
FDA Approves Indego for More Spinal Cord Injury Patients
“I cried, to tell you the truth,” he said. “Being upright, it was very fulfilling.”
It was his first time wearing the Indego, a robotic device that can be fitted to the legs to allow people with spinal cord injury to walk and participate in gait training.
Jimenez is now up to 1,200 steps with the Indego in a single session. He can even take some steps on his own with only the support of a walker.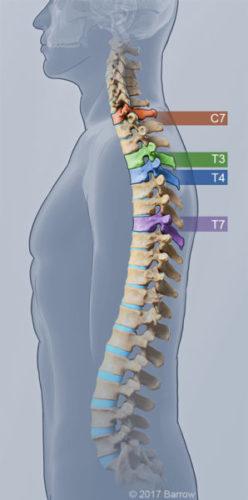
The U.S. Food and Drug Administration (FDA) has cleared an expanded indication for use for the Indego, which will allow even more people to benefit from the device.
Individuals with an injury at the level of the C7 vertebra or lower can now use the Indego in rehabilitation facilities, and those with an injury at the level of the T3 vertebra or lower can obtain a personal device for greater mobility at home and in the community. The FDA previously cleared the device for the T4 level and lower for rehab and the T7 level and lower for personal use.
Parker Hannifin, the maker of the device, said in a press release that the recent clearance followed the successful implementation of an FDA 522 Postmarket Surveillance Study—a type of study designed to continue monitoring the safety of a medical device after it has been placed on the market. The company is also conducting two clinical trials: one to evaluate the effectiveness of the Indego as a gait therapy tool for spinal cord injury patients and another to evaluate the safety of the device for stroke patients with paralysis on one side of the body.
Barrow Patient Builds Strength, Confidence With Indego
Jimenez, now 43, woke up without movement from the waist down after undergoing emergency spinal surgery in April. The New Yorker was visiting his mom in Phoenix when his left leg and foot started going numb. An MRI revealed an abnormal tangle of blood vessels, called an arteriovenous malformation (AVM), at the L5 level of his spine.

Without surgical treatment, a spinal AVM can permanently damage the spinal cord by depriving it of oxygen. The AVM can also rupture and cause bleeding into the spinal cord.
Jimenez started using the Indego as a Barrow patient and is now using the device as a study participant due to loss of insurance coverage. He visits the Barrow Outpatient Neuro-Rehabilitation Center once a week, in addition to maintaining his own exercise regimen. He credits the Indego with improving his strength, posture, and confidence.
“What’s nice about the device is it’s able to simulate a pretty near-normal gait pattern,” said Al Biemond, a physical therapist at Barrow. “In areas where Kevin has some strength, we can either turn up or turn down the amount of assistance that the device provides in different components of the walking pattern. It makes him work harder so that he has to strengthen certain muscles.”
Therapists can adjust the level of assistance provided by the device, as well as collect performance data for each patient, through a smartphone application. The Indego’s modular design also allows therapists to modify the device to fit various patients.
Regaining Independence After Spinal Cord Injury
The Indego works by mirroring natural human movements through postural cues. The user leans forward to initiate standing or walking and leans backward to initiate stopping or sitting. A fall-mitigation system kicks into gear if the user leans too far.
Although the 26-pound suit is not intended for all activities, such as playing sports or climbing stairs, it may enable people to perform many daily tasks and help them regain independence.
According to Parker Hannifin, the expanded clearance makes the personal Indego an option for more than 40 percent of Americans with spinal cord injury.
I think we’re eventually going to see these kinds of devices, whether it be Indego or others, in people’s homes. It just makes sense.
-Al Biemond, Barrow physical therapist
“I think one of the bigger barriers is going to be the cost associated with that and which of the insurance companies out there would even be willing to consider that as a reasonable request,” Biemond said. “I think some of the payer sources that might move forward with this are going to be those that serve veterans.”
Parker Hannifin said that several medical directors from Veterans Affairs encouraged the company to seek the expanded clearance from the FDA. The company said it is likely some of the 40,000 veterans with spinal cord injuries who are served by the VA will be among the immediate beneficiaries.
Biemond expects the barrier of insurance coverage to be broken down over time as more people request devices like the Indego and as competition drives down the price tag.
“Payer sources are always going to be hard, and this is true with any kind of new technology,” he said. “I think we’re eventually going to see these kinds of devices, whether it be Indego or others, in people’s homes. It just makes sense.”
