Imaging of Acoustic Neuromas
Brian D. O’Rourke, MD
Robert C. Wallace, MD
Division of Neuroradiology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
Acoustic neuromas are common benign lesions that can result in sensorineural hearing loss. These tumors have specific imaging characteristics that can help differentiate them from other lesions that occur in the cerebellopontine angle cistern of the posterior fossa. Magnetic resonance imaging is the most sensitive method of detection and also plays an important role in the follow-up of patients treated with both surgery and stereotactic radiosurgery. Although contrast-enhanced T1-weighted imaging remains the most commonly employed method for evaluation, high-resolution T2-weighted imaging provides a sensitive alternative for tumor detection.
Key Words: acoustic neuroma, cerebellopontine angle, vestibulocochlear nerve, internal auditory canal
Acoustic neuromas, common benign tumors accounting for 10% of primary intracranial tumors, are the most common tumor in the cerebellopontine angle cistern.[17] They almost always arise from Schwann cells of the vestibular portion of the vestibulocochlear nerve; therefore, vestibular schwannoma is the preferred term.
Patients usually present with unilateral hearing loss, often accompanied by tinnitus and vertigo. Sensorineural hearing loss is the result of extrinsic pressure from compression of the cochlear division of the eighth cranial nerve within the internal auditory canal by the tumor arising from the adjacent vestibular division. The accompanying facial nerve within the internal auditory canal is more tolerant of this local mass effect, and facial nerve symptoms such as ipsilateral facial paralysis are uncommon. Because this tumor grows slowly, symptoms evolve over months and years and the lesion can become quite large before it is detected. As the size of the tumor increases, it may extend into the posterior fossa. Symptoms related to increased intracranial pressure and cerebellar involvement such as ataxia may occur.
Treatment of vestibular schwannomas is aimed at removing the lesion or arresting its growth to prevent further neurologic impairment. Imaging plays a crucial role in both the detection and posttherapeutic management of these lesions.
Anatomy and Pathology
The anatomy of the facial and vestibulocochlear nerves must be understood to reliably evaluate and precisely localize pathology within the internal auditory canal and cerebellopontine angle. These nerves emerge from the brainstem at the pontomedullary junction and course through the cerebellopontine angle cistern. They enter the meatus, or porus acusticus, of the internal auditory canal as two separate structures. The shape of the facial nerve is tubular. Cross-sectional imaging viewed perpendicular to the long axis of the canal consistently shows the nerve in the anterosuperior quadrant of the internal auditory canal. The vestibular and cochlear components of the eighth cranial nerve usually enter the meatus as a single unit, and they divide into separate nerves within the internal auditory canal. The point of separation may vary among patients.[13] The vestibulocochlear nerve assumes a crescent shape along the posterior and inferior aspects of the middle portion of the canal as the two components begin to divide.
Only in the lateralmost 3 to 4 mm of the internal auditory canal do the vestibular and cochlear nerves divide into separate identifiable structures.[2,12,13] The cochlear division continues in the anterior-inferior quadrant of the canal beneath the facial nerve, and the vestibular nerves are located posterior to these two structures within the posterior half of the canal. At the fundus, the most lateral aspect of the canal, the vestibular nerve is separated into the superior and inferior divisions by the crista falciformis, a horizontal bony crest within the canal. The superior half of the canal is further divided into an anterior and a posterior compartment by a thin ossific septa referred to as Bill’s bar. The facial nerve is located anterior to Bill’s bar, and the superior division of the vestibular nerve courses in the posterior compartment. The four nerves classically described within the internal auditory canal are usually only delineated as individual nerves in the lateralmost portion of the canal.[13]
The cranial nerves are unique because their nerve sheaths are formed by oligodendrocytes (glial cells) proximally as they exit the brain parenchyma. Within several millimeters the transition to myelin-forming Schwann cells occurs. For the vestibulocochlear nerve, this glial-Schwann cell junction occurs near the porus acusticus. Most schwannomas originate at this location, but they may occur anywhere along the course of the nerve myelinated by Schwann cells.[14] Sensory nerves are more heavily myelinated than motor nerves. Therefore, these tumors tend to occur in association with sensory nerves such as the vestibular nerve. Trigeminal schwannomas are the second most common intracranial schwannoma accounting for about 5%, followed by facial schwannomas at a distant third.[9] Histologically, schwannomas are composed of Antoni type A and type B tissues. Antoni type A tissue is composed of compact palisades of spindle cells while Antoni type B tissue is a loose myxoid stroma with few cells and regions of cyst formation. Most vestibular schwannomas consist of predominantly type A tissue, until their size increases and their cellularity decreases.[5]
Neurofibromatosis type 2 (NF-2) is defined by bilateral acoustic neuromas. It is a neurocutaneous syndrome, or phacomatosis, that can be inherited in an autosomally dominant fashion or that can occur sporadically. In addition to the bilateral tumors of the internal auditory canal, NF-2 can be associated with multiple meningiomas, glial cell tumors, and other nerve sheath tumors.
Imaging
Tumor Characteristics
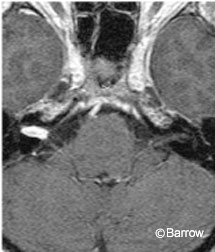
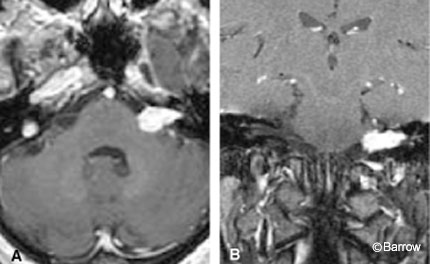
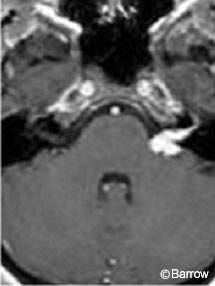
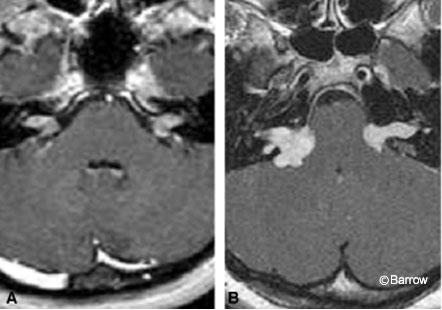
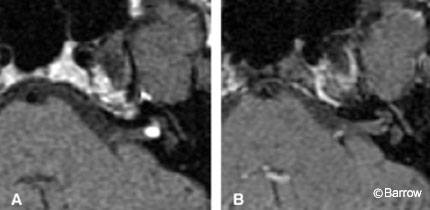
Acoustic neuromas are well-circumscribed, enhancing lesions that typically arise near the porus acusticus. They are small when their location is intracanalicular. Intracanalicular schwannomas are ovoid or cylindrical-shaped and have a convex medial margin (Fig. 1). As the tumor grows, it often extends into the cerebellopontine angle cistern where it can expand unimpeded by the osseous confines of the internal auditory canal. This growth pattern creates an “ice cream on a cone” appearance with the cone representing the intracanalicular component (Fig. 2).
[one_half]
[/one_half]
[one_half_last]
[/one_half_last]
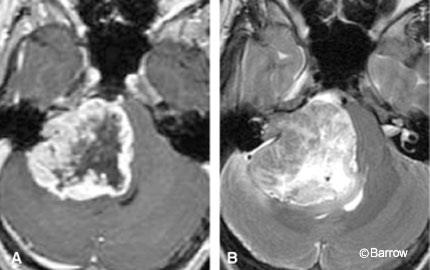
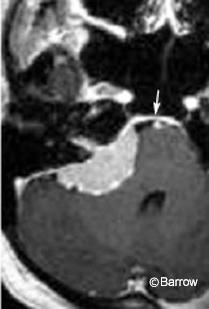
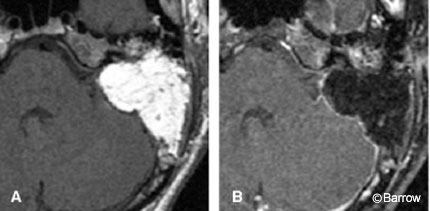
The round cisternal component typically remains centered at the auditory meatus and forms acute angles with the petrous bone. Because the tumor grows slowly, the bony canal may be remodeled and it may expand, particularly near the porus acusticus. This canalicular expansion is a late and unreliable radiologic sign because the width of the canal and degree of meatal flaring can vary considerably among normal patients. As the tumor enlarges, resulting areas of internal necrosis and hemorrhage can cause the formation of cysts, which appear as central regions of nonenhancement (Fig. 3).
T2-Weighted Imaging
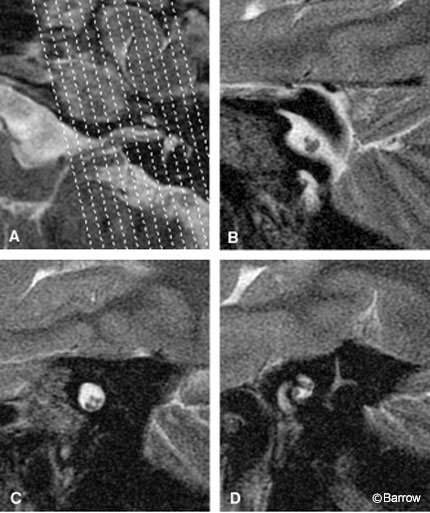
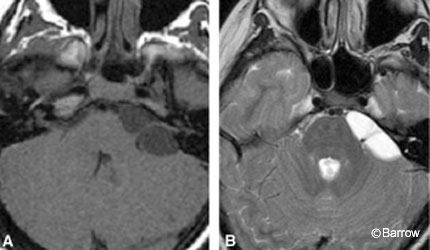
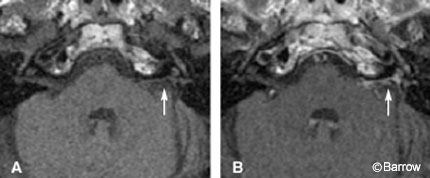
Magnetic resonance (MR) imaging is sensitive enough to define intracanalicular nerves for clinical purposes. Although contrast-enhanced T1-weighted imaging remains the most commonly employed method for evaluation, high-resolution T2-weighted imaging allows delineation of the individual nerves and evaluation of their relative sizes. When combined with knowledge of normal anatomy, this information can also be used to detect tumor. Sagittal oblique imaging in a plane perpendicular to the long axis of the internal auditory canal provides a cross-sectional view of the canal and allows individual nerves to be depicted (Fig. 4). The high T2-weighted signal of the surrounding cerebrospinal fluid (CSF) provides excellent contrast between the relatively T2- weighted hypointensity of the nerves. T2-weighted fast spin-echo (FSE) MR imaging acquired on a 1.5 Tesla-strength magnet with thin-section thickness, a small field of view, and a large matrix (512 x 512) provides excellent anatomic detail. Direct two-dimensional (2D) acquisition in the axial, coronal, and sagittal oblique planes through the internal auditory canal is typical. Three-dimensional volume acquisition can be obtained as a single axial acquisition, and that data set can then be used for multiplanar reconstructed images. The high-resolution anatomic detail of these T2-weighted images not only detects nerve sheath tumors but also allows tumor volume and the degree of nerve involvement to be evaluated. The ability of high-resolution T2-weighted imaging to depict tumor involvement of individual nerves may provide prognostic information that can help preserve hearing in selected patients.[15]
Contrast-Enhanced T1-Weighted Imaging
Differential Considerations
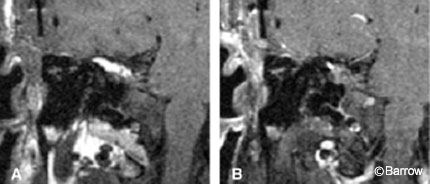
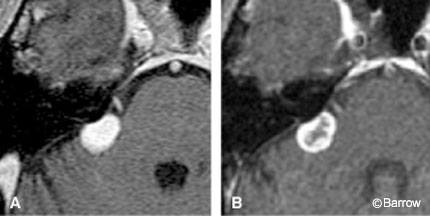
An enhancing lesion within the internal auditory canal typically represents a vestibular schwannoma. Other lesions, however, can occur in this location and should be excluded. Although rare, a schwannoma arising from the facial nerve can appear identical to a vestibular schwannoma on contrast-enhanced T1-weighted imaging. The distinction between the two lesions can affect treatment and is reliably made by imaging if enhancement extends into the labyrinthine segment of the facial nerve canal within the temporal bone (Fig. 5). Meningeal processes such as meningiomas, inflammation (meningitis, sarcoid), or metastatic disease can also demonstrate intracanalicular enhancement and mimic vestibular schwannomas as the leptomeninges and dura extend into the internal auditory canal (Fig. 6).
[one_half padding=”0 0 20px 0″]
[/one_half]
[one_half_last]
[/one_half_last]
In terms of cisternal vestibular schwannomas, meningiomas within the cerebellopontine angle cistern are probably the most common differential consideration. Both can appear as large, round, densely enhancing, extraaxial masses causing significant local mass effect. Meningiomas typically arise adjacent to the porus acusticus but sometimes extend into the internal auditory meatus itself. The presence of a ‘dural tail’ or an obtuse angle with respect to the petrous bone can also help differentiate a meningioma from a schwannoma (Fig. 7). Other common cerebellopontine angle masses such as epidermoids and arachnoid cysts are easily distinguished by their hyperintense T2-weighted signal characteristics and lack of enhancement (Fig. 8). On postcontrast T1-weighted imaging, an intracanalicular lipoma can appear as an ovoid hyperintense mass that can be differentiated from a schwannoma on precontrast T1-weighted images (Fig. 9).
[one_half]
[/one_half]
[one_half_last]
[/one_half_last]
Posttherapeutic Imaging
The treatment options of vestibular schwannoma include observation, surgical resection, and stereotactic radiosurgery. Clinical observation may be reserved for older patients using periodic MR imaging to monitor growth of the tumor. Imaging also plays an important role in the follow-up of patients undergoing surgery and stereotactic radiosurgery.
Surgery
Postcontrast T1-weighted MR imaging is crucial during postoperative management. The pattern of enhancement within the internal auditory canal can help differentiate expected normal postoperative appearance from residual tumor. The rate of inadvertent subtotal resection is only 0.5 to 1.0%.[18] Sometimes, however, small foci of tumor are intentionally left to preserve facial nerve function.[4] These small foci of tumor usually enhance in a nodular pattern. Some form of postoperative enhancement within the internal auditory canal is common, if not uniformly present.[19] Linear enhancement is almost always a normal finding associated with an uncomplicated acoustic neuroma resection. Nodular or mass-like enhancement within the internal auditory canal raises the possibility of residual tumor and should be followed. In the absence of radiation therapy, any enhancement pattern, including nodular and masslike, that remains stable or decreases in size or degree of enhancement on follow-up examinations is unlikely to represent residual tumor.
The type of surgical approach must be considered when evaluating images of a patient who has undergone resection of an acoustic neuroma. Three surgical approaches are commonly used to access the cerebellopontine angle and internal auditory canal: the retrosigmoid, translabyrinthine, and middle fossa craniotomy.[3] The retrosigmoid suboccipital approach can preserve hearing in appropriately selected patients because the labyrinth (cochlea, vestibule and semicircular canals) is left intact. The posterior wall of the internal auditory canal is drilled to expose the intracanalicular contents. However, the lateralmost aspect of the posterior wall is left intact to avoid damage to the posterior semicircular canal (Fig. 10).
The fundus of the canal cannot be visualized during surgery and is therefore associated with the highest risk of unintentional tumor residual. After this type of approach, nodular or mass-like enhancement in the fundus merits close follow-up.[19]
The middle fossa temporal approach also preserves hearing while allowing complete exposure of the fundus. This approach is usually reserved for patients with small tumors and near-normal hearing. Intense linear enhancement along the roof of the internal auditory canal is an expected postoperative finding because fascial grafts are typically used to close the bony defect (Fig. 11).
A translabyrinthine approach is reserved for large tumors when hearing preservation is no longer a concern because residual hearing has been destroyed. The mastoid, vestibule, and posterior wall of the internal auditory canal are resected to obtain complete exposure of the internal auditory canal, including the fundus. The cochlea is preserved. Surgical fat grafts are used to fill the mastoidectomy cavity to prevent CSF leaks. The T1-weighted hyperintense fat graft necessitates the use of fat-saturation pulse sequences to evaluate the surgical bed and remaining internal auditory canal for underlying enhancement (Fig. 12).
Stereotactic Radiosurgery
The rate of tumor control with radiation therapy ranges between 81 to 96%.[8,11] Only a small percentage of tumors continues to enlarge. MR imaging can show different patterns of tumor behavior, which can help predict the long-term effects of treatment. As many as 40% of treated tumors undergo a period of temporary enlargement after Gamma knife radiosurgery.[11] In these cases, the size of the tumor initially enlarges and peaks at one year. Sustained regression follows in the second year. To determine if acoustic neuromas have stabilized or regressed, the lesions must be imaged during the first 2 to 3 years after radiation therapy.
Summary
Acoustic neuromas, or vestibular schwannomas, are commonly encountered benign lesions that can result in profound hearing loss. Early detection and treatment can prevent progression of hearing loss and other neurologic deficits. MR imaging remains the most sensitive method of detection.
References
- Allen RW, Harnsberger HR, Shelton C, et al: Lowcost high-resolution fast spin-echo MR of acoustic schwannoma: An alternative to enhanced conventional spin-echo MR? AJNR Am J Neuroradiol 17:1205-1210, 1996
- Bergstrom B: Morphology of the vestibular nerve. II. The number of myelinated vestibular nerve fibers in man at various ages. Acta Otolaryngol 76:173-179, 1973
- Brackmann DE, Arriaga MA: Surgery of the posterior cranial fossa, in Cummings CW, Fredrickson JM (eds): Otolaryngology: Head and Neck Surgery. St. Louis, MO: CV Mosby, 1993, pp 3338-3352
- Cass SP, Kartush JM, Wilner HI, et al: Comparison of computerized tomography and magnetic resonance imaging for the postoperative assessment of residual acoustic tumor. Otolaryngol Head Neck Surg 104:182-190, 1991
- Gruskin P, Carberry JN: Pathology of acoustic tumors, in House WF, Luetje CM (eds): Acoustic Tumors. Baltimore: University Park Press, 1979, pp 85-148
- Jackler RK: Cost-effective screening for acoustic neuroma with unenhanced MR: A clinician’s perspective. AJNR Am J Neuroradiol 17:1226-1228, 1996
- Linskey ME, Lunsford LD, Flickinger JC: Radiosurgery for acoustic neurinomas: Early experience. Neurosurgery 26:736-744, 1990
- Linskey ME, Lunsford LD, Flickinger JC: Neuroimaging of acoustic nerve sheath tumors after stereotaxic radiosurgery. AJNR Am J Neuroradiol 12:1165-1175, 1991
- McCormick PC, Bello JA, Post KD: Trigeminal schwannoma. Surgical series of 14 cases with review of the literature. J Neurosurg 69:850-860, 1988
- Murphy MR, Selesnick SH: Cost-effective diagnosis of acoustic neuromas: A philosophical, macroeconomic, and technological decision. Otolaryngol Head Neck Surg 127:253-259, 2002
- Nakamura H, Jokura H, Takahashi K, et al: Serial follow-up MR imaging after gamma knife radiosurgery for vestibular schwannoma. AJNR Am J Neuroradiol 21:1540-1546, 2000
- Rasmussen AT: Studies of the VIIIth cranial nerve of man. Laryngoscope 50:67-83, 1940
- Rubinstein D, Sandberg EJ, Cajade-Law AG: Anatomy of the facial and vestibulocochlear nerves in the internal auditory canal. AJNR Am J Neuroradiol 17:1099-1105, 1996
- Russell DS, Rubinstein LJ: Pathology of Tumors of the Nervous System. Baltimore: Williams and Wilkins, 1989
- Schmalbrock P, Chakeres DW, Monroe JW, et al: Assessment of internal auditory canal tumors: A comparison of contrast-enhanced T1-weighted and steady-state T2-weighted gradient-echo MR imaging. AJNR Am J Neuroradiol 20:1207-1213, 1999
- Stone JA, Chakeres DW, Schmalbrock P: Highresolution MR imaging of the auditory pathway. Magn Reson Imaging Clin N Am 6:195-217, 1998
- Swatz JD, Harnsberger HR: Imaging of the Temporal Bone, 3rd ed. New York: Thieme, 1998
- Thedinger BA, Glasscock ME, III, Cueva RA, et al: Postoperative radiographic evaluation after acoustic neuroma and glomus jugulare tumor removal. Laryngoscope 102:261-266, 1992
- Weissman JL, Hirsch BE, Fukui MB, et al: The evolving MR appearance of structures in the internal auditory canal after removal of an acoustic neuroma. AJNR Am J Neuroradiol 18:313-323, 1997