
Stereotactic Radiosurgery in the Management of Acoustic Neuromas
Randall W. Porter, MD
C. Phillip Daspit, MD*
John J. Kresl, MD, PhD**
Christopher A. Biggs, MD, PhD**
David G. Brachman, MD**
Mark J. Syms, MD*
Division of Neurological Surgery and *Section of Neurotology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona **Department of Radiation Oncology, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona and Arizona Oncology Services, Phoenix, Arizona
Abstract
Stereotactic radiosurgery has become an important part of the armamentarium for the treatment of acoustic neuromas. Originally, it was used to treat elderly or medically infirm patients. Recently, however, it has become the primary treatment for patients who wish to avoid the potential complications associated with open neurosurgical procedures. Although radiographic elimination of the tumor is improbable, high rates of tumor control can be achieved with minimal complications. The risks of cerebrospinal fluid leakage, intracranial hemorrhage, myocardial infarction, and anesthesia are almost eliminated. The risk of facial palsy ranges from 1 to 2%. The rate of hearing preservation is 50%. Fractionated radiosurgery may increase the probability of preserving functional hearing.
Key Words: acoustic neuroma, cyberknife, Gamma knife, fractionated radiosurgery, radiosurgery
The earliest description of an acoustic neuroma was probably by Sandifort in 1777.[6,22] He described an autopsy of a patient with a tumor of the right auditory nerve;the tumor was firm and attached to the brain stem where the seventh and eighth cranial nerves exit. In 1830 Bell first diagnosed such a tumor in a living patient.[3] In 1894 Ballance first removed an acoustic neuroma successfully and reported the procedure in 1907.[1]
Before the early 1990s surgical resection was the mainstay for the treatment of acoustic neuromas. Operations performed in the 20th century chronicle the challenges and complications associated with surgical intervention. Early in the past century, acoustic neuromas were seldom diagnosed until they had attained a significant size and were associated with multiple cranial neuropathies, brain stem compression, and elevated intracranial pressure. Subtotal removal was the norm, and mortality rates were as high as 84%.[7] Preservation of the facial nerve was impossible until the mid20th century, and only minor improvements followed in the ensuing decades. Finally, however, the development of the operating microscope, refinement of surgical techniques, the introduction of sophisticated intraoperative monitoring of cranial nerves, and improved anesthetic techniques significantly improved outcomes.
The problems associated with microsurgical resection are well known. Dysfunction of the facial nerve and auditory nerves becomes apparent in the immediate postoperative period. Patients can suffer from depression, sleep disturbances, and fatigue for weeks to months.
A few patients do not return to gainful employment. However, improved instrumentation and surgical techniques and the combined expertise of neurosurgical and neurotological microsurgeons lead to the conclusion that total removal in a younger patient is more desirable than radiation therapy.The lack of long-term control data and the rare chance of malignant transformation make it problematic to recommend radiation in this age group. However, a young patient may still choose stereotactic radiosurgery after thoroughly discussing the goals,complications,and risks associated with both surgery and radiosurgery with the physician.
Informed consent is obtained before all surgeries. However, the disfiguring nature of a facial nerve palsy and its psychological impact on patients cannot and should not be underestimated. Patients often overestimate their ability to accept facial nerve palsy. After the initial recovery period, they can be quite dissatisfied with their outcome and would choose a different course of treatment given the opportunity to make the decision again. Other patients maintain that complete tumor resection is the only option,regardless of complications. However, this stance is the exception rather than the rule.
Stereotactic radiosurgery is no longer an option limited to the elderly or medically infirm. Young and middle-aged patients now consider radiosurgical alternatives to avoid the potential complications associated with surgical resection and to expedite recovery. The ability to reproduce similar outcomes at different institutions is another issue involved with the treatment of acoustic neuromas. Stereotactic radiosurgical techniques can easily be learned by competent stereotactic radiosurgeons and reliably reproduced at multiple institutions (Figs.1 and 2).
The same cannot be said for intracranial microsurgery.Years of training and independent experience are necessary to achieve the excellent surgical outcomes reported by master neurosurgeons and neurotologists. Consequently,stereotactic radiosurgery remains a viable option for those seeking durable tumor control while minimizing the risk of surgical complications. This article reviews the management of acoustic neuromas with stereotactic radiosurgery.
Gamma Knife Radiosurgery
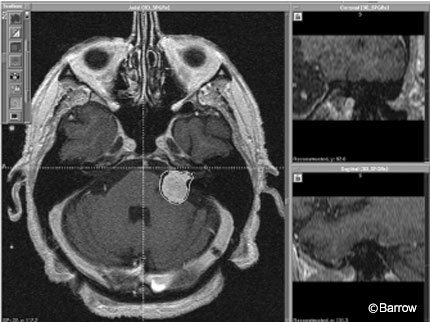
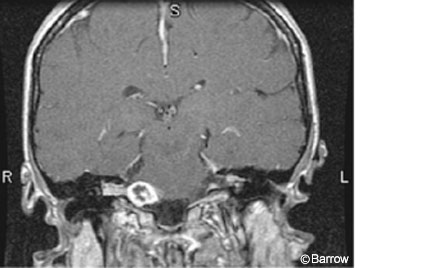
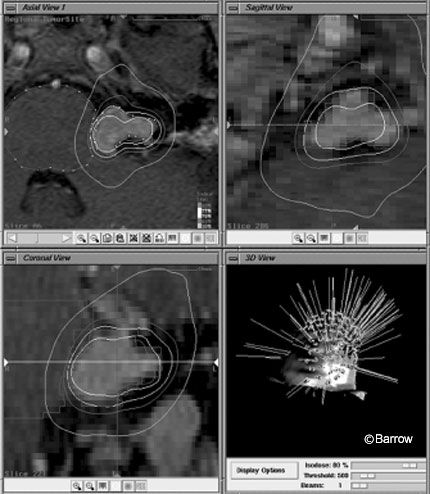
Initially, radiosurgery with single fraction doses of 16 Gy was associated with significant complication rates.[8] The rate of facial paralysis was 21%, higher than that associated with surgery. The rate of facial numbness was 27% and that of functional hearing loss was 49%.[12] Despite the low mortality rate associated with radiosurgery, the rate of cranial neuropathy was unacceptably high. Consequently, dosing was re-evaluated and modified. Improvements in stereotactic MRI techniques and delivery of treatment platforms and implementation of the 13 Gy marginal dose significantly reduced the complication rates associated with radiosurgery while the rate of tumor control remained the same (Figs. 3 and 4).
Flickinger et al.[8] reported tumor control rates as high as 97% with Gamma knife stereotactic radiosurgery. From 1992 to 1997, they treated 190 patients with a median follow up of 30 months. Tumor shrinkage occurred in 35% of the patients,and the 5-year actuarial rate of tumor control was 97%. Three patients later required surgical resection. Cranial neuropathies were infrequent; in particular,trigeminal neuropathy occurred in only 2.6% of the patients.The rate of facial weakness in patients receiving a marginal dose of 13 Gy or lower was 0% compared with 2.5% of those receiving a marginal dose of 14 Gy or higher. Serviceable hearing was preserved in 55 of the 75 (73%) patients.
Prasad et al.reported 96 cases treated with Gamma knife using 13 Gy to the margin. Tumor volumes ranged from 0.02 to 18.3 cm3.[19] Tumor volume decreased in 81% and increased in 6%. In the 57 patients who had undergone a previous surgery, tumor volume decreased in 65% and increased in 11%. Five patients suffered trigeminal neuropathy, which persisted in two. Three patients suffered facial paresis, and hearing deteriorated in 60% of the patients. However, no patient’s hearing deteriorated within the first 2 years of treatment.
Petit et al. treated 47 patients (median dose,12 Gy).[17] The mean maximum diameter of their tumors was 18 mm. With a median follow up of 3.6 years, tumor control was achieved in 96% of the patients. Transient facial weakness occurred in 4% and resolved within 6 months. No patients suffered trigeminal neuropathy,and no patients lost serviceable hearing (Gardner-Robertson Class I or II). Hearing deteriorated in 12% who were classified as having nonserviceable hearing before treatment.
Regis et al. evaluated the functional outcomes and quality of life of 224 consecutive patients.[20] Four years of followup was available in 104 patients. The mean length of hospitalization was 3 days after Gamma knife surgery and 23 days after microsurgery. These numbers, however, do not reflect contemporary neurosurgical practice in the United States. Compared with 56% of patients in the microsurgical group, all but one patient who underwent Gamma knife surgery subsequently were employed. With respect to neurological outcome, 30% lost functional hearing, 50% developed tinnitus, 4% developed hypesthesia, 63% developed vertigo, 26% developed imbalance, 27% developed ocular problems, and 9% developed problems with swallowing.
Bertalanffy et al.reported 40 patients with 41 tumors, 21 of whom had undergone a previous surgical resection.[4] The median maximal tumor diameter was 25 mm. The median marginal dose was 12 Gy,encompassing the 40 to 95% isodose line. Within 6 to 12 months of surgery, central loss of contrast enhancement was observed in 78% of the patients. Nine patients were lost to follow up. Of the remaining 31 patients, the diameter of the tumor was stable or decreased in 29 patients. Of the 14 patients with useful hearing before surgery, nine had adequate audiograms at follow-up. Cochlear function deteriorated in no patients. Of 13 patients with preoperative facial weakness, two suffered postradiosurgical aggravation of the condition. New facial palsy occurred in two patients 4 years after treatment. Of the 13 patients with tinnitus before surgery, the condition improved in six and deteriorated in two. Trigeminal hypesthesia did not appear as a new permanent symptom; it improved in three of nine patients and deteriorated in one. Vertigo was present in 23 patients before surgery. It increased in six, was stable in eight, and decreased in nine.
The gold standard regarding longterm data regarding stereotactic radiosurgery was presented at the 12th Leksell Gamma Knife Society meeting in Vienna by the Pittsburg group in 2004.[13] During a 17-year period, 829 patients with acoustic neuromas were treated with Gamma knife stereotactic radiosurgery using all three models. Between 1987 and 1992, dose planning, dose selection, and imaging improved and have since remained fairly stable. The only significant change has been the ability to use smaller isocenters with the automatic positioning system introduced in the past 3 to 5 years.
Contemporary marginal doses are usually 13 Gy. In the Pittsburgh series reported in Vienna,no patient sustained significant peritreatment morbidity. Most were treated as outpatients. Of the patients with serviceable hearing before treatment,it was preserved in 59%.The incidence of facial nerve palsy was about 1%.Trigeminal nerve problems (numbness or pain) occurred in fewer than 3% of patients whose tumor impinged on the trigeminal nerve. The rate of tumor control 9 to 15 years after treatment was 97%. If control rates remain stable in these patients after 20 to 30 years of follow-up, our ability to recommend this modality as a primary treatment option will be strengthened.
Linear Accelerator Radiosurgery
From 1988 to 1998,Foote et al.used the linear accelerator (LINAC) to treat 149 cases,139 of which were included in their analysis.[9] The median duration of follow-up was 36 months (median radiographic follow-up,34 months). The 2-year actuarial rate of facial and trigeminal neuropathies was 11.8% and 9.5%, respectively. With contemporary radiosurgical dosing techniques, facial neuropathy occurred in 5% and trigeminal neuropathy occurred in 2%. Radiographic tumor control was achieved in 93% of the tumors (7.5% enlarged, 34% were unchanged, 59% regressed). The 5-year actuarial rate of tumor control was 87%. The marginal dose cutoff for growth risk was 10 Gy.
Proton-Beam Radiation
Between 1992 and 2000 at the Massachusetts General Hospital,Weber et al. used proton beam stereotactic radiosurgery to treat 88 patients.[28] Two to four convergent fixed beams of 160-MeV protons were applied. Treatment diameters ranged from 2.5 to 35 mm. Seventeen patients had undergone a previous surgical resection. The median dose of 12 cobalt Gy equivalents was prescribed with a 70 to 108% isodose line. The median length of follow-up was 38.7 months. The 2- and 5-year actuarial rates of tumor control were 95.3% and 94%,respectively. Three percent of the patients underwent shunting for hydrocephalus. Of 21 patients with serviceable hearing before surgery, 7 (33%) retained serviceable hearing after treatment. The 5-year rate of preservation of normal facial nerve function was 91%, and the 5-year rate of preservation of normal trigeminal nerve function was 89%. Inhomogeneity coefficient dose and prescribed dose were associated with a significant risk of long-term facial neuropathy. The authors concluded that proton-beam stereotactic radiosurgery was an effective means of controlling tumors.
From 1991 to 1999, Bush et al. treated 30 patients with acoustic neuromas (mean tumor volume, 4.3 cm3) with proton-beam radiotherapy.[5] Daily fractions were given in 1.8 to 2.0 cobalt Gy equivalents. Patients with serviceable hearing received 54 cobalt Gy equivalents in 30 fractions. Patients without useful hearing received 60 cobalt Gy equivalents in 30 to 33 fractions. No tumor growth was found on follow-up MRI (mean, 34 months). Serviceable hearing (Gardner-Robertson grade I or II) was maintained in 31%. No transient or permanent treatment-related trigeminal or facial nerve dysfunction occurred.

Fractionated Stereotactic Radiosurgery and Radiotherapy
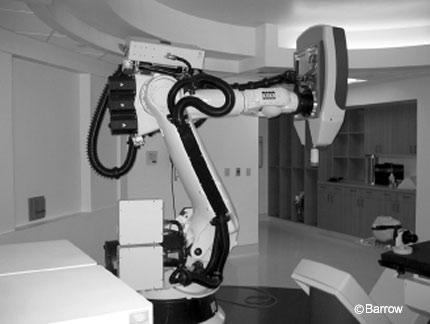
Although single-fraction stereotactic radiation is an important treatment option for acoustic neuromas,new stereotactic techniques that do not require the use of head frames have been developed (Figs. 5 and 6).
With improved imaging quality and with the ability to track patients in space and to reposition patients daily with submillimeter accuracy (Fig. 7), the use of fractionated beams applied according to radiobiological principles may increase the rate of cranial nerve preservation and lower the chance of morbidity.
At the Cyberknife meeting in Napa Valley, November 2003, the Stanford group reported 61 patients treated with fractionated stereotactic radiosurgery using the Cyberknife with more than 2 years of follow-up. Ninety patients excluded from the study had less than a 2-year follow-up. No acoustic neuroma increased in size and 48% decreased after treatment. Hearing was preserved in 80%, stable in 76%,and improved in 4%.Transient facial twitching occurred in two patients. No permanent facial paralysis or was 33 months. The 5-year local control rate was 100% of patients undergoing single-fraction treatment compared to 94% in the fractionated group. At 5-years,the rate of facial nerve preservation was 93% of those undergoing singlefraction radiosurgery compared to 97% of those undergoing fractionated treatment (25 Gy administered over five fractions at the 80% isodose line).There was no significant difference in the rate of hearing preservation. However, there was a significant difference in the rate of trigeminal neuropathy: 8% for patients undergoing single-fraction radiosurgery compared with 2% of those undergoing fractionated radiosurgery.
Williams[29] reported 150 patients with follow-up greater than 1 year treated with three different fractionation schemes. One hundred thirty-one patients with acoustic neuromas less than 3 cm in diameter received the 5 Gy in five consecutive daily fractions. Patients with tumors larger than 3 cm and smaller than 3.9 cm (mean volume,8.7 cc) received a total of 30 Gy in 10 fractions, and patients with tumors larger than 4 cm received a total of 40 Gy in 20 fractions. All treatments were prescribed with an 80% isodose line. The decrease in tumor size was 14%, 15%, and 8% for those treated with 25, 30, and 40 Gy regimens, respectively. No patient’s acoustic neuroma grew, and no patient developed facial weakness. Two patients experienced a transient decrease in facial sensation. The rates of hearing preservation were similar for both larger and smaller tumors.
Poen et al. reported 33 patients with acoustic neuromas who received 21 cGy in three fractions during a 24-hour period using conventional frame-based LINAC radiosurgery.[18] The median tumor diameter was 20 mm. Thirty-two patients were accessible for follow-up, and 21 patients had serviceable hearing before treatment. The median followup was 2 years. The tumor regressed or stabilized in 97% and progressed in 1 patient (3%). Sixteen percent of the patients suffered trigeminal neuropathy. One patient (3%) developed facial nerve injury (House-Brackmann grade 3) 7 months after stereotactic radiosurgery. Useful hearing (Gardner-Robertson Class 1-2) was preserved in 77%. At their last follow-up, all patients who had undergone pretreatment (Gardner-Robertson Class 1 or 2) had maintained serviceable hearing.
Sawamura et al. treated 101 patients with an acoustic neuroma with fractionated stereotactic radiosurgery. Over 5 to 6 weeks, patients received 40 to 50 Gy administered in 20 to 25 fractions.[23] The median tumor size was 19 mm (median follow-up,45 months; actuarial tumor control growth rate, 91%). Three patients with progressive tumors underwent a salvage operation,and useful hearing was preserved in 71%.Transient facial nerve palsy occurred in 4%, trigeminal neuropathy occurred in 14%, and balance disturbance occurred in 17%. No patient developed a new permanent facial weakness after treatment.
Radiosurgical Treatment of Acoustic Neuromas Associated with Neurofibromatosis-2
Preservation of hearing function in patients with neurofibromatosis type 2 (NF-2) is paramount because these patients have bilateral tumors and are at risk for complete deafness in both ears. Subach et al. reported their experience with 40 patients who underwent stereotactic radiosurgery, 35 for solitary tumors.[26] The other five patients underwent treatment for both lesions. Thirteen patients had undergone previous resection. The median tumor volume was 4.8 mm[3], and the mean tumor marginal dose was 15 Gy. Tumor control was achieved in 98% of the patients: 36% of the tumors regressed and 62% were stable. During a median follow-up of 36 months, tumors grew in 2% of the patients. More than a 5-year follow-up was available in 10 patients: Five tumors were smaller and five were unchanged. Seven percent of the patients underwent surgical resection. Serviceable hearing was preserved in 43% of patients. This rate improved to 67% after the contemporary neurosurgical dose of 13 Gy was introduced. Facial nerve function was normal in 81%,and trigeminal nerve function was normal in 94%.
Kida et al.[11] reported their experience with radiosurgery for the treatment of bilateral acoustic neuromas associated with NF-2. They treated patients with growing tumors less than 30 mm in diameter, patients with an ipsilateral deaf ear, and patients at risk of brain stem compression. Twenty patients with a mean tumor diameter of 24 mm were treated with Gamma knife radiosurgery with a mean marginal dose of 13 Gy. Eight patients had serviceable hearing in their treated ear. The tumor regressed in 60% of the patients (mean follow-up, 36 months). Contralateral untreated tumors enlarged in 40% of the cases. Serviceable hearing was preserved in 33% of the treated tumors. Facial nerve function deteriorated in 10%.
Rowe et al. reported their radiosurgical experience with 96 patients with NF-2.[21] Serviceable hearing deteriorated in 40% and 20% became deaf. Facial nerve and trigeminal nerve neuropathy occurred in 5% and 2 % of the patients, respectively.
Malignant Transformation
Despite excellent tumor control rates after stereotactic radiosurgery, malignant transformation has been reported.[2,10,14,16,25,27] Shin et al. reported a 26-year-old woman who had undergone subtotal resection of her tumor through a suboccipital approach.[25] At that time,the tumor had shown no sign of atypia. One month after surgery,17 Gy with a 50% isodose line was delivered to the tumor margin to prevent regrowth of the residual tumor. Six years after radiosurgery, the patient noticed progressive facial dysesthesia. At that time MRI showed regrowth of the tumor,and she underwent a second surgical resection. Histological examination showed a dense cellular mass of atypical cells,pleomorphism,and high mitotic activity,which was interpreted as a malignant schwannoma. After the second resection, the tumor grew rapidly despite surgical debulking and adjunctive chemotherapy. The patient died 10 months later with carcinomatous meningitis. A point mutation, which had not been present at the initial surgical resection, was found in the TP 53 gene. A high expression of the mutant P53 protein was present in the second but not first tumor.
Hanabusa et al.reported a 57-year-old woman with a right-sided hearing disturbance who underwent a retrosigmoid approach.[10] A typical benign neuroma was found. Residual tumor growth was noted 4 years after the surgery, and the patient underwent Gamma knife stereotactic radiosurgery. Six months later the tumor had grown. In a second operation, the tumor was accessed through a translabyrinthine approach. Abnormal mitotic figures indicating malignancy were observed on histological studies. The patient died 6.5 years after her initial surgery.
Malignant nerve sheath tumors of the vestibulocochlear nerve have been reported in only five patients. Four had a malignant triton tumor and one had a malignant triton tumor with NF-2.[2,10,14,16,25,27] This rare soft-tissue type sarcoma exhibits rhabdoid myoblastic differentiation. Seventy percent of these cases are associated with NF.
Finally, Shamisa et al.[24] reported a malignant glial tumor in a patient 7.5 years after Gamma knife stereotactic radiosurgery. A glioblastoma developed in the inferior temporal lobe adjacent to the area of radiosurgery.
Conclusions
Stereotactic radiosurgery for acoustic neuromas is an attractive alternative to control tumors smaller than 3 cm exhibiting significant growth on serial imaging. No prospective, randomized trials comparing the outcomes of microsurgical and radiosurgical treatment have been performed,and studies with a follow-up longer than 10 years are not widely available. Contemporary stereotactic radiosurgery techniques have minimized cranial neuropathies. Although possible,malignant transformation is extremely rare. Fractionated stereotactic radiosurgery may improve functional outcomes with respect to serviceable hearing,trigeminal neuropathy, and facial neuropathy while providing tumor control rates similar to those associated with single-fraction treatment.
References
- Balance CA: Some Points in the Surgery of the Brain and its Membranes. London: Macmillan, 1907
- Bari ME, Forster DM, Kemeny AA, et al: Malignancy in a vestibular schwannoma. Report of a case with central neurofibromatosis, treated by both stereotactic radiosurgery and surgical excision, with a review of the literature. Br J Neurosurg 16:284-289, 2002
- Bell C: Appendix of Cases. London: Longman, Rees, Orme, Brown, and Green, 1830, pp 112-114
- Bertalanffy A, Dietrich W, Aichholzer M, et al: Gamma knife radiosurgery of acoustic neurinomas. Acta Neurochir (Wien) 143:689-695, 2001
- Bush DA, McAllister CJ, Loredo LN, et al: Fractionated proton beam radiotherapy for acoustic neuroma. Neurosurgery 50:270-273, 2002
- Cushing H: Tumors of the Nervus Acusticus and the Syndrome of the Cerebellopontine Angle. Philadelphia: WB Saunders, 1917
- Dandy WE: An operation for the total removal of cerebellar pontine (acoustic) tumors. Surg Gynecol Obstet 41:121-148, 1925
- Flickinger JC, Kondziolka D, Niranjan A, et al: Results of acoustic neuroma radiosurgery: an analysis of 5 years’ experience using current methods. J Neurosurg 94:1-6, 2001
- Foote KD, Friedman WA, Buatti JM, et al: Analysis of risk factors associated with radiosurgery for vestibular schwannoma. J Neurosurg 95:440-449, 2001
- Hanabusa K, Morikawa, Murata T, et al: Acoustic neuroma with malignant transformation. Case report. J Neurosurg 95:518-521, 2001
- Kida Y, Kobayashi T, Tanaka T, et al: Radiosurgery for bilateral neurinomas associated with neurofibromatosis type 2. Surg Neurol 53:383-389, 2000
- Kondziolka D, Lunsford LD, McLaughlin MR, et al: Long-term outcomes after radiosurgery for acoustic neuromas. N Engl J Med 339:1426-1433, 1998
- Lunsford LD, Kondziolka D, Niranjan A, et al: Gamma knife radiosurgery for acoustic neuromas. 12th International Meeting of the Leksell Gamma Knife Society, May 2004, Vienna, Austria, p 40
- Lustig LR, Jackler RK, Lanser MJ: Radiation-induced tumors of the temporal bone. Am J Otol 18:230-235, 1997
- Meijer OW, Vandertop WP, Baayen JC, et al: Single-fraction vs. fractionated LINAC-based stereotactic radiosurgery for vestibular schwannoma: a single-institution study. Int J Radiat Oncol Biol Phys 56:1390-1396, 2003
- Muracciole X, Cowen D, Regis J: Radiosurgery and brain radio-induced carcinogenesis: Update [French]. Neurochirurgie 50:414-420, 2004
- Petit JH, Hudes RS, Chen TT, et al: Reduceddose radiosurgery for vestibular schwannomas. Neurosurgery 49:1299-1306, 2001
- Poen JC, Golby AJ, Forster KM, et al: Fractionated stereotactic radiosurgery and preservation of hearing in patients with vestibular schwannoma: a preliminary report. Neurosurgery 45:1299-1305, 1999
- Prasad D, Steiner M, Steiner L: Gamma surgery for vestibular schwannoma. J Neurosurg 92:745-759, 2000
- Regis J, Pellet W, Delsanti C, et al: Functional outcome after gamma knife surgery or microsurgery for vestibular schwannomas. J Neurosurg 97:1091-1100, 2002
- Rowe JG, Radatz MW, Walton L, et al: Clinical experience with gamma knife stereotactic radiosurgery in the management of vestibular schwannomas secondary to type 2 neurofibromatosis. J Neurol Neurosurg Psychiatry 74:1288-1293, 2003
- Sandifort E: Observationes Anatomico-pathologicae. Leiden: Lugduni Batavorum, 1777, pp 116-120
- Sawamura Y, Shirato H, Sakamoto T, et al: Management of vestibular schwannoma by fractionated stereotactic radiotherapy and associated cerebrospinal fluid malabsorption. J Neurosurg 99:685-692, 2003
- Shamisa A, Bance M, Nag S, et al: Glioblastoma multiforme occurring in a patient treated with gamma knife surgery. Case report and review of the literature. J Neurosurg 94:816-821, 2001
- Shin M, Ueki K, Kurita H, et al: Malignant transformation of a vestibular schwannoma after gamma knife radiosurgery. Lancet 360:309-310, 2002
- Subach BR, Kondziolka D, Lunsford LD, et al: Stereotactic radiosurgery in the management of acoustic neuromas associated with neurofibromatosis Type 2. J Neurosurg 90:815-822, 1999
- Thomsen J, Mirz F, Wetke R, et al: Intracranial sarcoma in a patient with neurofibromatosis type 2 treated with gamma knife radiosurgery for vestibular schwannoma. Am J Otol 21:364-370, 2000
- Weber DC, Chan AW, Bussiere MR, et al: Proton beam radiosurgery for vestibular schwannoma: tumor control and cranial nerve toxicity. Neurosurgery 53:577-586, 2003
- Williams JA: Fractionated stereotactic radiotherapy for acoustic neuromas. Acta Neurochir (Wien) 144:1249-1254, 2002
