
Role of Motor Cortex in Control of Walking*
Irina N. Beloozerova, PhD
Mikhail G. Sirota, PhD
Division of Neurobiology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
*Courtesy of Irina N. Beloozerova, PhD
The Beloozerova Laboratory: Irina Beloozerova received her PhD in physiology from the Lomonosov University in Moscow, Russia, for her studies on the role of the motor cortex in the control of walking. She then studied the physiology of the spinal cord with Serge Rossignol at the University of Montreal, Canada, and the physiology of the somatosensory cortex with Harvey Swadlow at the University of Connecticut. Mikhail Sirota obtained his PhD in physiology from the Lomonosov University in Moscow, Russia, where he studied the mechanisms of initiation of walking in the laboratory of Mark Shik. He then headed the Laboratory of Microgravity Neurophysiology at the Institute of Biomedical Problems in Moscow, Russia and studied the effects of weightlessness in outer space on motor coordination. Zinayida Tamarova obtained her PhD in physiology from the A. A. Bogomoletz Institute of Physiology, in Kiev, Ukraine, working in the laboratory of Platon Kostuk on the spinal mechanism underlying visceromotor reflexes. She then studied mechanisms of synaptic transmission in the spinal cord and influences of higher brain centers on motoneurons with Alexander Shapovalov in Leningrad, Russia and central mechanisms of pain with Yury Limansky in Kiev, Ukraine.
Abstract
Walking includes voluntary components that are necessary to overcome obstacles and to change directions. In cats and humans, these movements are not possible without the contribution of the motor cortex. One focus of our laboratory is the role of the motor cortex in the control of walking. The activity of the motor cortex of cats was recorded during simple and complex (i.e., involving overcoming obstacles) walking. The activity of the motor cortex changed dramatically when cats had to negotiate obstacles compared to simple walking conditions. Lesions to the motor cortex made cats incapable of negotiating obstacles correctly. During complex walking it appears that the activity of the motor cortex contains commands addressed to the spinal cord and that these commands adjust the spinal locomotor mechanisms to specific structures of the pathway.
Key Words: locomotion, motor cortex
Abbreviations used: SD, standard deviation; TTX, tetrodotoxin
Walking includes voluntary components that are necessary for overcoming obstacles and for changing the direction of movements. The point at which to step often must be chosen carefully. The system of reception with which the spinal cord is equipped is sufficient to ensure walking on a flat surface; however, it is incapable of adapting movements to a complex environment.[13] Natural locomotion is impossible without the contribution of supraspinal centers to control stepping movements. One focus of research in our laboratory is the involvement of the motor cortex in the control of walking in a complex environment.
Commands from supraspinal motor centers reach the spinal cord by various descending tracts.[1,4,5] The activity of nerve cells that contribute their long branches, axons, to the reticulospinal, vestibulospinal, rubrospinal, and pyramidal tracts was studied in walking animals. [2,3,7,8,15,16,17] The activity of these neurons is modulated rhythmically, and the modulation is related to the stepping rhythm. The activity of the motor cortex changes during voluntary movements and, in some cases, before the movements begin.[9-12,14,19] Injury to the motor cortex damages the capacity to perform some voluntary limb movements or at least greatly interferes with the performance of these movements.
We recorded the activity of neurons in the motor cortex in cats during walking. Two conditions were tested: (1) walking on a flat horizontal surface (simple walking) and (2) walking while overcoming various obstacles (complex walking). The latter requires visual inspection of the environment and appropriate adaptation of steps. We compared the activity of neurons of the motor cortex under simple and complex conditions, assuming that the difference in that activity would be related to the difference in the tasks, thereby revealing the cortical “voluntary” motor command underlying precise stepping.
Methods
Positive reinforcement (food) was used to adapt cats to the experimental situation and to engage them in locomotor behavior.[18,20] A rectangular enclosure (2.5 m x 0.5 m) served as an experimental chamber. A longitudinal wall divided the box into two corridors through which the cats passed sequentially and repeatedly in either direction. In one corridor the floor was flat; the other corridor contained a horizontal ladder or a set of barriers (Fig. 1).
Once trained, the cat underwent surgery during which a plastic base was fixed to the skull. A microdriver and an amplifier were then attached to the base. The bone above the motor cortex was replaced with a plastic plate that had about 100 small holes prefilled with sterile bone wax. Subsequently, the recording electrode was introduced through the holes into the brain.
The activity of a neuron during a step was assessed as was its activity before locomotion. For analysis of neuronal activity, a step was divided into 10 intervals, and a postevent time histogram of spike distribution throughout the step was obtained for 20 to 200 successive steps. The difference between the maximum and minimum firing frequency associated with a step divided by the mean frequency was used as an index to the depth of rhythmic step-related modulation.
Results
Simple Walking
During standing, the discharge frequency of motor cortical neurons was 1 to 30 impulses (mean ± SD, 9.8 ± 1). During the transition from standing to locomotion, the average activity of 36% of the neurons increased about two-fold and it increased three to five-fold in 20%. We analyzed the activity of neurons immediately before the first step, defined as the number of spikes in each 100 ms interval during the 2 s preceding the first step. During this period the activity of 70% of the neurons remained unchanged, but the activity of 25% of the neurons increased (Fig. 2).
The activity of 252 neurons in the motor cortex was recorded during simple walking. The activity of 89% of these neurons was modulated in relation to the stepping movements; that is, activity increased during one step phase and decreased during the next (Fig. 2). The maximum activity in 65% of the “modulated” neurons coincided with the swing phase of the contralateral forelimb (as in Fig. 2A) and in 35% of the neurons, with the stance phase (as in Fig. 2B). The mean depth of frequency modulation was 25 ± 2 %.

Complex Walking
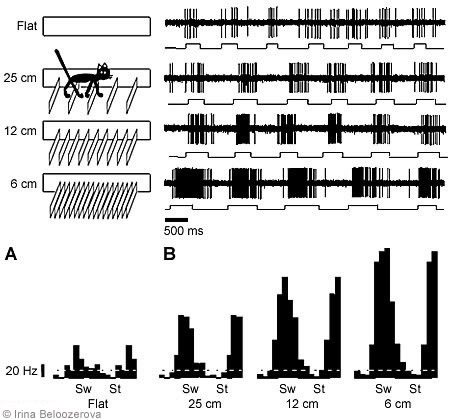
Overstepping Barriers. The possible positions of the cat’s feet on the floor were restricted by arranging barriers along the cat’s path. The height of the barriers was 70 mm, and the distance between successive barriers was 250 mm (i.e., equal to an average cat’s step; Fig. 3). To walk along the box, the cat had to step between the barriers.
Under these conditions, the activity of 68 neurons in the motor cortex was recorded. In 79% of the neurons, the average activity during walking with barriers differed from that during simple walking: Activity increased in 50% and decreased in 29%. The depth of frequency modulation changed in 84% of the neurons, increasing in 59% and decreasing in 25%. In 91% of cases, the phases of maximum activity were the same during both simple and complex locomotion (Fig. 3).
We also tested the activity of the same neurons with smaller interbarrier intervals (i.e., the cats had to step with greater accuracy). In all neurons tested, the average activity and depth of modulation increased as the interbarrier interval decreased (Fig. 3).
Walking on Rungs of a Horizontal Ladder. The horizontal ladder had 50-mm wide rungs, and the distance between successive rungs was 250 mm. When the cats walked on the ladder, the possible position of a limb landing on the supporting surface was restricted to the rungs. In contrast, during the previous test conditions, cats could place their feet anywhere between the barriers.
The activity of 108 neurons in the motor cortex was recorded while cats walked along the ladder. Compared to simple walking, walking on the ladder changed the average level of activity in 81% of the neurons: Activity increased in 71% and decreased in 10%. The depth of frequency modulation changed in 80% of the neurons: Modulation increased in 56% of the neurons and decreased in 24% of the neurons: The phases of maximum and minimum activity persisted in most neurons.
Effects of Lesioning or Chemical Inactivation of the Motor Cortex on Walking
Neither bilateral permanent lesions of the motor cortex, nor reversible inactivation by cooling, nor the neurotoxin TTX hampered simple walking. In contrast, cats with a lesion or inactivated motor cortex could no longer step over obstacles (they knocked them over). They also could not walk on the ladder (they missed the rungs). After lesioning, their inability to perform a complex walk persisted 5 to 7 days. Attempts at walking then became more successful. After about 1 week, cats managed to perform the complex walking tasks without making mistakes. This finding suggests that some other undamaged brain structures assumed the function of the motor cortex after injury to control the accuracy of stepping.
Discussion
Simple Walking
In many neurons a tonic change in activity was observed before a cat took its first step. This change seems to be associated with the postural adjustments involved in the transition from standing to locomotion. When a cat walked on the flat horizontal surface with no obstacles, the activity of most motor cortical cells was modulated rhythmically, with the modulation related to the rhythm of the stepping movements. Various neurons in the motor cortex showed maximum activity during various phases of a step, but most neurons were most active during the swing phase of the corresponding limb.
What is the source of the rhythmic signal modulation of the activity of neurons in the motor cortex? Armstrong and Drew[3] blocked nerves with novocaine and found that elimination of somatic sensation did not abolish the rhythmic modulation of the neurons. This finding suggested that the source of modulatory signals is not at the periphery but rather involves central neuronal mechanisms. In experiments with exercise belt walking,[4] neurons of tracts descending from the brainstem to the spinal cord were modulated rhythmically in relation to the stepping cycle. The source of their modulatory signals proved to be the spinal neuronal mechanism generating the stepping rhythm. Spinal influences on these neurons are mediated by the cerebellum — another brain structure closely involved with the control of movements. Neurons in the motor cortex likely receive modulatory commands via the same route because their rhythmical modulation decreased considerably after a nucleus that mediates cerebellar influences on the motor cortex was destroyed.[6]
Complex Walking
Our main finding is that the pattern of activity of most cortical motor neurons changed dramatically when cats had to perform stepping movements with accuracy. The information concerning the peculiarities of the walking pathway was obtained via the visual system. Therefore, the activity of neurons in the motor cortex changes considerably when the visual system is involved in the control of walking.
A lesion to the motor cortex renders an animal incapable of performing space-linked stepping under the control of vision. Consequently, it seems likely that the activity of neurons in the motor cortex involving pronounced rhythmical modulation, as observed in our experiments, constitutes the cortical commands addressed to the spinal cord. These commands suitably adjust the operation of the spinal mechanisms to external conditions. Understanding the formation and the content of these commands may help in the design of brain-machine interfaces that will help people with damage to the motor cortex or corticospinal pathway.
References
- Armstrong DM: Supraspinal contributions to the initiation and control of locomotion in the cat. Prog Neurobiol 26:273-361, 1986
- Armstrong DM, Drew T: Discharges of pyramidal tract and other motor cortical neurones during locomotion in the cat. J Physiol 346:471-495, 1984
- Armstrong DM, Drew T: Locomotor-related neuronal discharges in cat motor cortex compared with peripheral receptive fields and evoked movements. J Physiol 346:497-517, 1984
- Arshavsky Y, Gelfand IM, Orlovsky GN: Cerebellum and Rhythmical Movements. Berlin, New York: Springer-Verlag, 1986
- Arshavsky YI, Gelfand IM, Orlovsky GN, et al: Messages conveyed by descending tracts during scratching in the cat. I. Activity of vestibulospinal neurons. Brain Res 159:99-110, 1978
- Arshavsky YI, Orlovsky GN, Pavlova GA, et al: Messages conveyed by descending tracts during scratching in the cat. II. Activity of rubrospinal neurons. Brain Res 159:111-123, 1978
- Asanuma H, Babb RS, Mori A, et al: Input-output relationships in cat’s motor cortex after pyramidal section. J Neurophysiol 46:694-703, 1981
- Beloozerova IN, Sirota MG: Role of motor cortex in control of locomotion, in Gurfinkel VS, Ioffe ME, Massion J, et al (eds): Stance and Motion. Facts and Concepts. New York: Plenum Press, 1988, pp 163-176
- Beloozerova IN, Sirota MG: The role of the motor cortex in the control of accuracy of locomotor movements in the cat. J Physiol 461:1-25, 1993
- Beloozerova IN, Sirota MG: The role of the motor cortex in the control of vigour of locomotor movements in the cat. J Physiol 461:27-46, 1993
- Brooks VB: Some examples of programmed limb movements. Brain Res 71:299-308, 1974
- Evarts EV: Relation of pyramidal tract activity to force exerted during voluntary movement. J Neurophysiol 31:14-27, 1968
- Georgopoulos AP, Ashe J, Smyrnis N, et al: The motor cortex and the coding of force. Science 256:1692-1695, 1992
- Georgopoulos AP, Kettner RE, Schwartz AB: Primate motor cortex and free arm movements to visual targets in three-dimensional space. II. Coding of the direction of movement by a neuronal population. J Neurosci 8:2928-2937, 1988
- Grillner S, Rossignol S: On the initiation of the swing phase of locomotion in chronic spinal cats. Brain Res 146:269-277, 1978
- Mountcastle VB, Atluri PP, Romo R: Selective output-discriminative signals in the motor cortex of waking monkeys. Cereb Cortex 2:277-294, 1992
- Orlovsky GN, Shik ML: Control of locomotion: A neurophysiological analysis of the cat locomotor system. International Review of Physiology 10:281-317, 1976
- Pryor K: Lads Before the Wind. New York: Harper and Row, 1975
- Scott SH, Kalaska JF: Reaching movements with similar hand paths but different arm orientations. I. Activity of individual cells in motor cortex. J Neurophysiol 77:826-852, 1997
- Skinner BF: The Behavior of Organisms: An Experimental Analysis. New York: Appleton-Century, 1938
Related Readings
- Beloozerova IN, Sirota MG, Swadlow HA: Activity of different classes of neurons of the motor cortex during locomotion. J Neurosci 23:1087-1097, 2003
- Beloozerova IN, Sirota MG, Orlovsky GN, Deliagina TG: Activity of pyramidal tract neurons in the cat during postural corrections. J Neurophysiol 93:1831-1844, 2005
- Beloozerova IN and Sirota MG: Cortically controlled gait adjustments in the cat. Ann N Y Acad Sci 860:550-553, 1998
- Beloozerova IN and Sirota MG: Integration of motor and visual information in the parietal area 5 during locomotion. J Neurophysiol 90:961-971, 2003
- Beloozerova IN and Sirota MG: The role of the motor cortex in the control of vigour of locomotor movements in the cat. J Physiol 461:27-46, 1993
- Beloozerova IN and Sirota MG: The role of the motor cortex in the control of accuracy of locomotor movements in the cat. J Physiol 461:1-25, 1993
- Prilutsky BI, Sirota MG, Gregor RJ, Beloozerova IN: Quantification of motor cortex activity and full-body biomechanics during unconstrained locomotion. J Neurophysiol, in press
- Sirota MG, Swadlow HA, Beloozerova IN: Three channels of corticothalamic communication during locomotion. J Neurosci 25(25):5915-5925, 2005
