Acoustic Neuromas: Symptoms and Diagnosis
Louis J. Kim, MD
Jeffrey D. Klopfenstein, MD
Randall W. Porter, MD
Mark J. Syms, MD*
Division of Neurological Surgery and *Section of Neurotology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Download a .pdf of this article.
Abstract
Acoustic neuromas are the most common cerebellopontine angle tumor in adults. While their natural history is not fully understood, a subset of tumors demonstrates significant growth over time, necessitating treatment. Clinical manifestations include hearing loss, tinnitus, vertigo, dysequilibrium, and cranial nerve neuropathies. Late findings include brainstem compression, hydrocephalus, and facial paresis. Audiometric testing is useful for diagnostic screening and pretreatment planning. Magnetic resonance imaging is the diagnostic gold standard. Its widespread use has led to increasingly earlier diagnosis of acoustic neuromas, including asymptomatic lesions.
Key Words: acoustic neuroma, audiometry, radiological imaging, sensorineural hearing loss
A thorough understanding of the clinical manifestations of acoustic neuromas is essential for diagnosis. Once this lesion is suspected, the appropriate diagnostic tests are vital for treatment planning and pretreatment discussions with the patient. This article reviews the clinical presentation and diagnostic evaluation of acoustic neuromas.
Epidemiology
Acoustic neuromas account for about 75% of tumors discovered in the cerebellopontine angle (CPA). Their prevalence and incidence remain unclear.[15] Schuknecht analyzed 1400 temporal bone specimens and found a 0.57% incidence of occult acoustic neuromas, or about 570 tumors per 100,000 people.[28] In another temporal bone study, Leonard and Talbot[20] found a 0.8% incidence of occult acoustic neuromas. However, the 1991 National Institutes of Health Consensus Statement estimated 2,000 to 3,000 new, clinically apparent cases of unilateral acoustic neuromas each year,[1] or an incidence of about one tumor per 100,000 per year.
Given the disparate data between occult postmortem lesions and clinically apparent lesions, the true incidence of acoustic neuromas is thought to range between these two numbers. A large number of clinically silent tumors probably never require medical attention during an individual’s lifetime. With the widespread use of magnetic resonance (MR) imaging, however, the diagnosis of clinically occult lesions will increase. The approach to these diagnosed but clinically silent lesions will rely heavily on our understanding of the natural history of these tumors.
Natural History
Deen et al.[7] followed 68 elderly patients (mean age, 67.1 years) with acoustic neuromas conservatively for a mean of 3.4 years; 71% demonstrated no tumoral growth. Eventually 15% required treatment. Glasscock and coworkers[13] followed 34 patients for a mean of 2.4 years. The mean growth rate of 56% of the tumors was 2.9 mm/year. Mirz et al.[21] reported 50 patients. The mean growth rate of 64% of the tumors was 1.6 mm/ year. During a mean follow-up of 4.8 years in 80 patients with no prior treatment, 42% exhibited no growth.[26] The overall growth rate in these nonsurgical patients was 0.91 mm/year. Among 49 patient who underwent subtotal surgical resection, 68.5% exhibited no new growth.[26] The overall growth rate in this group was 0.35 mm/year. Three patients (6.1%) subsequently required reoperation due to regrowth. Yamakami et al.[33] reported 903 patients followed a mean of 3.1 years: 51% exhibited an average growth rate of 1.87 mm in the first year, and 20% subsequently required surgical intervention.
A cadre of small, retrospective, natural history studies has been reported, but long-term prospective data are lacking. Therefore, the exact natural history of acoustic neuromas remains uncertain. A subset of tumors appears subject to rapid growth while another subset exhibits no growth over time. Based on current studies, however, it is reasonable to use serial imaging studies for small tumors, particularly in patients over 65 years. If rapid growth, new symptoms, or both are identified, intervention can be instituted.
Clinical Manifestations
In 1917 Harvey Cushing reviewed his acoustic neuroma series and extrapolated a progression of neurological symptoms orresponding to tumor enlargement: gradual auditory and labyrinthine dysfunction, occipitofrontal pain, cerebellar ataxia, adjacent cranial nerve palsies, increased intracranial pressure, dysphagia, dysarthria, and brainstem compression with respiratory compromise.[6]
Jackler and Pitts[17] used three stages to describe acoustic neuroma growth in anatomical terms. The canalicular stage, characterized by hearing loss, tinnitus, and vertigo, occurs during early growth of the tumor from the lateral fundus of the internal acoustic canal to the porus acusticus. Early displacement of cranial nerves against the bony wall of the canal occurs. During the cisternal stage, auditory and vestibular function progressively declines and headache occurs from dural irritation. The tumor grows from the porus acusticus into the 1- to 2-cm subarachnoid cistern adjacent to the brainstem. The facial and vestibulocochlear nerves and anterior inferior cerebellar artery are displaced progressively. Progressive nerve dysfunction results when tumor growth in the CPA exceeds that of the portion in the internal auditory canal and the facial nerve is stretched over the bony anterior lip of the porus. Late in this stage as the trigeminal nerve becomes distorted, midfacial and corneal anesthesia can manifest. The brainstem compressive stage is demarcated by tumor growth extensive enough to displace brainstem structures, to obstruct the fourth ventricle, to deform the trigeminal nerve and tentorium superiorly, and to deform the nerves of the jugular foramen inferiorly. At this stage, facial twitch and weakness, worsening headache, papilledema, and diplopia related to obstructive hydrocephalus may occur.
Selesnick et al.[30] correlated symptoms with duration of their onset before diagnosis. Tinnitus and hearing loss occurred 4 years before diagnosis, vertigo 3.6 years earlier, headache 2.2 years earlier, dysequilibrium 1.7 years earlier, trigeminal symptoms 0.9 years earlier, and facial nerve symptoms 0.6 years earlier. The stages of acoustic neuroma growth also were correlated with symptoms. The canalicular stage occurred 4 years before diagnosis, the cisternal stage 1.5 years later, and the brainstem compressive stage 0.9 years later. The authors stress the importance of the 1.5-year gap between the canalicular and cisternal stages, during which earlier diagnosis might permit treatment of a considerably smaller and less symptomatic tumor.
Hearing Loss
Progressive, high-frequency unilateral or asymmetric sensorineural hearing loss, the most common symptom of acoustic neuromas, is reported to occur in more than 95% of patients.[15,29] It usually develops over months to years and is associated with impairment of speech disproportionate to the pure tone loss. In 10% of cases, sudden hearing loss occurs and is attributable to the vascular interruption of the internal auditory artery.[5,17] Based on audiometry, speech discrimination scores, and speech reception thresholds, normal hearing has been observed in 3 to 15% of acoustic neuroma cases.[4,25] In these patients, dysequilibrium was the most common symptom.[27] The mechanism for hearing loss is unclear, but it appears to be related to the compressive effects of tumor on the vestibulocochlear nerve with resultant injury to neuronal elements, vascular compromise, or both.
 The Gardner-Robertson hearing classification system was established to standardize the measurement of preoperative and postoperative hearing (Table 1).[12] Hearing is assessed using speech reception thresholds and speech discrimination scores. In this 5-point grading scale, useful hearing is scored as I or II, nonuseful hearing (absent speech discrimination) as III or IV, and complete hearing loss as V.
The Gardner-Robertson hearing classification system was established to standardize the measurement of preoperative and postoperative hearing (Table 1).[12] Hearing is assessed using speech reception thresholds and speech discrimination scores. In this 5-point grading scale, useful hearing is scored as I or II, nonuseful hearing (absent speech discrimination) as III or IV, and complete hearing loss as V.
The Committee on Hearing and Equilibrium of the American Academy of Otolaryngology-Head and Neck Surgery has also provided guidelines for reporting pre- and postoperative hearing function (Table 2). According to these recommendations, hearing thresholds are reported as the average of puretone hearing thresholds by air conduction at 0.5, 1, 2 and 3 kHz. In addition, speech discrimination at levels of 40 dB or maximum comfortable loudness should be documented pre- and postoperatively. [2]
Tinnitus
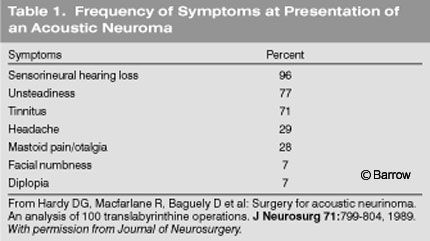
According to Hardy et al.,[15] tinnitus is associated with 71% of acoustic neuromas at presentation. Unlike hearing loss, tinnitus has a low impact on patient functioning; therefore, it rarely serves as the impetus to seek medical attention. Nonetheless, unilateral tinnitus without obvious cause warrants investigation of the auditory brainstem response (ABR) or MR imaging. Tinnitus in the absence of hearing loss is extremely rare.[10] The pathophysiology of tinnitus associated with acoustic neuromas is thought to be similar to that of hearing loss, that is, neural or vascular compression.
Vertigo
Vertigo, the illusion of motion, has been reported in 58% of patients.[24] According to a University of California—San Francisco (UCSF) series, vertigo occurred in 19% of patients overall but was inversely proportional to the size of the tumor. Vertigo was present in 27% of patients with tumors less than 1 cm compared to 19% of patient with tumors 1 to 3 cm and 10% of patients with tumors larger than 3 cm. Dysequilibrium, the sense of floating or unsteadiness, was more common (48% overall),[29] and its incidence was proportional to the size of the tumor.
Given that acoustic neuromas arise from the vestibular nerves, it is paradoxical that vertigo is not a more consistent finding. The pathophysiology of vertigo is believed to be related to the compressive effects of the tumor on the vestibular nerve or vascular supply. Because nerve attenuation usually is gradual, the loss of function is readily compensated for by the contralateral vestibular system. Thus, symptoms of vertigo are usually mild unless a large tumor also affects cerebellar functioning.
Cerebellar Dysfunction
Cerebellar symptoms, including incoordination, ataxia, and dysequilibrium, usually occur late in the course of tumor growth. Symptoms tend to be continuous and unremitting. Incoordination typically is worse in the lower extremities than in the upper extremities. Cerebellar involvement tends to include the flocculus, which is a component of the vestibulo-ocular reflex. Disruption of the flocculus by tumor compression can lead to dysequilibrium, truncal ataxia, and nystagmus. Volitional limb movement remains normal when only the flocculus is involved. However, if the lesion grows large enough to affect the cerebellar hemispheres, severe lateral compression can result in ipsilateral ataxic limb movements, intention tremor, and, in extreme situations, hypotonia.
Trigeminal Nerve Involvement
Typical trigeminal nerve dysfunction related to acoustic neuromas includes hypesthesia or paresthesias of the midfacial region. Although the incidence of trigeminal symptoms ranges from 33% to 71%,[29] several studies have reported a strong correlation between tumor size and trigeminal symptoms.[22,29,32]
The classic description of early trigeminal involvement with an acoustic neuroma is loss of the corneal reflex. Increasing nerve dysfunction related to tumor growth can produce midfacial tingling that can progress to numbness. In extremely large tumors, the muscles of mastication can atrophy. According to Jannetta,[18] 10% of trigeminal neuralgia cases are related to neoplasms, a proportion of which includes acoustic neuromas. In giant tumors, contralateral tic douloureux can serve as a false localizing sign. Symptoms arise due to direct nerve compression in the superior CPA just inferior to the tentorium.
Headaches
Cephalgia is related to compression and local irritation of the neural, vascular, and dural contents of the internal auditory canal or petrous bony dura. Incidence is also related to tumor size. In the UCSF study, no tumors smaller than 1 cm caused headaches. In contrast, headaches occurred in 20% of those with 1- to 3-cm tumors and in 43% of those with tumors larger than 3 cm.[29]
Hydrocephalus
Symptoms from hydrocephalus related to tumor obstruction of the fourth ventricle include headache, nausea, vomiting, diplopia, papilledema, and changes in mental status. Its incidence is low, occurring in 4% of cases according to the UCSF series.[29] Significant tumor growth is usually required to produce obstructive hydrocephalus.
Facial Nerve Involvement
Facial nerve dysfunction occurs late in the course of tumor growth, and frank facial nerve palsy is usually preceded by facial twitch and paresis. The facial nerve can tolerate gradual tumor growth surprisingly well, despite being splayed and thinned over the surface of the tumor. This effect is especially likely to occur just medial to the porus acusticus, the anterior bony lip of which can compress the distorted nerve as it enters the internal auditory canal. Neural compression causing facial nerve dysfunction typically requires a large tumor mass. Therefore, small CPA tumors associated with facial nerve dysfunction should signal clinicians to suspect another pathology such as a facial nerve schwannoma or cavernous malformation involving the internal auditory canal. The incidence of facial nerve dysfunction associated with acoustic neuromas is about 10%.
The sensory fibers of the facial nerve are often affected, but patients seldom notice a clinical effect. Hitselberger’s sign, hypesthesia of the posterior external auditory canal and concha skin, can be detected with careful examination.[2]
Lower Cranial Nerve and Brainstem Findings
Glossopharyngeal and vagus nerve compression from tumor can cause hoarseness, dysphagia, and aspiration. Hypoglossal nerve compression can lead to dysarthria. These findings are always late in the course of an acoustic neuroma because the tumor must be very large to extend caudally toward the jugular foramen and hypoglossal canal. Contralateral weakness is typically a long tract sign related to brain stem compression or cerebellar tonsillar herniation through the foramen magnum. However, ipsilateral weakness can also occur.
Diagnosis
Audiologic testing is useful not only for the diagnosis of acoustic neuromas but also for pretreatment planning based on the extent of useful hearing. Both pure tone audiometry and speech audiometry are the first-line tests. Puretone sensorineural hearing loss is reflected in the shape of the audiogram: flat, high-tone loss, low-tone loss, and trough-shaped loss. Johnson found that 66% of patients experienced high-tone loss, while the other types each occurred in 10%.[19] Analysis of speech discrimination scores showed that only 28% of patients exhibited speech discrimination better than 62%. The ability of audiometric testing to identify all patients with an acoustic neuroma is imperfect.[29] Therefore, test results should be interpreted cautiously. Persistent clinical suspicion of an acoustic neuroma despite normal audiologic tests warrants the patient undergoing gadolinium-enhanced MR imaging.
Acoustic reflex testing, which monitors middle ear impedance as an indirect measure of the contraction of middle ear muscles in response to a loud sound, is independent of the patient, which avoids biased test results. Acoustic reflex decay is defined as 50% decay of a tone administered 10 dB above threshold.[29] The sensitivity of the combined use of acoustic reflex and decay for detecting acoustic neuromas is as high as 97%. Low specificity, however, precludes these tests from having an accurate predictive value for acoustic neuromas.
The most useful and accurate audiologic test for acoustic neuromas remains the ABR. The prerequisite for this test is hearing function sufficient to generate an adequate ABR. The test uses computer averaging over time to filter background noise to generate an electroencephalogram created by the response of the auditory pathway to an auditory stimulus. The waveform represents specific anatomical points along the auditory neural pathway: the cochlear nerve and nuclei (waves I and II), superior olivary nucleus (wave III), lateral lemniscus (wave IV), and inferior colliculi (wave V).
For clinical use, the most reliable waves generated are the synchronous discharges of wave I from the cochlear nerve and of wave V from the inferior colliculus. Waveform characteristics used to detect abnormalities include amplitude, latency, and the interval difference between the two sides. The most reliable indicator for acoustic neuromas from the ABR is the interaural latency differences in wave V: The latency in the abnormal ear is prolonged (Fig. 1).
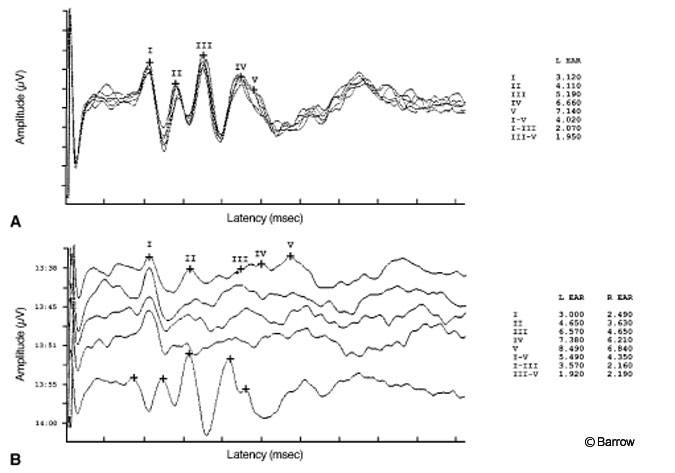
The advantages of ABR include its relative imperviousness to general anesthesia and the patient’s level of consciousness. However, the ABR varies with temperature, head circumference, and menstrual cycle.[8] Several reports have documented the specificity of ABR for the diagnosis of acoustic neuroma to be as high as 98% with a specificity of 90%.[11,16,22,23,25] The rate of falsenegative ABRs has been reported as 2% for acoustic neuromas and as high as 25% for other CPA tumors.[31]
Stacked derived-band ABR may identify small, intracanalicular acoustic neuromas with greater sensitivity than a standard ABR.[9] This technique uses derived-band ABRs obtained up to normal hearing levels. The stacked ABR uses the amplitude of wave V as the measurement point: Wave V of each derived-band ABR is temporarily aligned and the time-shifted responses are summed.
Rarely, a patient with an acoustic neuroma has normal pure-tone hearing, acoustic reflexes, and ABRs. Thus, any patient under a high index of clinical suspicion for an acoustic neuroma should undergo MR imaging.
Imaging Studies
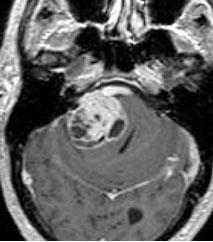
Gadolinium-enhanced MR imaging offers excellent resolution of soft tissues, the tumor’s size and morphology, and the relationship between normal and abnormal anatomy. It is the imaging study of choice to rule out an acoustic neuroma. Tumor enhancement is usually homogenous. Areas of cystic structure, necrosis, and hemorrhage can be present in larger masses.
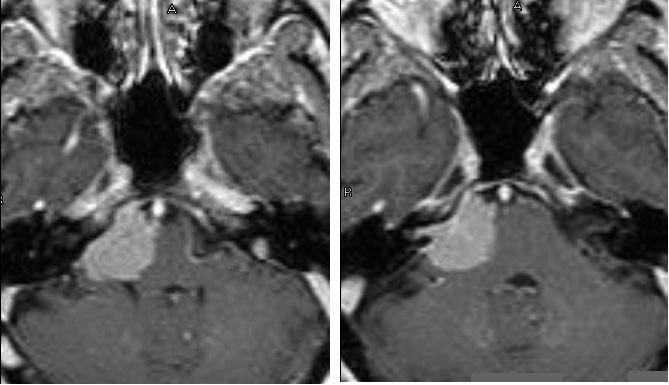
Acoustic neuromas are usually lobular, well-circumscribed, and centered over the porus acusticus. They may or may not demonstrate tumor enhancement into the internal auditory canal. Larger tumors maintain their lobular shape, leading to deformation of the adjacent cranial nerves, brainstem, and cerebellum (Fig. 2).
Acoustic neuromas are differentiated from CPA meningiomas, the second most common mass in this region in adults, by the absence of a dural tail. The latter typically represents enhancement where a meningioma attaches to its dural base. Dural-based meningiomas also lack a flattened, broad base along the petrous face (Fig. 3).[3] Both tumors can extend into the internal auditory canal.
The differential diagnoses for CPA masses also include epidermoids, choroid plexus papillomas, metastases, arachnoid cysts, schwannomas involving other cranial nerves, neurenteric cysts, cavernous malformations, cholesterol granulomas, aneurysms, and dolichoectatic vertebrobasilar arteries.
Computed tomography (CT) is a common screening tool to rule out mass lesions of the brain. Acoustic neuromas appear iso- to hypodense on nonenhanced CT and enhance homogenously after contrast injection. However, CT is inferior to MR imaging in its resolution of soft tissue anatomy and is subject to beam-hardening artifact in the bony confines of the posterior fossa. The latter point has become less troublesome with the advent of multislice spiral CT scanners.
Nonetheless, CT retains its usefulness in pretreatment planning because it offers superior imaging of bony anatomy. Bone windows on CT can show enlargement of the internal auditory canal related to tumor growth and can exquisitely detail the relationship of the tumor to the mastoid air cells, transverse sigmoid junction, jugular bulb, internal auditory canal, and bony labyrinth.[14] Understanding these relationships facilitates surgical planning.
Conclusions
Acoustic neuromas are the most common CPA tumors in adults. Although their natural history is still not clearly defined, a subset can demonstrate significant growth over time. The stages of acoustic neuroma growth are well correlated with symptom development. Depending on their size, acoustic neuromas typically present with unilateral sensorineural hearing loss and various degrees of cranial nerve symptoms and cerebellar symptoms. Audiometric testing can provide useful diagnostic and pretreatment planning information. Even if audiometric tests are negative, imaging studies should be obtained if clinical suspicion remains. Because of its excellent resolution of soft tissue, MRI is the definitive diagnostic tool. For preoperative planning, CT provides superior bony detail in relation to the tumor.
References
- The National Institutes of Health Consensus Development Conference on Acoustic Neuroma. Consens Statement 9(4):1-24, 1991
- Committee on Hearing and Equilibrium guidelines for the evaluation of hearing preservation in acoustic neuroma (vestibular schwannoma). Otolaryngol Head Neck Surg 113:179-180, 1995
- Aoki S, Sasaki Y, Machida T, et al: Contrast-enhanced MR images in patients with meningioma: Importance of enhancement of the dura adjacent to the tumor. AJNR Am J Neuroradiol 11:935-938, 1990
- Beck HJ, Beatty CW, Harner SG, et al: Acoustic neuromas with normal pure tone hearing levels. Otolaryngol Head Neck Surg 94:96-103, 1986
- Chaimoff M, Nageris BI, Sulkes J, et al: Sudden hearing loss as a presenting symptom of acoustic neuroma. Am J Otolaryngol 20:157-160, 1999
- Cushing H: Tumors of the Nervus Acousticus and the Syndrome of the Cerebellopontile Angle. Philadelphia: WB Saunders, 1917
- Deen HG, Ebersold MJ, Harner SG, et al: Conservative management of acoustic neuroma: An outcome study. Neurosurgery 39:260-264, 1996
- Dehan CP, Jerger J: Analysis of gender differences in the auditory brainstem response. Laryngoscope 100:18-24, 1990
- Don M, Masuda A, Nelson R, et al: Successful detection of small acoustic tumors using the stacked derived-band auditory brain stem response amplitude. Am J Otol 18:608-621, 1997
- Erickson LS, Sorenson GD, McGavran MH: A review of 140 acoustic neurinomas (neurilemmoma). Laryngoscope 75:601-627, 1965
- Flood LM, Brammer RE, Graham MD, et al: Pitfalls in the diagnosis of acoustic neuroma. Clin Otolaryngol 9:165-170, 1984
- Gardner G, Robertson JH: Hearing preservation in unilateral acoustic neuroma surgery. Ann Otol Rhinol Laryngol 97:55-66, 1988
- Glasscock ME, III, Pappas DG, Jr., Manolidis S, et al: Management of acoustic neuroma in the elderly population. Am J Otol 18:236-241, 1997
- Graham MD, Sataloff RT: Acoustic tumors in the young adult. Arch Otolaryngol 110:405-407, 1984
- Hardy DG, Macfarlane R, Baguley D, et al: Surgery for acoustic neurinoma. An analysis of 100 translabyrinthine operations. J Neurosurg 71:799-804, 1989
- Harner SG, Laws ER, Jr.: Diagnosis of acoustic neurinoma. Neurosurgery 9:373-379, 1981
- Jackler RK, Pitts LH: Acoustic neuroma. Neurosurg Clin N Am 1:199-223, 1990
- Jannetta PJ: Neurovascular compression in cranial nerve and systemic disease. Ann Surg 192:518-525, 1980
- Johnson EW: Auditory test results in 500 cases of acoustic neuroma. Arch Otolaryngol 103:152-158, 1977
- Leonard JR, Talbot ML: Asymptomatic acoustic neurilemoma. Arch Otolaryngol 91:117-124, 1970
- Mirz F, Jorgensen B, Fiirgaard B, et al: Investigations into the natural history of vestibular schwannomas. Clin Otolaryngol 24:13-18, 1999
- Moffat DA, Hardy DG, Baguley DM: Strategy and benefits of acoustic neuroma searching. J Laryngol Otol 103:51-59, 1989
- Pensak ML, Glasscock ME, III, Josey AF, et al:Sudden hearing loss and cerebellopontine angle tumors. Laryngoscope 95:1188-1193, 1985
- Pool JL, Pava AA: The Early Diagnosis and Treatment of Acoustic Nerve Tumors. Springfield, IL:Charles C Thomas, 1957
- Roland PS, Glasscock ME, III, Bojrab DI, et al:Normal hearing in patients with acoustic neuroma. South Med J 80:166-169, 1987
- Rosenberg SI: Natural history of acoustic neuromas. Laryngoscope 110:497-508, 2000
- Saunders AZ, Jackson RT: CPA tumors with normal routine audiometry and positive reflex and BSER tests. Am J Otol 2:318-323, 1981
- Schuknecht HF: Pathology of vestibular schwannoma (acoustic neuroma), in Silverstein H, Norrel H (eds): Neurological Surgery of the Ear. Birmingham: Aesculapius Publishing, 1977, pp 193-197
- Selesnick SH, Jackler RK: Clinical manifestations and audiologic diagnosis of acoustic neuromas. Otolaryngol Clin North Am 25:521-551, 1992
- Selesnick SH, Jackler RK, Pitts LW: The changing clinical presentation of acoustic tumors in the MRI era. Laryngoscope 103:431-436, 1993
- Telian SA, Kileny PR, Niparko JK, et al: Normal auditory brainstem response in patients with acoustic neuroma. Laryngoscope 99:10-14,1989
- Thomsen J, Tos M: Acoustic neuroma: Clinical aspects, audiovestibular assessment, diagnostic delay, and growth rate. Am J Otol 11:12-19, 1990
- Yamakami I, Uchino Y, Kobayashi E, et al: Conservative management, gamma-knife radiosurgery, and microsurgery for acoustic neurinomas: A systematic review of the outcome and risk of three therapeutic options. Neurol Res 25:682-690, 2003