
Cerebral Amyloid Angiopathy: Review and Case Report
Hamidreza Aliabadi, MD*
Gerard Chamberlin, MPh**
Murray Flaster, MD, PhD**
Divisions of *Neurological Surgery and **Neurology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
A 75-year-old woman with a history of coronary artery disease, hyperlipidemia, and early dementia was found on the floor next to her bed but had no recollection of the event. She exhibited a flat affect, inappropriate cheerfulness, and mild right visual neglect. At an outside institution, computed tomography of the head showed a 0.9-cc intraparenchymal hemorrhage. She was transferred to our institution where her motor and sensory examinations were normal. Her score on the Mini Mental State Examination was 25. She exhibited cognitive deficits consistent with mild dementia. Further diagnostic imaging indicated that she had experienced a third lobar hemorrhage; the previous two hemorrhages had been unrecognized. Her case is presumed to represent hemorrhage caused by cerebral amyloid angiopathy, the clinical diagnosis of which is reviewed.
Key Words: Alzheimer’s disease, cerebral amyloid angiopathy, dementia, intracerebral hemorrhage, intraparenchymal hemorrhage
Cerebral amyloid angiopathy is characterized by the accumulation of amyloid in the adventitia and media of small and mid-sized vessels. This microangiopathy has a predilection for leptomeningeal and parenchymal arteries, particularly of the parietal and occipital lobes, but can occasionally be found in veins.[6] Cerebral amyloid angiopathy has been found in neurologically normal individuals, and the angiopathy increases with age. It often is concomitant with Alzheimer’s disease and has been described as a morphologic feature of this disease. We report a patient whose intraparenchymal hemorrhage, comprehensive neuroimaging, and clinical condition support the diagnosis of presumed cerebral amyloid angiopathy.
Illustrative Case
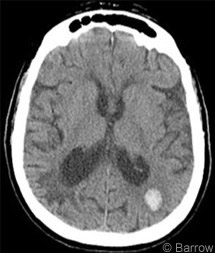
A 75-year-old woman with a history of coronary artery disease, hyperlipidemia, and early dementia was found on the floor next to her bed. By ambulance she was transported to the emergency room of an outside hospital where computed tomography (CT) of the head showed an acute 1.0×1.2×1.5-cm left parietal intraparenchymal hemorrhage (Fig.1). Prominent small vessel disease was present in the periventricular white matter, and chronic changes of encephalomalacia were present in the right parietal lobe. No significant mass effect, hydrocephalus, or collections of extraaxial fluid were present.
After a myocardial infarction was ruled out, the patient was transferred to our institution for further neurosurgical evaluation. On arrival, she was awake, alert, and oriented to person, place, time, and situation. However, she was amnestic to the event. Her home medications were lopressor, digoxin, and aspirin. She had no known drug allergies. Her surgical history was notable for a hysterectomy. She was married and lived at home with her husband. She drank alcohol only occasionally and used no tobacco products.
Her mental examination was consistent with mild-to-moderate pre-existing dementia. Her score on the Mini Mental State Examination was 25 of 30 points. She showed deficits in memory recall and drawing of intersecting images. She had difficulty (although minimal) spelling “world” backward (4 of 5 points obtained on this part of the examination). She recalled none of four words after 2 minutes.
The patient’s eyes opened spontaneously, and she verbalized without difficulty or dysphasia. She followed all commands briskly and bilaterally. Her Glasgow Coma Scale score was 15 (E4, V5,M6). Her pupils were equally round and reactive to light. Her extraocular muscle and cranial nerve functions were intact, and her face was midline. She had no recollection of a previous stroke or hemorrhage and had no evidence of prior deficits. Her sensory and motor examinations were normal. Deep tendon reflexes were +2 throughout.
Her metabolic profile was as follows: sodium, 138 mEq/L; potassium, 3.7 mEq/L; chloride, 106 mEq/L; bicarbonate, 26 mEq/L;blood-urea-nitrogen level,13 mg/dL;creatinine,0.7 mg/dL; and glucose, 102 mg/dL. Her white blood cell count was 7.1 x 103/mL, hemoglobin was 12.6 gm/dL, hematocrit was 37.2 %, and platelets were 198 x 103/mL. Her coagulation panel was normal, with an international normalized ratio of 0.9, prothrombin time of 10.4 sec, and partial thromboplastin time of 25 sec. Her bleeding time was normal (3 minutes),and her level of thyroid stimulating hormone was normal (3.283 UIU/mL). Her level of vitamin B12 was low (92 pg/mL).
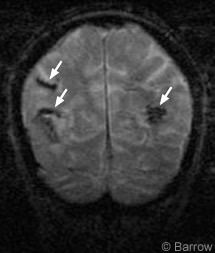
Transthoracic echocardiography showed that the size of the left ventricle was normal as was the ejection fraction. Aortic sclerosis and slight mitral annular calcification were present. Electroencephalography revealed slowing over both hemispheres with the left hemisphere slower than the right. Magnetic resonance (MR) imaging of the brain and MR angiography of the head and neck showed no acute ischemic events, the hemorrhage as expected from her CT scan, and significant stenosis of the inferior division of the right middle cerebral artery. The MR angiography study of the neck was normal. A coronal gradient-echo MR imaging study showed two previous lobar areas of magnetic susceptibility consistent with prior intraparenchymal hemorrhages, indicating that this episode was the patient’s third parenchymal hemorrhage (Fig.2).
Neuropsychological evaluation revealed severe impairment in basic memory, learning, visuoperceptual organization, and problem-solving with more moderate deficits in attention, concentration, and visual scanning. She had constructional dyspraxia with both hands and had “limited insight.” These findings were compatible with the possible diagnosis of mild-to-moderate preexisting dementia.
Discussion
Clinical Manifestations and Diagnosis
Clinically, cerebral amyloid angiopathy often manifests as intracerebral hemorrhage (ICH) and accounts for as many as 15% of all ICHs in patients older than 60 years and as many as half of nontraumatic lobar ICHs in patients older than 70 years.[5] Amyloid deposits weaken the vessel walls and increase the risk of microaneurysms and hemorrhage.[3] ICH can manifest with a variety of symptoms, including headache, vomiting, partial seizures, and coma. Confusion and transient neurological events such as weakness, paresthesia, or visual misperceptions have also been associated with cerebral amyloid angiopathy.[5] Unless cerebral amyloid angiopathy manifests with ICH, it is usually diagnosed postmortem by the microscopic demonstration of amyloid beta protein in vessel walls.[6]
During life cerebral amyloid angiopathy can only be diagnosed definitively by biopsy and histological examination.[4] Our patient’s case elucidates a typical presentation of a presumed amyloid hemorrhage without the luxury of a surgical specimen. Her case history, physical examination, and imaging results are classic for a clinical diagnosis of cerebral amyloid angiopathy. In neurosurgically treated cases, samples of brain tissue can be obtained for microscopic examination during evacuation of a hematoma or a cortical biopsy.
Otherwise, tissue is obtained from postmortem studies. Amyloid beta protein stains eosinophilic under light microscopy. When viewed under polarized light after staining with Congo red, the protein yields the apple-green birefringence characteristic of amyloid. Staining with thioflavin T and S results in the amyloid beta protein fluorescing when exposed to ultraviolet light.[5]
Treatment
There is no definitive treatment for cerebral amyloid angiopathy. However, patients with such hemorrhages are treated like any patient with an intraparenchymal hemorrhage. The goals of treatment are to prevent complications from developing, to maintain normal intracranial pressure, and to reverse anticoagulation to prevent extension of the hemorrhage. When co-existing vasculitis is present, patients can be treated with long-term steroids and immunomodulants such as cyclophosphamide.[5] Avoidance of aspirin and nonsteroidal anti-inflammatory agents with the possible exception of cox-2 inhibitors is advisable.
Surgical treatment in these patients is controversial. A deteriorating neurological status, intractable intracranial hypertension, and the size of the hemorrhage are major factors. We typically estimate the volume of the hemorrhage to help determine the need for surgery. Patients with a hemorrhagic volume between 20 and 60 cc who exhibit a progressive decline in consciousness may be good candidates for surgical evacuation of the hematoma.[5] Otherwise, conservative treatment and medical management are keys in treating hemorrhage related to cerebral amyloid angiopathy. Neuropsychological testing helps assess for the presence of cognitive impairment.
Our patient was deficient in vitamin B12. Given that a B12 deficiency is associated with a reversible form of dementia, her levels were corrected with injectable vitamin B12. However, her primary dementia appeared to have been related to Alzheimer’s disease or multivascular disease.
Cerebral Amyloid Angiopathy and Dementia
As many as 85% of individuals with Alzheimer’s disease are thought to have cerebral amyloid angiopathy.[5] In fact, the amyloid beta protein deposited in the vessels of individuals with cerebral amyloid angiopathy is derived from the amyloid precursor protein, as is the protein found in the core of the neuritic plaques associated with Alzheimer’s disease.[1]
Alzheimer’s disease and cerebral amyloid angiopathy can occur independently of each other, and no causality between the two diseases has been determined. However, the relationship between the two conditions may be related to similar risk factors. The apolipoprotein E epsilon 4 (ApoE E4) allele has been linked to early ICH and Alzheimer’s disease.[2] Individuals who are homozygous for the ApoE E4 allele are at much greater risk of later developing Alzheimer’s disease than those who are not.
The link between dementia and cerebral amyloid angiopathy has not been completely elucidated. In the Honolulu-Asia Aging Study, Pfiefer et al. analyzed the autopsy data of Japanese-American men.[6] Those with Alzheimer’s disease and cerebral amyloid angiopathy performed worse on the Cognitive Abilities Screening Instrument than men with only Alzheimer’s disease. Thus, cognitive impairment may be worsened by the presence of both cerebral amyloid angiopathy and Alzheimer’s disease. However, conflicting results from other studies failed to find an association between cognitive function and cerebral amyloid angiopathy. The variability of results could be related to different methods of assessing cognitive function or to intrinsic differences among the populations studied.[6] More research is needed to understand the impact of cerebral amyloid angiopathy on Alzheimer’s disease and dementia.
References
- Cotran RS, Kumar V, Collins T: Robbins Pathologic Basis of Disease. Philadelphia: W.B. Saunders, 1999
- Greenberg SM, Briggs ME, Hyman BT, et al: Apolipoprotein E epsilon 4 is associated with the presence and earlier onset of hemorrhage in cerebral amyloid angiopathy. Stroke 27:1333-1337, 1996
- Hershey LA: Cerebrovascular disease, in Duthie EH, Jr., Katz PR (eds): Practice of Geriatrics. Philadelphia: W.B. Saunders, 1998, pp 328-335
- Knudsen KA, Rosand J, Karluk D, et al: Clinical diagnosis of cerebral amyloid angiopathy: Validation of the Boston criteria. Neurology 56:537-539, 2001
- Merino JG, Hachinski VC: Amyloid angiopathy. www.emedicine.com/neuro/topic628.htm, 2003 (This website link is provided for your convenience only. Barrow Neurological Institute neither endorses nor is responsible for the content on the site in any way.)
- Pfeifer LA, White LR, Ross GW, et al: Cerebral amyloid angiopathy and cognitive function: The HAAS autopsy study. Neurology 58:1629-1634, 2002
