with clinical syndromes compatible with cerebral concussion have challenged the traditional concept that imaging studies are always negative in mild cerebral injuries.[14,18,24,32]
The advent of MR imaging further challenged the concept of the brain being “normal” in cerebral concussion. MR imaging has demonstrated abnormalities that CT is unable to detect.[13,18,32] The superiority of MR imaging over CT is the result of the high level of contrast between the brain signal and that of cerebrospinal fluid (CSF), the lack of a computer-averaging artifact from adjacent bone, and the capacity for multiplanar imaging.[13] Compared to CT, MR imaging offers better resolution of the normal brain parenchyma and injured areas compared to normal cerebral tissue. Cerebral edema, shear injuries, and encephalomalacia are exquisitely demonstrated because they increase the water content of cerebral tissue, which, in turn, is easily detected on T2-weighted images.[12,13,19,21] As many as 25% of patients with normal CT scans and mild head injuries show abnormalities on MR imaging.[1,19] Newer “fast-generation” MR imagers will make this modality more accessible for the evaluation of trauma victims. Additional information derived from the MR imaging study of patients with cerebral concussions should improve our understanding of brain injuries.
Neuropsychology
With the acceptance of the GCS as the gold standard for quantifying loss of consciousness, the concept of traumatic brain injury (TBI) evolved during the late 1980s and early 1990s.[1] By necessity TBIs were divided into mild, moderate, and severe according to the patient’s GCS score. The mild forms of TBI encompass the classic concussive syndromes and also mild cerebral contusions. By definition a mild TBI is characterized by five features: a GCS score between 13 and 15, a brief period of unconsciousness or dazed consciousness, posttraumatic amnesia, negative neuroimaging studies, and recovery within weeks to months without sequelae or the need for specific medical intervention.[1] Based on the preceding discussion, the category of mild TBI should probably include only patients with a GCS score of 15 and normal MR images.
In 1968 Oppenheimer reported diffuse axonal injury in patients with mild forms of head injuries who died from unrelated causes.[23] Based on neuropathological material and experimental models, the duration of coma and the duration of posttraumatic amnesia are probably the best indicators of the severity of diffuse axonal injury.[1,22,23] In turn, the severity of axonal injury determines a patient’s final functional outcome.
Although mild TBI is one of the most common neurologic disorders,[1,9] its evaluation and management are not formally taught in medical school or residency programs. Postgraduate teaching in neurology fails to reflect the high prevalence of TBI.[1] Unfortunately, the disorder is considered an unpleasant clinical exercise that is neither intellectually compelling nor associated with academic rewards while too often associated with litigation.[1] Thus, TBI patients are cared for by neurologists, internists, or family physicians who received no instruction in their care during residency programs or by neurosurgeons who are “too busy” to care for them in their private practices. Consequently, old myths persist and patients return to work, school, or competition earlier than advisable. The neuroforensic implications of this situation can be devastating for patients.
The clinical picture associated with mild TBI is not only the result of the head injury but a complex syndrome in which signs and symptoms of vestibular, cerebral, and psychological dysfunction coexist with soft tissue injuries, primarily of the head, face, and cervical region.[1] Inexperienced physicians can sometimes misinterpret this complex picture as malingering or “craziness.” Early deficits in arousal mechanisms clear quickly, but impairments in attention last longer. Awareness always recovers first, followed by the gradual recovery of other more sophisticated cognitive functions. Amnesia of variable duration is always present. After patients have regained a normal level of consciousness, they complain of many disabilities, most of which are related to attention, concentration, and memory. Yet they perform normally on simple clinical tests such as counting backward from 100, serial sevens, and three-word recollection. Such “normal results” on cursory testing of cognitive functions usually foster the suspicion of exaggeration for secondary gain, or malingering.[1]
Highly motivated professionals with the mildest forms of concussion or young athletes with the vernacular “dings” recover in a few days. If consciousness is lost briefly (GCS = 15 in the emergency room and posttraumatic amnesia less than 1 hour), complete recovery can require 6 to 12 weeks. When unconsciousness persists more than 10 minutes and posttraumatic amnesia persists 4 to 6 hours, recovery can require more than 3 months. Ten to 15% of patients with cerebral concussions sustain a permanent disability. Patients older than 50 years may require much longer to recover and some may never recover completely.[1,17,26,33]
Grading Scales
 All sports are associated with a risk of injury. In contact sports and sports associated with high-speed and high-body inertia, the potential to injure the central nervous system and spine is even higher. Repeated injuries can be cumulative and produce delayed neurophysiological and neuropsychological deficits. In the heat of competition, athletes and coaches alike can lose their objectivity. Fans, teammates, peers, and the news media can pressure injured athletes to return to competition under potentially harmful or detrimental conditions. The deaths or devastating injuries suffered by 19 football players in 1904 outraged President Theodore Roosevelt and prompted the creation of the National Collegiate Athletic Association to establish rules and regulations for safer athletic competition.
All sports are associated with a risk of injury. In contact sports and sports associated with high-speed and high-body inertia, the potential to injure the central nervous system and spine is even higher. Repeated injuries can be cumulative and produce delayed neurophysiological and neuropsychological deficits. In the heat of competition, athletes and coaches alike can lose their objectivity. Fans, teammates, peers, and the news media can pressure injured athletes to return to competition under potentially harmful or detrimental conditions. The deaths or devastating injuries suffered by 19 football players in 1904 outraged President Theodore Roosevelt and prompted the creation of the National Collegiate Athletic Association to establish rules and regulations for safer athletic competition.
Nonetheless, an average of 8 to 10 people die from sports each year. Occasionally as many as 30 athletes have died in a year as a result of head and cervical spine injuries.[27] Most fatalities occur in football or hockey and are caused by direct blows to the head. Fatalities also occur in other sports and gymnastics as a result of collisions or falls.[16] The overall incidence of cerebral concussions in all sports is unknown. The incidence of concussion among high school football players is estimated at 20% and at 10% among college football players. The severity of sports injuries ranges from the minor dings with only transient impairment to sudden deaths in the field.[15,16]
 This broad range prompted national organizations interested in the well being of athletes to develop a scale to assess the severity of cerebral concussions for both therapeutic and prognostic purposes. The earliest scales for grading the severity of both cerebral concussion and TBI relied on vague descriptive terms (mild, moderate, and severe) that could not be quantified. Combining the resources of the Colorado Medical Society (CMS), the AAN, the AANS, high school and college coaches, and sports medicine organizations, a grading scale for sports-induced cerebral concussions was developed in 1997. The intent of establishing the severity of an injury was to implement guidelines for the management of concussions in athletes in the field, to prevent catastrophic outcomes, and to avoid cumulative brain injuries.[16]
This broad range prompted national organizations interested in the well being of athletes to develop a scale to assess the severity of cerebral concussions for both therapeutic and prognostic purposes. The earliest scales for grading the severity of both cerebral concussion and TBI relied on vague descriptive terms (mild, moderate, and severe) that could not be quantified. Combining the resources of the Colorado Medical Society (CMS), the AAN, the AANS, high school and college coaches, and sports medicine organizations, a grading scale for sports-induced cerebral concussions was developed in 1997. The intent of establishing the severity of an injury was to implement guidelines for the management of concussions in athletes in the field, to prevent catastrophic outcomes, and to avoid cumulative brain injuries.[16]
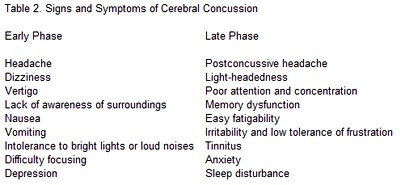
Three grades of cerebral concussion were accepted for the evaluation and treatment of injured athletes (Table 1). Patients with a Grade 1 concussion experience transient confusion but do not lose consciousness. All symptoms (Table 2) resolve in less than 15 minutes. Patients with Grade 2 concussions also experience transient confusion and remain conscious, but signs and symptoms last longer than 15 minutes. A Grade 3 concussion implies loss of consciousness regardless of its duration.
The AAN published a practice parameter in Neurology in 1998.[2] This publication was intended to provide specific recommendations to coaches and sideline physicians for the assessment and treatment of injured athletes. Athletes with Grade 1 concussions should be removed from competition. They should be examined immediately and at 5-minute intervals for the development of mental status abnormalities or postconcussive symptoms both at rest and during exertion. Athletes may return to the arena if mental status abnormalities clear within 15 minutes. A second Grade 1 concussion during the same competition, however, precludes athletes from further competition during the same day. They may return to competition only if they remain asymptomatic at rest and during exercise for 1 week.
Athletes with Grade 2 concussions should be removed from competition for the remainder of the day. They should be examined onsite frequently for signs of increasing intracranial pressure and reexamined the next day. They should return to competition only after a physician who has performed a full neurological examination has cleared the athlete. This medical clearance should be obtained after the athletes have been asymptomatic at rest and during exertion for one full week. Before returning to competition, neuroimaging studies are recommended for all athletes whose headache or symptoms of postconcussive syndrome persist longer than 1 week. Any abnormality on CT or MR imaging terminates the season for the athletes. If athletes sustain a second Grade 2 concussion during the same season, they should not be allowed to compete until they have been asymptomatic at rest and during exertion for at least 2 weeks.
Athletes with Grade 3 concussions should be transferred from the field to the nearest emergency room by ambulance where a thorough evaluation should be performed. Athletes who are discharged home from the emergency department should be observed closely by a responsible adult for at least 24 hours or until all symptoms have resolved. Athletes with a Grade 3 concussion who lost consciousness for seconds only should be withheld from competition until they have been asymptomatic at rest and during exertion for at least 1 week. Athletes who lose consciousness for minutes should return to competition only after they have been asymptomatic at rest and during exertion for at least 2 weeks. Athletes who sustain a second Grade 3 concussion during the same season should not return to competition until they have been asymptomatic a minimum of one full month.
We believe that this grading scale is simple enough to be accepted as a standard tool for grading cerebral contusions regardless of the place of injury. Paramedics, firemen, policemen, and nurses could use it to assess minor closed head injuries at the scene. Like the GCS, this grading system permits personnel in the field to record and transmit a numerical value to emergency room personnel that can be used later to determine the incidence and severity of these lesions.
Conclusions
During the past 2,000 years, the concept of cerebral concussion has evolved from a simplistic view of a “shaken brain” to a more complex concept of axonal injury based on histological, neuroradiological, and neuropsychological evidence. This new perspective incorporates different grades of severity for so-called “minor” brain injuries that can range from totally reversible lesions to those associated with permanent neurological deficits and disability. The acceptance of a grading scale for cerebral concussions would standardize the rating of the severity of these injuries, an advance that would be valuable for clinical research and the evaluation of outcomes. A grading scale for cerebral concussions would also facilitate medicolegal and forensic determinations for claims that involve work injuries and motor vehicle accidents. Finally, a grading scale would eliminate vague terms such as mild, moderate, and severe, which are currently in use and impossible to quantify.
References
- Alexander MP: Mild traumatic brain injury: Pathophysiology, natural history, and clinical management. Neurology 45:1253-1260, 1995
- American Association of Neurology: Practice Parameter: The management of concussion in sports. Report of the quality standards subcommittee. Neurology 48:581-585, 1997
- Bruce DA, Alavi A, Bilaniuk L, et al: Diffuse cerebral swelling following head injuries in children: The syndrome of “malignant brain edema.”
J Neurosurg 54:170-178, 1981 - Cantu RC: Head and spine injuries in the young athlete. Clin Sports Med 7:459-472, 1988
- Crooks DA: The pathological concept of diffuse axonal injury in head trauma. J Pathol 165:5-10, 1991
- Dagi TF: The management of head trauma, in Greenblatt SH (ed): A History of Neurosurgery. Park Ridge, IL: American Association of Neurological Surgeons, 1997, pp 289-343
- Denny-Brown D, Russell WR: Experimental cerebral concussion. Brain 64:93-106, 1941
- Flamm ES: From signs to symptoms: The neurosurgical management of head trauma from 1517 to 1867, in Greenblatt SH (ed): A History of Neurosurgery. Park Ridge, IL: American Association of Neurological Surgeons, 1997, pp 65-81
- Goldstein M: Traumatic brain injury: A silent epidemic (editorial). Ann Neurol 27:327, 1990
- Goodrich JT: Neurosurgery in the ancient and medieval worlds, in Greenblatt SH (ed): A History of Neurosurgery. Park Ridge, IL: American Association of Neurological Surgeons, 1997, pp 37-64
- Gurdjian ES, Voris HC: Report of Ad Hoc Committee to study head injury nomenclature. Clin Neurosurg 12:386-387, 1966
- Jenkins A, Teasdale G, Hadley MD, et al: Brain lesions detected by magnetic resonance imaging in mild and severe head injuries. Lancet 2:445-446, 1986
- Jordan BD, Zimmerman RD: Computer tomography and magnetic resonance imaging comparisons in boxers. JAMA 263:1670-1674, 1990
- Katz DI, Alexander MP, Seliger GM, et al: Traumatic basal ganglia hemorrhage: Clinicopathologic features and outcomes. Neurology 39:897-904, 1989
- Kelly JP, Nichols JS, Filley CM, et al: Concussion in sports. Guidelines for the prevention of catastrophic outcome. JAMA 266:2867-2869, 1991
- Kelly JP, Rosenberg JH: Diagnosis and management of concussion in sports. Neurology 48:575-580, 1997
- Leininger BE, Gramling SE, Farrell AD, et al: Neuropsychological deficits in symptomatic minor head injury patients after concussion and mild concussion. J Neurol Neurosurg Psychiatry 53:293-296, 1990
- Levi L, Guilburd JN, Lemberger A, et al: Diffuse axonal injury: Analysis of 100 patients with radiological signs. Neurosurgery 27:429-432, 1990
- Levin HS, Williams DH, Eisenberg HM, et al: Serial MRI and neurobehavioural findings after mild to moderate closed head injury. J Neurol Neurosurg Psychiatry 55:255-262, 1992
- Lyons AS, Petrucelli RJ, II: Medicine. An Illustrated History. New York: Harry N. Abrams, 1978
- Newton MR, Greenwood RJ, Britton KE, et al: A study comparing SPECT with CT and MRI after closed head injury. J Neurol Neurosurg Psychiatry 55:92-94, 1992
- Ommaya AK, Gennarelli TA: Cerebral concussion and traumatic unconsciousness. Correlation of experimental and clinical observations of blunt head injuries. Brain 97:633-654, 1974
- Oppenheimer DR: Microscopic lesions in the brain following head injury. J Neurol Neurosurg Psychiatry 31:229-306, 1968
- Parodi CI, Cammarata S, Pizio N, et al: Traumatic basal ganglia hemorrhage with slight clinical signs and complete recovery (letter). J Neurol Neurosurg Psychiatry 55:72, 1992
- Povlishock JT, Becker DP, Cheng CLY, et al: Axonal change in minor head injury. J Neurol Neurosurg Psychiatry 42:225-242, 1986
- Rimel RW, Giordani B, Barth JT, et al: Disability caused by minor head injury. Neurosurgery 9:221-228, 1981
- Schneider RC: Football head and neck injury. Surg Neurol 27:507-508, 1987
- Sensmingler R: Hipocrates Aforismos. Mexico: Premia, 1989
- Spetzler RF, Wilson CB, Weinstein P, et al: Normal perfusion pressure breakthrough theory. Clin Neurosurg 25:651-672, 1978
- Teasdale G, Jennett B: Assessment of coma and impaired consciousness: A practical scale. Lancet 2:81-84, 1974
- Ward AA, Jr: The physiology of concussion. Clin Neurosurg 12:95-111, 1964
- Willberger JE, Jr., Rothfus WE, Tabas J, et al: Acute tissue tear hemorrhages of the brain: Computed tomography and clinicopathological correlations. Neurosurgery 27:208-213, 1990
- Yarnell PR, Lynch S: Retrograde memory immediately after concussion. Lancet 1:863-864, 1970
