
Functional MR Imaging During Finger Tapping
Sterling C. Johnson, PhD
George P. Prigatano, PhD
Division of Neurology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
To examine the rapid dynamic cerebral changes associated with increasingly difficult motor tasks, functional magnetic resonance imaging studies of eight healthy normal adults were obtained as they performed a version of the Halstead Finger Oscillation (Tapping) Test. While undergoing whole brain echo-planar imaging, subjects were asked to tap their right hand for 15 seconds alternating with 15 seconds of rest. Over the course of the 150-second experiment, the task became more difficult as the muscles fatigued with the repetitive, rapid finger movement. A motor map typical of simple hand-motor experiments, including activations in the contralateral motor cortex and ipsilateral cerebellum, and, to a lesser extent, in the supplementary motor area and ipsilateral motor cortex, was obtained. The effect of task difficulty, as assessed by statistical analysis of the interaction of time and tapping, increased activation in regions of the contralateral and ipsilateral primary motor cortex, left lateral premotor cortex, and left dorsolateral prefrontal cortex. The results highlight the rapid modifiability of motor systems and have implications for the study of recovery of function after stroke or trauma.
Key Words: functional magnetic resonance imaging, Halstead Finger Tapping Test, motor cortex, neuropsychology, neurorehabilitation
Numerous studies have documented the potential usefulness of the Halstead Finger Tapping Test (HFTT)[30] as a clinical neuropsychological measure.[18,25,26,37] Finger tapping has been related to the severity of traumatic brain injury (TBI),[6] the lateralization of cerebrovascular accidents,[26] and recovery of function after mild and moderate TBIs.[14] The HFTT is very sensitive to the effects of aging[37] and related to disorders of impaired awareness after brain injury.[25] Finger tapping has even been related to employability after various forms of brain injury.[7,15] But what brain structures are key to performing this “simple” task, and do patterns of cerebral activation change as the task progresses? For example, many patients report fatigue after several trials of tapping. Are certain patterns of brain activation associated with such effort?
The primary motor cortex has long been thought of as having a modifiable architecture,[1,17,35] and recent animal studies have shown that motor cortex is capable of rapid (on the order of minutes) and long-lasting reorganization.[8-10,36] Such plasticity may partially underlie functional recovery after the motor cortex has been injured by stroke or trauma. The mechanism may include acquired function in intact perilesional tissue or remote motor regions such as contralesional homologous motor cortex.[20,21] Recent functional imaging studies of patients who have suffered unilateral stroke support these ideas.[4,31]
In the past decade, the functional neuroanatomy of the human motor system has been well defined with functional magnetic resonance (fMR) imaging.[24,28,29] However, little work on the rapid plasticity of the human motor cortex as reflected by fMR imaging has been reported. Existing studies focus on motor learning rather than motor effort.[2,3,16,20,24,33,34,38] In a study of repetitive hand motor movement, Samuel etal.[33] found that activation of the rostral supplementary motor area decreased as the task progressed. That task, however, did not involve increasing difficulty or fatigue.
In pilot studies of the HFTT, individuals found it slightly more difficult to finger tap in the scanner than in a standard clinical setting because of the magnetic effect on the spring/lever. It was also much more difficult to perform the task at the end of the experiment than at the beginning. These findings suggested that areas of the brain activated as the difficulty of task increased could be examined. Although other studies have examined rapid cerebral change associated with motor learning, this report examined the changes that occurred as the difficulty of the task increased. Activation patterns similar to those found in other basic motor paradigm studies were expected,[19,27,29] as were changes in the pattern of activation within the motor system as the demands of the task increased over the course of the experiment.[11]
Materials and Methods
Subjects
Eight healthy normal volunteers underwent fMR imaging while performing the HFTT as described below. Their mean age was 32 years [standard deviation (SD), 7.9 years] and their mean level of education was 18 years (SD, 1.3 years). Four participants were female and six were right-handed. All subjects were screened to rule out preexisting conditions (e.g., major psychiatric, neurological, or medical disease, including prior head trauma and high blood pressure) that could have affected the results.
Procedure
A modified HFTT procedure was used. While undergoing echoplanar imaging, subjects tapped with their right index finger as fast as possible on the HFTT device. Each trial was 15 seconds and alternated with 15 seconds of rest. Five cycles (consisting of 15 seconds of tapping and 15 seconds of rest) were completed with the right hand and followed by five cycles with the left hand during the same scanning session. Use of the left hand was associated with considerable task-correlated motion artifact, and those data were not analyzed.
Image Acquisition
A gradient-echo, echo-planar pulse sequence with the following parameters was used: echo delay (TE)=40, repetition time (TR)=3000 ms, number of repetitions=105, flip angle=90Þ, acquisition matrix=64×64, field of view=240 mm, and slice thickness=3.8 mm. Thirty-six slices of the brain were acquired axially at 105 contiguous time points with near isotropic voxel resolution of 3.75×3.75×3.8 mm.
Postprocessing of fMRI Data
Images were processed and analyzed using SPM99, developed at the University College of London (www.fil.ion.ucl.ac.uk). For each data set, images were realigned to correct for subvoxel motion, normalized to the Talairach standard atlas space as defined by the International Consortium for Brain Mapping, and spatially smoothed with a Gaussian filter to 8 mm (full width at half maximum). This procedure compensated for anatomical variation between subjects and conformed with appropriate statistical inferences using the theory of random fields.[39]
Statistical Analyses
For this relatively small sample, a fixed-effect group analysis[12] was performed using the general linear model.[13] This approach permits the main effect of tapping versus resting as well as the linear increases and decreases in activity over the course of the experiment (known as time-by-condition interactions) to be examined.[38] For the robust main effect of tapping versus rest, Bonferroni correction was used to address the problems associated with multiple comparisons. The corrected probability threshold was set as T=4.81. For the less robust time-by-condition interactions, an uncorrected threshold of T=2.34 was used.
The latter comparison differs from most conventional analyses. Typically, fMR imaging studies have examined only the agreement of voxel behavior with active and control conditions. These analyses assume that a consistent steady state is reached during each active epoch. Given the accumulating evidence that the brain adapts quickly to task demands (in seconds to minutes), this assumption may not always be valid. Therefore, the interaction analysis seemed appropriate to model explicitly any change in the brain’s response to the motor task over the course of the 2.5 minutes of right-hand movement versus resting.
Results
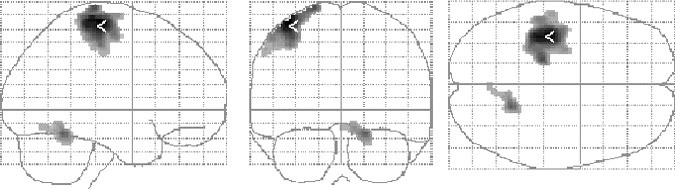
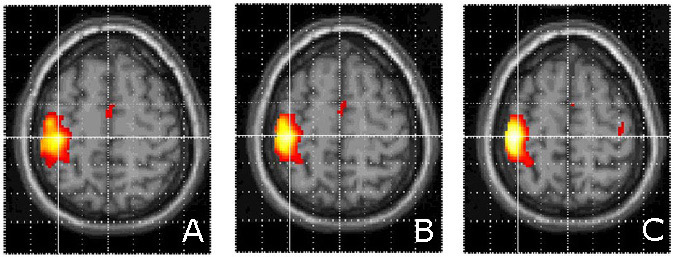
All voxels that survived a stringent threshold (T=4.81) during finger-tapping with the right hand are shown in Figure 1. The clusters corresponded to the M1 hand motor cortex and ipsilateral cerebellum. Activations at a lower threshold (T=3.12) were superimposed on a representative anatomical scan (Fig. 2). Other areas within the cortical motor system, including the supplementary motor and ipsilateral primary motor cortex, were activated as is typical of simple motor activation paradigms.
Task Difficulty
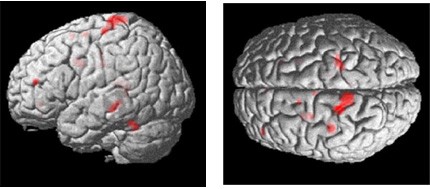
All subjects reported that the task was more difficult at the end of the 150-second experiment than it was at the beginning. Thus, the data were analyzed to determine the brain regions that were associated with task difficulty by introducing into the model a linearly increasing covariate over the time course of the experiment. The results revealed interactions in the left motor cortex (Talairach coordinates, –20, –28, 72) just dorsal to the main effect. Other regions included ipsilateral (right) motor cortex (22, –30, 64), left lateral premotor (–58, –16, 42), and left dorsolateral prefrontal cortex (–40, 46, 16). These effects are illustrated in Figures 3 and 4.
Discussion
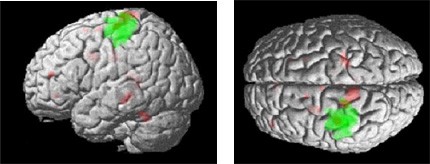
These results highlight the normal neuroanatomy underlying right hand motor function as well as interesting areas of dynamic change that occurred as the task became more difficult over the course of the 150-second experiment. As with prior studies of simple motor paradigms, the primary motor network, including primary motor cortex, lateral premotor cortex, supplementary motor cortex, and cerebellum, was activated. The search for dynamic increases in activity over the course of the experiment demonstrated extended activation in M1 in both the contralateral and ipsilateral cortex. Activity was also increased in the left lateral premotor and left dorsolateral prefrontal cortex.
These results support earlier suggestions that area M1 exhibits plasticity within a brief period,[22,23] in this case, within 150 seconds of strenuous, right-handed, repetitive finger tapping. These findings are also consistent with a recent positron emission tomography (PET) study that correlated frontal lobe activity with level of exertion.[5]
To our knowledge this is the first report of fMR imaging obtained while subjects performed the HFTT. The observed pattern of activation may help explain certain clinical findings. The cortex was activated bilaterally when normal subjects performed the HFTT, perhaps partially explaining why unilateral cerebral lesions, particularly during the acute stages of recovery, can be associated with bilateral slowing on this task.[26] Second, multiple regions in the frontal cortex seem to become activated during rapid finger tapping as fatigue develops over repeated trials. With head injuries damage to the frontal cortex is often widespread.[40] Patients with such injuries often finger tap slowly[6] and exhibit poor self-awareness.[25] Perhaps patients with impaired self-awareness tend to finger tap slowly because both dimensions are related to diffuse underlying pathology of the frontal lobe. Further studies are needed to assess patterns of cerebral activation in brain-injured patients who show impaired self-awareness and who finger tap slowly.
Several PET and fMR imaging-activation studies have examined motor recovery after stroke (reviewed elsewhere[32]). This is an important area of research because a better understanding of how the brain recovers from an insult could enhance the timing and content of neurorehabilitation interventions. This new body of research still lacks longitudinal studies of recovery studied with the high temporal and spatial resolution of fMRI. A similar method to the one employed here could be used to detect longitudinal changes several months to years after stroke.
The HFTT paradigm requires subjects to tap the lever as fast as possible. Undoubtedly, subjects tapped at different rates. For this preliminary investigation, performance data to determine each participant’s rate of tapping could not be collected in the scanner. This point is of concern because the rate of tapping can affect the magnitude and extent of brain activation.[27] Additionally, our HFTT device was slightly more difficult to use in the magnetic field than in a standard clinical setting. Unfortunately, due to the constraints of the magnetic field, we were unable to measure the force required to depress the tapping device in the scanner. Furthermore, the sample was not highly representative of the normal population in terms of their level of education and the extent of left-handedness in the sample. Normative data on the HFTT suggest that normal individuals achieve 5 to 10% fewer taps with their nondominant hand, a tendency that could have influenced the results of this preliminary study. These limitations are currently being addressed.
Acknowledgments
We gratefully acknowledge the assistance of James G. Pipe, PhD, Joseph E. Heiserman, MD, PhD, and Stephen F. Flitman, MD, for their assistance with the fMR imaging and Sarah Hahn, PhD, and Lanie Zigler, PhD, for recruiting subjects.
References
- Brown T, Sherrington C: On the instability of a cortical point. Proc R Soc London Serv B 85:250-277, 1912
- Classen J, Liepert J, Hallett M, et al: Plasticity of movement representation in the human motor cortex. Electroencephalogr Clin Neurophysiol Suppl 51:162-173, 1999
- Classen J, Liepert J, Wise SP, et al: Rapid plasticity of human cortical movement representation induced by practice. J Neurophysiol 79:1117-1123, 1998
- Cramer SC, Moore CI, Finklestein SP, et al: A pilot study of somatotopic mapping after cortical infarct. Stroke 31:668-671, 2000
- Dettmers C, Lemon RN, Stephan KM, et al: Cerebral activation during the exertion of sustained static force in man. Neuroreport 7:2103-2110, 1996
- Dikmen S, Machamer J, Winn H, et al: Neuropsychological outcome at 1-year post head injury. Neuropsychology 9:80-90, 1995
- Dikmen SS, Temkin NR, Machamer JE, et al: Employment following traumatic head injuries. Arch Neurol 51:177-186, 1994
- Donoghue JP: Plasticity of adult sensorimotor representations. Curr Opin Neurobiol 5:749-754, 1995
- Donoghue JP: Limits of reorganization in cortical circuits. Cereb Cortex 7:97-99, 1997
- Donoghue JP, Suner S, Sanes JN: Dynamic organization of primary motor cortex output to target muscles in adult rats. II. Rapid reorganization following motor nerve lesions. Exp Brain Res 79:492-503, 1990
- Friston KJ: Analysing brain images: Principles and overview, in Frackowiak RSJ, Friston KJ, Frith CD, et al. (eds): Human Brain Function. London: Academic Press, 1997, pp 25-42
- Friston KJ, Holmes AP, Price CJ, et al: Multisubject fMRI studies and conjunction analyses. Neuroimage 10:385-396, 1999
- Friston KJ, Holmes AP, Worsley KJ, et al: Statistical parametric maps in functional imaging: A general linear approach. Human Brain Mapping 2:189-210, 1995
- Haaland KY, Temkin N, Randahl G, et al: Recovery of simple motor skills after head injury. J Clin Exp Neuropsychol 16:448-456, 1994
- Heaton RK, Chelune GJ, Lehman RA: Using neuropsychological and personality tests to assess the likelihood of patient employment. J Nerv Ment Dis 166:408-416, 1978
- Karni A, Meyer G, Jezzard P, et al: Functional MRI evidence for adult motor cortex plasticity during motor skill learning. Nature 377:155-158, 1995
- Lashley KS: Temporal variation in the function of the gyrus precentralis in primates. Am J Physiol 65:585-602, 1923
- Lezak MD: Neuropsychological Assessment. New York: Oxford University, 1995
- Mattay VS, Weinberger DR: Organization of the human motor system as studied by functional magnetic resonance imaging. Eur J Radiol 30:105-114, 1999
- Nudo RJ: Recovery after damage to motor cortical areas. Curr Opin Neurobiol 9:740-747, 1999
- Nudo RJ, Milliken GW: Reorganization of movement representations in primary motor cortex following focal ischemic infarcts in adult squirrel monkeys. J Neurophysiol 75:2144-2149, 1996
- Pascual-Leone A, Grafman J, Hallett M: Modulation of cortical motor output maps during development of implicit and explicit knowledge. Science 263:1287-1289, 1994
- Pascual-Leone A, Tarazona F, Catala MD: Applications of transcranial magnetic stimulation in studies on motor learning. Electroencephalogr Clin Neurophysiol Suppl 51:157-161, 1999
- Passingham R: Functional organisation of the motor system, in Frackowiak R, Friston K, Frith C, et al (eds): Human Brain Function. San Diego: Academic Press, 1997, pp 243-274
- Prigatano GP: Impaired awareness, finger tapping, and rehabilitation outcome after brain injury. Rehabilitation Psychology 44:145-159, 1999
- Prigatano GP, Wong JL: Speed of finger tapping and goal attainment after unilateral cerebral vascular accident. Arch Phys Med Rehabil 78:847-852, 1997
- Rao SM, Bandettini PA, Binder JR, et al: Relationship between finger movement rate and functional magnetic resonance signal change in human primary motor cortex. J Cereb Blood Flow Metab 16:1250-1254, 1996
- Rao SM, Binder JR, Bandettini PA, et al: Functional magnetic resonance imaging of complex human movements. Neurology 43:2311-2318, 1993
- Rao SM, Binder JR, Hammeke TA, et al: Somatotopic mapping of the human primary motor cortex with functional magnetic resonance imaging. Neurology 45:919-924, 1995
- Reitan RM, Wolfson D: The Halstead-Reitan Neuropsychological Test Battery: Theory and Clinical Interpretation. Tucson, AZ: Neuropsychology Press, 1993
- Rossini PM, Caltagirone C, Castriota-Scanderbeg A, et al: Hand motor cortical area reorganization in stroke: A study with fMRI, MEG and TCS maps. Neuroreport 9:2141-2146, 1998
- Rowe JB, Frackowiak RS: The impact of brain imaging technology on our understanding of motor function and dysfunction. Curr Opin Neurobiol 9:728-734, 1999
- Samuel M, Williams SC, Leigh PN, et al: Exploring the temporal nature of hemodynamic responses of cortical motor areas using functional MRI. Neurology 51:1567-1575, 1998
- Samuel M, Williams SCR, Leigh PN, et al: An fMRI study of the temporal course of cortical motor area activation, in American Academy of Neurology (ed): 49th Annual Meeting of the American Academy of Neurology. Boston: American Academy of Neurology, 1997, 48.
- Sanes J, Donoghue J: Plasticity and primary motor cortex. Ann Review Neuroscience 23:393-415, 2000
- Sanes JN, Suner S, Donoghue JP: Dynamic organization of primary motor cortex output to target muscles in adult rats. I. Long-term patterns of reorganization following motor or mixed peripheral nerve lesions. Exp Brain Res 79:479-491, 1990
- Spreen O, Strauss E: A Compendium of Neuropsychological Tests. New York: Oxford University, 1998
- Toni I, Krams M, Turner R, et al: The time course of changes during motor sequence learning: A whole-brain fMRI study. Neuroimage 8:50-61, 1998
- Worsley KJ, Marrett S, Neelin P, et al: A unified statistical approach for determining significant signals in images of cerebral activation. Human Brain Mapping 4:58-73, 1996
- Zimerman RA, Bilaniuk LT: CT and MR: Diagnosis and evolution of head injury, stroke, and brain tumors. Neuropsychology 3:191-230, 1989
