
Thoracoscopic Approaches for the Treatment of Anterior Thoracic Spinal Pathology
Authors
Curtis A. Dickman, MD†
Camilla Mican, MD*
†Division of Neurological Surgery, Barrow Neurological Institute *Division of Cardiothoracic Surgery, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
Microsurgical approaches for the treatment of pathology of the ventral thoracic spine involving the disk spaces and vertebral bodies can be performed with minimally incisional surgery. Narrow portals are placed in the intercostal spaces to insert working tools and a rigid rod lens endoscope for visualization, magnification, and illumination. Video-assisted thoracoscopic techniques can be used for a variety of spinal surgeries: thoracic microdiskectomies, thoracic vertebrectomies, anterior releases of spinal deformity, interbody fusion, reconstruction of the spine, and placement of internal fixation devices. These techniques have distinct advantages compared to transthoracic and posterolateral approaches to anterior spinal pathology. Small incisions are made in the intercostal spaces, without retracting any ribs. This approach can reduce postoperative pain, shorten the length of hospitalization, improve cosmetic appearances, and allow early resumption of activity. This report describes the surgical techniques involved in this unique operative exposure of the spine.
Key Words : diskectomy, minimally invasive surgery, spine, thoracoscopy, vertebrectomy, video-assisted endoscopic surgery
Minimally invasive surgery has revolutionized almost all areas of surgery. The use of endoscopes permits surgical maneuvers to be performed through small incisions that facilitate a patient’s recovery. Endoscopes have been used for microsurgical magnification and illumination, as arthroscopes in orthopedics; as laparoscopes in general surgery, gynecology and urology; as thoracoscopes in cardiothoracic surgery; and as ventriculoscopes in cranial neurosurgery. They also have been used as diagnostic and therapeutic tools in more traditional endoscopic approaches in otolaryngology, urology, pulmonology, and gastroenterology, among other specialties.
Significant clinical demands exist for minimally invasive surgery. Patients prefer these techniques because they reduce recovery time and have cosmetic benefits. Experience with thoracoscopy versus thoracotomy for diseases of the chest has demonstrated that small incisions that minimize dissection and retraction of the chest wall decrease blood loss and reduce postoperative pain.1,5-7 These techniques shorten length of stays in the intensive care unit as well as overall hospitalization, hasten recovery times and a patient’s return to work, and lower the costs of medical care.1,3-5 Already, thoracoscopic surgery has almost replaced thoracotomy in thoracic surgery for a variety of diagnostic and therapeutic purposes.1,5-7 Biopsy of lesions of the lung and chest wall, pulmonary nodule resections, empyema drainage, resection of adhesions, hemothorax drainage, lymph node biopsy, tumor staging and resection, sympathectomies, and other procedures are regularly performed using thoracoscopy.
Minimally incisional surgery is a more appropriate term than minimally invasive surgery for these operative procedures because the “conventional” operations with extensive anatomical dissections can be performed through the small incisions. This article describes how a variety of surgical procedures used to treat pathology of the anterior thoracic spine can be adapted to a thoracoscopic approach.
Video-assisted thoracoscopic surgery can be used for a variety of spinal indications. The nerve roots and the spinal cord can be decompressed, bone grafts can be placed for interbody fusion and vertebral body reconstruction, and internal fixation devices can be applied to stabilize the spine. Thoracoscopy can be used to perform thoracic sympathectomies, to resect thoracic disk herniations, to biopsy thoracic vertebral body lesions, to release complex spinal curvatures for reduction of scoliosis, to perform vertebrectomies, to resect tumors, to debride infections, and to treat spinal fractures. The thoracic spine is exposed easily by temporarily deflating one lung using a double-lumen endotracheal tube. The collapsed lung creates a large empty working space in the pleural space in which to access the thoracic spine.
Thoracoscopic Technique
Surgeons should be extremely familiar with endoscopic surgical techniques and with the anatomy of the thorax and the mediastinum as well as with the anatomy of the thoracic spine, the spinal cord, and the paraspinal structures. Thoracoscopic spinal surgery ideally is performed in conjunction with a cardiothoracic surgeon experienced in techniques of thoracoscopic surgery. This collaborative approach is recommended to prevent and treat complications and to optimize the benefit to patients.
The thoracic spine can be approached using either a right or a left thoracoscopic access, depending on the location and eccentricity of the pathology and on the regional anatomy. When possible, a right-sided approach is preferable because more spinal surface area is available behind the azygous vein than behind the aorta. The relative position of the great vessels should be inspected on preoperative computed tomography (CT) or magnetic resonance (MR) imaging studies. If exposure is needed at the level of T10 to T12, a left-sided approach is preferred because the liver causes the right diaphragm to ride higher, limiting visibility to the spine. Exposure from T1-T2 to the T12-L1 interspace is possible. The upper thoracic spine is approached from the upper intercostal spaces, near the axillae. The lower thoracic spine is accessed by retracting the diaphragm. The reader should refer to an anatomy textbook for a comprehensive review of thoracic and mediastinal anatomy. The following discussion focuses on details relevant to thoracoscopic spinal surgery and paraspinal anatomy.
Patient Positioning and Operating Room Setup
Video–monitoring screens are placed at several locations throughout the surgical suite to allow the entire surgical team to directly visualize the operative procedure as it is performed.
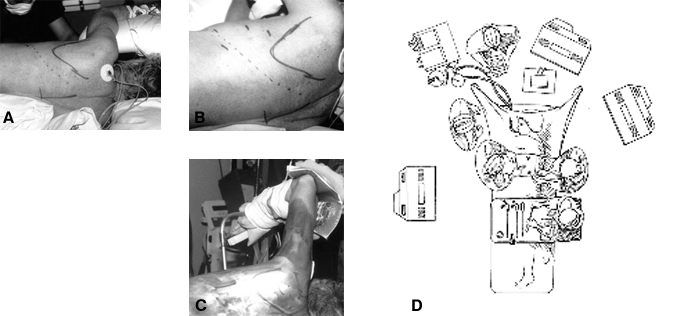
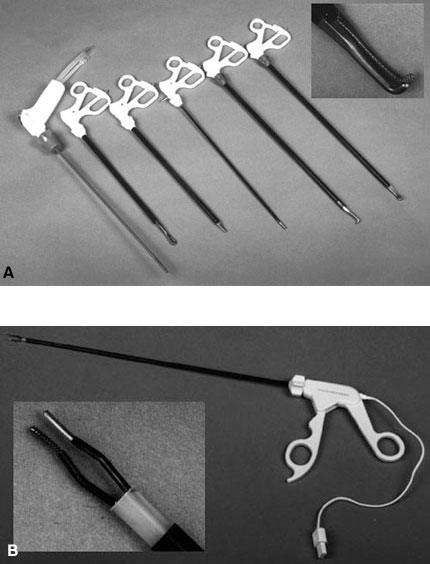
The patient is intubated with a double-lumen endotracheal tube and placed in a lateral decubitus position on the operating table (Fig. 1). The dependent axilla is padded and the upper arm is abducted, elevated anteriorly, supported, and padded. The lower leg is flexed and a pillow is placed between the patient’s legs to avoid compression neuropathies. The surgeon stands anteriorly to the patient’s chest. Cables and tubes for suctioning, irrigating, coagulating and the endoscope are routed from the sterile field at the top of the table to minimize tangling when devices are passed between the surgeon and the assistants.
The patient is placed in a true lateral position on the operating table to maintain the surgeon’s orientation in relationship to the spine. Intraoperatively, the table may be rotated anteriorly to allow the deflated lung to fall away from the spine, obviating the need to retract the lung. The superficial sites of the trochars are marked on the skin in the anterior and middle axillary lines, triangulated over the site of the spinal pathology. The scapula and the site for a potential thoracotomy incision are marked on the chest wall. The locations of the portal incisions are adjusted based on the level of the spinal pathology. In case an open thoracotomy is needed, a thoracotomy tray and a sponge “stick” are prepared and opened on the sterile field.
Portal Insertion
Usually, four portals are needed in the intercostal spaces (Fig. 2). The portals should not be positioned too closely together, or the tools will “fence” with each other over the surface of the chest. The portals are triangulated over the levels of the pathology. When possible, rigid portals are avoided because they have been associated with intercostal neuralgia from intercostal nerve compression.6-9 Therefore, flexible portals are used in most circumstances.
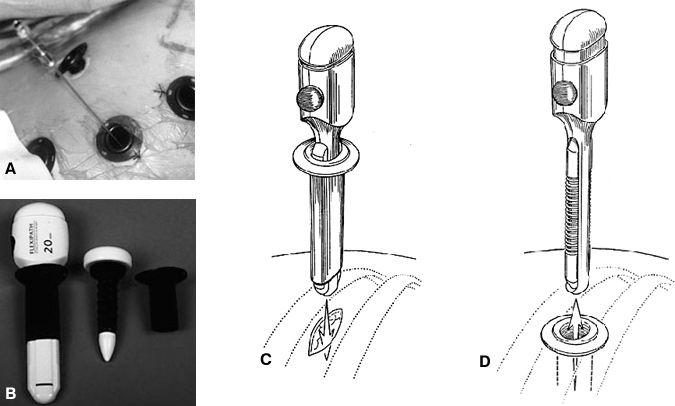
The technique used to insert the portals in the intercostal spaces closely resembles the techniques used for chest tube insertion, except the incisions through the muscles are not tunneled. The skin incisions are made directly over the intercostal entry sites. Portal incisions (15 to 20 mm long) are made over the intercostal spaces, parallel to the upper surface of the rib. The incisions are extended through the parietal pleura. Using a trochar, the flexible portals are inserted through the intercostal spaces. The incisions are positioned directly above the rib to avoid the neurovascular bundle, which is located along the undersurface of each rib. After the first portal is inserted, subsequent portals are placed under direct visualization with the endoscope. Penetration through the diaphragm must be avoided when any portals are inserted below the level of T7.
Thoracoscopic Imaging
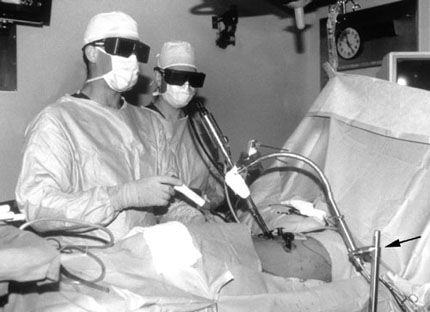
A 1-cm diameter rigid-rod lens endoscope with a 0Þ or 30Þ angle is used to provide visualization, magnification, and illumination. The images are projected from the thoracoscopic camera to video screens within the operating room. A table-mounted endoscope holder (Fig. 3) is used to stabilize the position of the thoracoscope intraoperatively. This endoscope holder is very useful—it fixates the image and frees the operative assistant’s hands for other tasks. A high resolution endoscope with a three chip camera or a three-dimensional endoscope (Endolive, 3D Video Endoscope, Carl Zeiss, Inc., Thornwood, NY) optimizes the visualization of the spine and hence appreciation for the complex three-dimensional intraoperative relationships of the spinal anatomy.
Fogging of the endoscope is prevented by prewarming the endoscope, using warm irrigation solutions, and wiping the lens with a sterile fog reduction/elimination detergent antifogging solution (FRED; Dexide Inc., Fort Worth, TX). The FRED solution is applied to a sponge on the sterile drapes. The endoscope lens is wiped intermittently with this solution to optimize visibility throughout the procedure. Irrigating and windshield wiper like cleaning mechanisms have been integrated into several types of endoscopes to clean debris from the surface of the lens. Because the operative view and illumination are limited, small amounts of blood over the tissue surfaces can absorb a large amount of light, thereby diminishing the quality of the endoscopic images. Powerful, adjustable xenon or halogen light sources are used to provide adequate illumination. Strong suction and pressurized irrigation devices are used to clear the surgical field rapidly.

Initial Spinal Exposure
Usually, the first thoracoscopic portal is placed in the sixth or seventh intercostal space after the ipsilateral lung has been deflated. Subsequently, the thoracoscope is inserted to visualize the contents of the thoracic cavity. If the operating table and the patient are rotated anteriorly, the deflated lung will usually fall away from the spine, minimizing the need for lung retraction. If necessary, the lung is gently retracted medially with an atraumatic endoscopic fan retractor (Fig. 4). The fan retractor can be opened, closed, and angled by the surgeon; however, care must be exerted to avoid lacerating the lung inadvertently with this tool. There is a risk of catching the lung tissue within the blades while closing the fan retractor. This problem can be avoided by directly observing all manipulations of the fan retractor with the endoscope. The fan retractor should be lifted away from the lung surface before the blades are closed. If the lung is torn intraoperatively, the air leak can be repaired with an endoscopic stapler. Fortunately, pulmonary injury is rare if the lung is retracted gently and the retractor is used carefully.
Spinal Localization
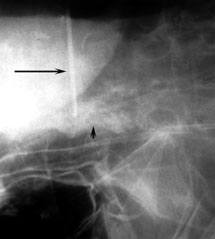
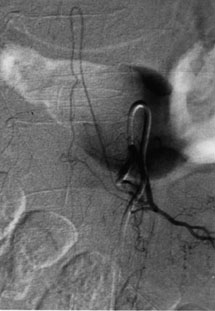
After the ribs and surface of the spine have been visualized, the ribs are counted to confirm the localization. The first rib can be palpated but usually cannot be visualized within the thoracic apex. The first visually identifiable rib is the second rib. Each subsequent rib is directly visualized and palpated. The ribs are counted sequentially to identify the level of the spinal pathology. A long, blunt tipped needle is inserted into the disk space, and a radiograph is obtained to localize the level of pathology intraoperatively (Fig. 5). We prefer fluoroscopy to radiography because it reduces the time needed to identify the level of the pathology. Anteroposterior (AP) images are used rather than lateral images. AP views allow the surgeon to count the ribs and vertebrae precisely to confirm their localization.
Pleural and Paraspinal Tissue Dissection
Endoscopic Debakey forceps are used to grasp and elevate the parietal pleura from the surface of the spine and ribs. The pleura is dissected sharply from the surface of the spine and ribs with microscissors (i.e., endoshears or endoscissors). The segmental vertebral vessels are preserved as they pass over the surface of the middle of the vertebral bodies. For an anterior release or an anterior thoracic diskectomy, the segmental vessels usually do not need to be ligated.
For vertebrectomies, however, the segmental artery and vein should be ligated with hemoclips and divided sharply. Hemoclips are used rather than just coagulation because these segmental vessels are direct branches from the great vessels. Right-angle dissection forceps and hemoclips are used to mobilize and ligate the segmental vessels before dividing them with scissors.
If the exposure is on the left side in the lower thoracic region, the surgeon should consider identifying the artery of Adamkiewicz via preoperative spinal angiography (Fig. 6). The pleural dissection remains localized over the levels of the spinal pathology and laterally along the proximal 2 to 3 cm of the corresponding ribs. The sympathetic chain should be preserved. It can be mobilized laterally over the surfaces of the ribs as needed.
Standard spinal tools have been modified and existing laparoscopic and thoracoscopic soft tissue dissection instruments have been used to facilitate thoracoscopic spinal surgery. All of the tools for thoracoscopic surgery need to be longer than their counterparts for conventional spinal surgery because the working distance from the chest wall to the spine ranges from 14 to 30 cm.
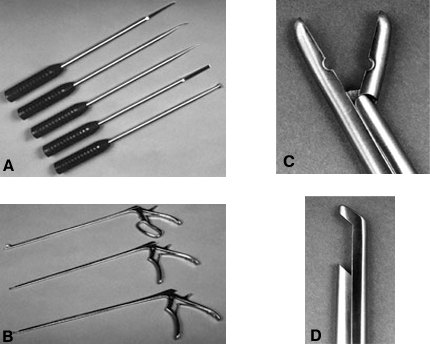
Laparoscopic and thoracoscopic soft tissue dissection tools that are available commercially are suitable for a variety of tasks. Several basic tools that are useful for spinal paraspinal soft tissue dissection include microscissors (endoshears), fine tissue clamps (Debakey forceps), Babcock clamps, Allis clamps, right-angle tissue forceps, suction irrigation tools, and peanut dissectors (Fig. 7).
Long thoracoscopic bone and disk dissection tools include curved and straight curettes, Penfield instrumentation, nerve hooks, Cobb periosteal elevators, rib dissectors, Kerrison rongeurs, Leksell rongeurs, disk space rongeurs, osteotomes, and bone graft impactors (Fig. 8).
Long drill bits are needed for the bone dissection of the vertebrae and ribs (Fig. 9). Midas Rex (Midas Rex™ Pneumatic Tools, Fort Worth, TX) provides drill bits that are 25 cm and 40 cm long: the RX bit, the L bit, and R attachments.These attachments have a long protective sheath that prevents injury to the lung and paraspinal soft tissues. A pistol grip handle is attached to the proximal portion of the drill to stabilize the drill precisely. Three-point control of the drill is achieved by grasping the handpiece and pistol grip with two hands and further stabilizing the shaft of the drill against the chest wall within the portal.
Accurate, fine control of the tip of the drill is provided using these maneuvers. A high speed drill is essential to permit accurate dissection. Drill bits are available in several shapes for bone dissection. Cone-shaped bits are used for fine dissection (-8 bit), ball-shaped bits for cutting large bone surfaces (-31 and -32 bits), and cylinder-shaped bits for cutting smooth, wide bone channels (-11, -12 bits)
Hemostatic Agents
Hemostasis can be achieved by a variety of methods that are similar or identical to hemostatic techniques used in conventional surgery (Fig. 10). Vascular clips or sutures, bipolar cauterization, unipolar cauterization, cotton-tipped peanut dissectors, bone wax, cottonoid patties, Gelfoam®, Endoavetine®, and Nuknit® are used to achieve hemostasis. Straight and right-angled vascular clip appliers are available for ligation of small and medium size blood vessels. Bone bleeding is controlled with small pieces of bone wax that are applied with an endoscopic peanut dissector. Epidural hemostasis is achieved with bipolar coagulation or small pieces of hemostatic agents, which are applied gently with cottonoid patties. The end of the string of the cottonoid should be clamped with a hemostat to prevent losing the cottonoid within the thorax. Monopolar cautery should be used sparingly near the spine and avoided near the dura, nerve roots, and sympathetic chain.
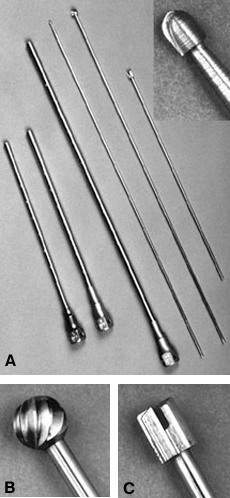
Figure 9. (A) Long drill attachments for bone dissection. (Left to right) The L, the R, and the RX attachments (25-cm, 25-cm and 40-cm long, respectively), and the -8, -31, and -12 bits. (Inset) Close-up of the -8 bit for fine microsurgical bone dissection. (B) Close-ups of the -31 bit for cutting large cavities in the bone, and (C) the cylinder-shaped -12 bit for mortising the fusion bed.
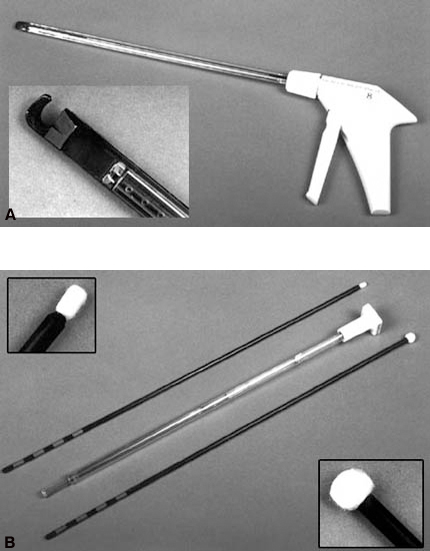
Figure 10. Hemostatic tools. (A) Right-angle clip applier for ligating the segmental vessels and a close-up of its tip (inset). (B) Peanut (top) and cherry (bottom) dissectors can be used to retract tissue, to tamponade bleeding temporarily, and to apply bone wax to bleeding bone. The Endoavitene delivery system is a hollow tube with a plunger (center). (Insets) Close-ups of the tips of the (top) peanut and (bottom) cherry dissectors.
The surgeon always must be prepared to obtain immediate hemostasis if a complication such as injury to a great vessel occurs. A tightly rolled 4 x 4 sponge on a long clamp (sponge stick) and a thoracotomy tray should be available. If a large vessel bleeds, the endoscope should be repositioned from the depths of the operating field with the tip moved superficially near the chest wall so that the lens does not become covered with blood. The sponge stick is used to gently tamponade the bleeding vessel until the chest can be opened. Routine intraoperative hemostasis can be obtained easily with endoscopic techniques; however, it is important to be prepared for an immediate thoracotomy if a major vascular complication occurs.
The 12 thoracic vertebrae have cylindrical, slightly oval vertebral bodies. The size of the thoracic vertebrae increases progressively from the first to the twelfth thoracic level. The height of each vertebral body is slightly shorter than its AP diameter and width. The thoracic disk spaces are relatively narrow, reflecting the limited motion that occurs across the thoracic motion segments.
The middle of the thoracic vertebral body, between each disk space, has a slightly concave surface. The segmental arteries and veins course over the middle of the vertebral bodies. The disk spaces and end plates form a convex surface. The surface contours are important clues for determining the anatomical relationships intraoperatively.
The pedicles connect the vertebral bodies with the remainder of the posterior arch (i.e., transverse processes, pars interarticularis, facets, laminae, and spinous processes) (Fig. 11). The pedicles are dense oval cylinders of bone that have a cancellous center.
The pedicles are adjacent to the upper third of the vertebral body. Understanding the relationship of the pedicle to the disk space, vertebral body and spinal canal is critical for intraoperative anatomical orientation. The pedicles of two adjacent vertebrae form the boundaries of the neural foramen through which the nerve roots traverse. The pedicle therefore surrounds the lateral aspect of the dura over the spinal cord. The nerve roots within the neural foramen are surrounded by a large amount of epidural fat, a rich epidural venous plexus, and radicular arteries. Therefore, the dura is best exposed by removing the thoracic pedicles rather than dissecting through tissue within the neural foramen. The upper surface of the pedicles is contiguous with the superior surface of the vertebral end plates. Consequently, tracing the upper surface of the pedicle anteriorly leads one into the disk space. To adequately expose a disk space for neural decompression, a portion of the pedicle of the caudal vertebrae must be removed to visualize the dura (Fig. 12).
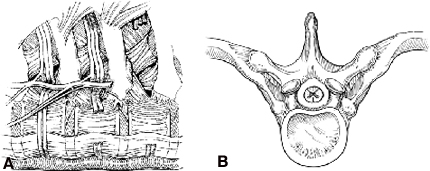
The rib heads provide essential landmarks for localization. The sympathetic ganglia and sympathetic chain are located just lateral to the rib heads beneath the parietal pleura. The ribs articulate with the transverse processes and the pedicles by strong ligamentous attachments. The costotransverse and costovertebral ligaments are dense, thick, and relatively inelastic. The rib forms a triangular space overlying the transverse process, the pedicle, and the vertebral bodies. The rib head, which articulates with the base of the pedicle and the vertebral body just caudal to or at the level of the disk space, serves to orient the surgeon to the relative position of the disk space and the pedicles. The T9 rib leads to the T8-T9 disk space; the T8 rib leads to the T7-T8 disk space, and so on. The costovertebral joint is a shallow ball and socket type joint. The glistening surfaces of the cartilage in this joint are a helpful anatomical feature for verifying that the rib head has been resected completely.
The key to unlocking the spinal canal and visualizing the nerve roots, dura, and spinal cord is the costovertebral triangle created between where the rib joins the transverse process and the vertebral body. The proximal 2 to 3 cm of the rib is removed en bloc to expose the surface of the pedicle. The pedicle is then removed to unlock the lateral aspect of the thecal sac. Removing the pedicle early in the dissection allows the dura to be visualized clearly so the surgeon can remain oriented to the position of the spinal cord to protect it during the dissection.
A predictable anatomical relationship exists among the intercostal vein, artery, and nerve. The segmental artery and vein course over the middle of the concave surface of the vertebral body. At the neural foramen, the segmental nerve joins the segmental vessels. As the neurovascular bundle extends laterally, from cephalad to caudal, the vein, artery, and nerve run in the groove on the undersurface of each rib.
Spinal Operative Techniques
Thoracic Microdiskectomy
The parietal pleura is dissected from the surface of the spine, and if needed the segmental vessels to the region of interest are exposed, isolated, and mobilized or divided. The disk space is exposed at the level of the pathology. The pleura is incised over the medial surface of each rib, and Cobb periosteal elevators are used to expose a 2- to 3-cm segment of the proximal rib and rib head. The muscular attachments and neurovascular bundle are detached from the margins of the rib using subperiosteal dissection with periosteal elevators and large curved curettes. Usually, the neurovascular bundle is detached easily from the undersurface of the rib by careful dissection with curved curettes. Bleeding from the intercostal artery or vein can be controlled with bipolar cauterization. The intercostal nerve is identified and preserved.
The costotransverse and costovertebral ligaments are divided sharply using curved curettes and a Cobb periosteal elevator. The rib head is detached from its articulation with the vertebral body. After detaching the soft tissue attachments from the rib, a Midas Rex® drill with a R-8 bit is used to create an osteotomy to transect the rib. A thin layer of the outer cortex of the rib is preserved. The osteotomy is completed using a sharp osteotome to avoid catching soft tissue in the drill bit. The rib head and the proximal rib are removed en bloc. Enough of the proximal rib should be removed to insure that the neural foramen and pedicle have been exposed satisfactorily. The pedicle is identified and the surfaces of the pedicle are exposed using a periosteal elevator and curved curettes. The epidural fat and nerve root are identified within the neural foramen. Epidural venous bleeding from the neural foramen is common but easily controlled with bipolar coagulation, Gelfoam, or Endoavetine.
The amount of the pedicle that needs to be resected depends on whether the herniated disk material has migrated. If the herniation remains at the level of the disk space, the superior half of the pedicle must be removed. If the disk has migrated caudally, the entire pedicle must be resected. When the pedicle is removed, the lateral aspect of the thecal sac is exposed. This exposure enhances visibility thereby enabling the surgeon to preserve and protect the dura and spinal cord. Typically, the pedicle of the caudal vertebrae adjacent to the disk space must be removed. If the T9-T10 disk is being removed, the T10 pedicle is resected. The pedicle is removed with drills and Kerrison rongeurs.
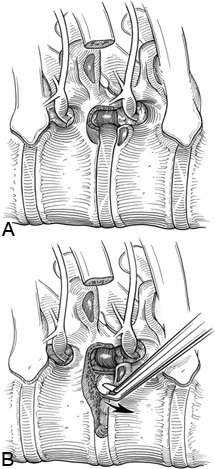
After the pedicle has been removed and the dura has been clearly identified, the diskectomy is performed. The annulus fibrosus of the involved disk space is incised with a microscissors or a Cobb periosteal elevator. Disk curettes and disk rongeurs are used to remove the disk material from the posterior edge of the disk space adjacent to the spinal canal. This dissection creates a cavity in which to curette the herniated disk material, moving it away from the spinal cord.
The vertebral end plates, calcified disk material, or osteophytes may need to be removed with a drill or Kerrison rongeurs. The depth of the decompression is assessed with direct visualization and can be verified with intraoperative radiographs or fluoroscopic images (Fig. 5). The decompression can readily be extended completely across the spinal canal, all the way to the contralateral pedicle (Fig. 13). Interbody fusion is usually not necessary after a routine thoracoscopic diskectomy. However, if fusion is needed, the proximal rib, which was harvested during the spinal exposure, can be used for a bone graft.
Anterior Release of Spinal Deformities
Anterior release is used to treat patients with chronic progressive spinal curvatures such as scoliotic or kyphotic deformities.2 These deformities tend to be longstanding and occur predominantly in young patients. Anterior release refers to the transection of the soft tissue attachments (i.e., annulus, anterior longitudinal ligament, and scar tissue) between vertebrae before corrective forces are applied for reduction with hardware. The tension band or tethering effect of these soft tissues, which would resist the reduction of the deformity, is removed.
For scoliotic deformities, the spine is approached from the convex side of the predominant thoracic curvature. The convex apex of the curved spine is more superficial within the thoracic cavity, and the disk spaces are widened on the convex spinal surface; both features make it desirable to approach this spinal surface. If the spinal curvature is approached on the concave surface, the disk spaces are narrowed and inaccessible, and the spine is farther away from the chest wall. These features make the spinal dissection difficult.
Anterior release consists of a simplified thoracic microdiskectomy at multiple levels (Fig. 14). The major advantage is that the dura does not need to be exposed or decompressed, and the segmental vessels do not usually have to be sacrificed. The involved disk spaces are identified radiographically and visually with the thoracoscope.
The pleura is incised directly over the disk spaces. The segmental vessels are preserved if possible. The rib heads and proximal ribs are not resected unless a severe complex curvature is being corrected, or unless a thoracoplasty is also being performed to treat an accompanying chest wall deformity. The rib heads, however, may be detached by sectioning the costotransverse and costovertebral ligaments to additionally release the attachments to the vertebrae.
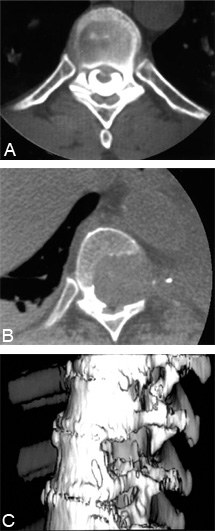
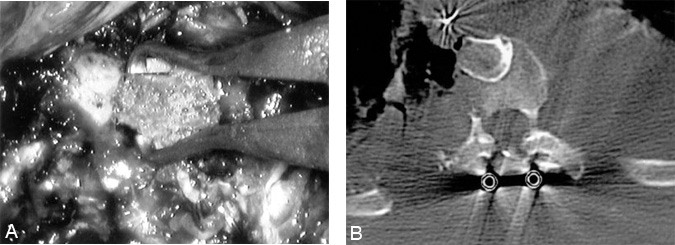
Figure 14. (A) Corticocancellous, autologous iliac crest bone struts are compressed into the disk spaces after the spinal deformity is reduced and fixated. (B) Postoperative computed tomography of an interbody fusion graft after an anterior release and reduction of a chronic kyphotic deformity in a 14-year-old female.
The annulus of each disk space is incised with a Cobb periosteal elevator. Disk material is loosened from the interspaces with Cobb elevators and disk curettes. The disk material is removed with disk rongeurs. The cartilaginous end plates are decorticated with a drill. The ball shaped Midas attachments are useful for this task.
After the intervertebral end plates have been decorticated, morcellized autologous bone grafts (i.e., rib or iliac crest) are placed into the interspaces to achieve a bone fusion. A staged, posterior reduction, internal fixation with a universal hook rod instrumentation system and fusion are performed.
We have found it useful to perform the interbody fusion with solid blocks of bone, using a staged second thoracoscopic approach. The thoracoscopic anterior release is performed. The patient is then repositioned prone and the posterior reduction, fixation, fusion, and harvest of bone graft are performed.
Autologous bone blocks are saved sterilely for the interbody fusion (from rib resection or iliac crest harvest). Immediately after the reduction the patient is repositioned laterally, and the thoracoscopic portals are reinserted into their original incisions. The size and depth of the intervertebral defects are measured. Solid grafts are sized and compressed into the defects with bone graft impactors, wedging the grafts between the adjacent vertebrae.
This strategy is simple. The reinsertion of the thoracoscopic portals and insertion of the grafts can be performed very rapidly, without additional morbidity. The solid bone grafts promote fusion better than the morcellized bone because they are compressed against the adjacent fusion surfaces. The solid grafts also provide structural support: They bear axial loads and can reduce some of the stresses on the spinal instrumentation, thereby reducing the risk of hardware failure.
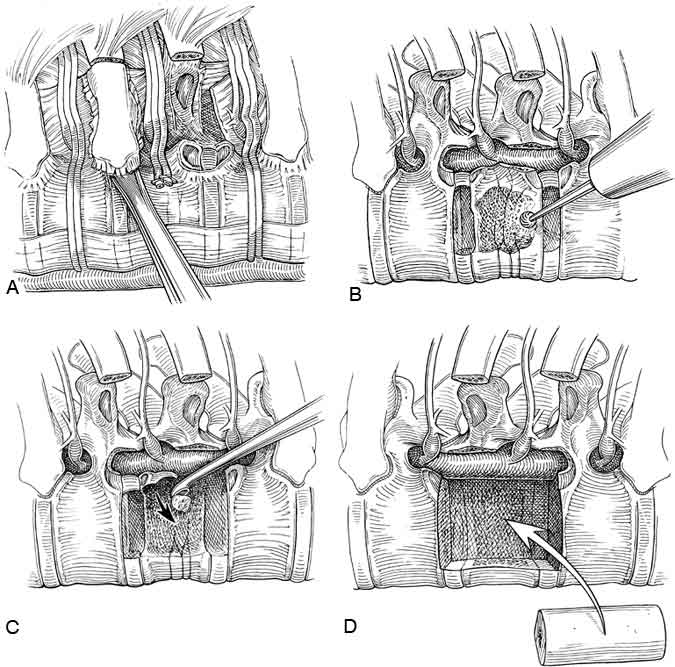
Thoracic Vertebrectomy
Corpectomies are performed in a manner similar to the exposure for diskectomies3 (Fig. 15). The pleura is dissected widely from the surfaces of the involved vertebrae and the adjacent ribs. The segmental vessels are ligated and transected. The proximal rib attachments to each of the respective vertebral levels are identified and the proximal ribs are removed.
The pedicles of the involved vertebrae are removed with a Kerrison rongeur. For protection, the dura and the nerve roots are identified clearly before proceeding with the resection of the vertebral bodies. Diskectomies are performed to define the cephalad and caudal boundaries of the bone dissection.
A large cavity is created within the center of the involved vertebral bodies with a high speed drill, osteotomes, curettes, and bone rongeurs.
The posterior longitudinal ligament and any pathology compressing the spinal cord can be visualized clearly and then decompressed safely by curetting the material away from the spinal cord and into the cavity made in the vertebrae. This sequence of dissection, clearly visualizing the dura throughout the operative procedure, maximizes the safety of the procedure.
Vertebral Reconstruction with Bone Grafts
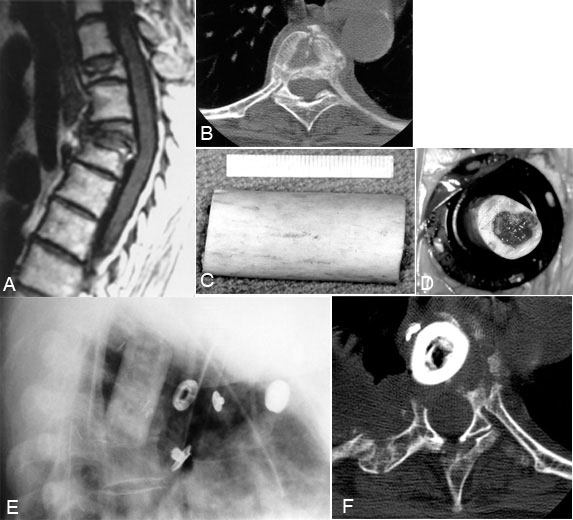
Osseous defects from a corpectomy can be reconstructed using allograft bone shafts, autologous iliac crest struts, or a combination of allograft and autograft (Fig. 16). If allograft is needed, a whole diameter humerus shaft usually fits well within the thoracic vertebrectomy site. The length, width, and depth of the bone graft and the vertebrectomy defect are precisely measured. The bone graft is sized so that it is just slightly longer than the length of the vertebrectomy defect.
One end of the graft is cut with a slightly beveled surface to allow the graft to be wedged into position. The bone graft is inserted, “end-on,” through a 20-mm flexible portal into the thoracic cavity. The bone graft is grasped with an endoscopic Babcock clamp and positioned into the vertebrectomy defect. Bonegraft impactors and mallets are used to compress the bone grafts precisely into the vertebrectomy bed. The relationship of the graft to the dura must be observed throughout the insertion of the graft.
Vertebral Reconstruction with Methylmethacrylate
If tumors have been resected, reconstruction can be performed relatively easily with methylmethacrylate (Fig. 17).4 The dimensions of the vertebrectomy defect are measured. The adjacent normal vertebrae are used to anchor the methylmethacrylate. A sterile silastic tube is cut so that it is 5 to 6 mm longer than the end plates at the margin of the vertebrectomy defect.
The end plates of the adjacent normal vertebrae are penetrated, and large holes are made into the adjacent vertebrae with drills and curettes to fit the tube diameter. The silastic tube, which serves as a template for the methylmethacrylate until it sets, is telescoped into the bodies of the adjacent vertebrae. A hole is cut in the middle of the tube to allow injection of the methylmethacrylate.
A long, wide bore needle with a pressure syringe is used to inject the methylmethacrylate. Slow-setting cranioplasty methylmethacrylate is preferred to rapid-setting methylmethacrylate because it allows the polymer to be injected and it produces much less heat as it sets, thereby minimizing the risk to the spinal cord. The methylmethacrylate is injected until it completely fills the silastic tubing and “toothpastes” through the ends into the adjacent vertebrae.
The silastic tube acts like a mold while the methylmethacrylate hardens. Extrusion of the methylmethacrylate into the adjacent bone is mandatory to provide an anchor to prevent the methylmethacrylate from loosening or becoming displaced. Additional methylmethacrylate can be added ventrally and laterally to the tube; however, care should be taken to ensure that the dura and spinal cord are not compressed.
Internal Fixation of the Thoracic Spine
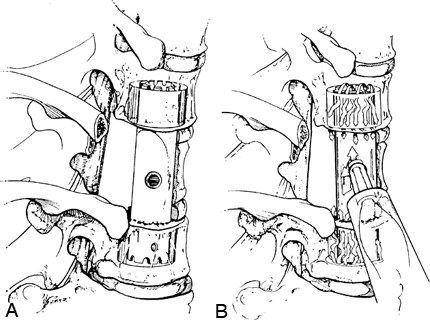
Internal fixation of the reconstructed spinal segments can be performed with screw plates using a thoracoscopic operative approach (Fig. 18). A low profile, dynamic, titanium screw plate is available with long tools that can be used to insert the fixation device percutaneously (Z-plate; Sofamor-Danek, Memphis, TN).10 This locking screw plate has components that make it relatively easy to insert thoracoscopically. The screws and plate are applied to the lateral surfaces of the vertebral bodies positioned away from the great vessels, heart, and mediastinum. The plate is 18-mm wide, 5-mm thick, and is manufactured in graduated 1-cm lengths. The plate easily fits sideways through a 20-mm flexible portal.
Before surgery begins, the portals for insertion of the bolts and screws can be determined by fluoroscopy, which will eliminate the need for additional portal incisions. Intraoperative fluoroscopy and direct visualization with the thoracoscope are required to guide the trajectory of the screws and bolts precisely into the bones.
Plate application is relatively simple. After the corpectomy and decompression of the spinal cord are completed, the dura is visualized directly. The surfaces of the adjacent vertebrae are prepared so that osteophytes and irregular contours of the bone are removed to allow the plate to seat flush against the bone surfaces. The rib heads adjacent to the plate must be removed to permit a flush plate fit.
The preoperative MR imaging or CT is used to measure the length of the bolts and screws, so that they achieve bicortical purchase but do not penetrate more than 1 to 2 mm beyond the opposite vertebral cortical surface. The vertebrae above and below the vertebrectomy are measured individually. Bolts and screws of the appropriate length are selected. The screws should be 5 mm longer than the bolts to compensate for the height of the plate.
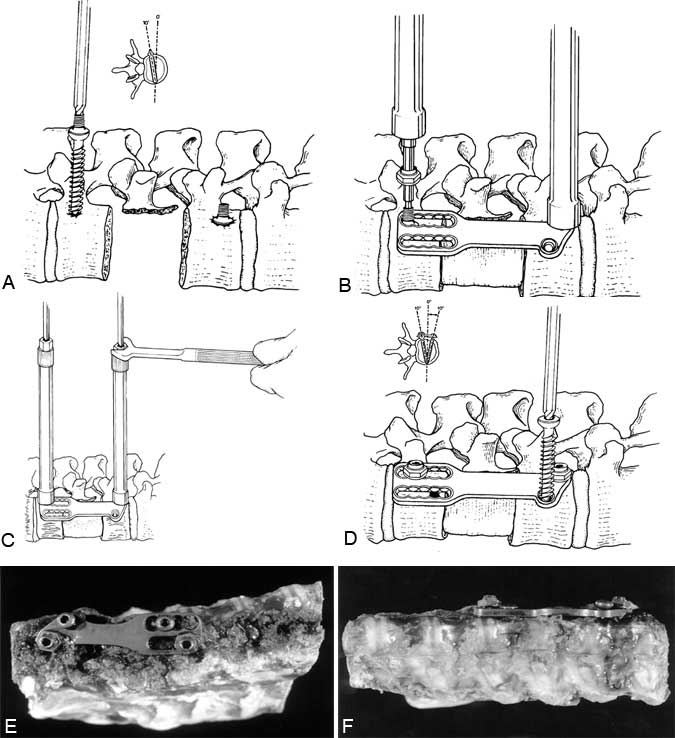
Intraoperative fluoroscopy and direct observation are essential to determine the appropriate trajectory for the bolts and screws. Directly visualizing the dura with the thoracoscope and precisely positioning the patient in a true lateral position allow the surgeon to confirm the exact trajectory of the screws. The bolts and screws are placed parallel to the vertebral end plates on AP fluoroscopic imaging. Transversely, the bolts are directed 10Þ anteriorly and the screws are directed 10Þ posteriorly. The bolts are inserted 1 cm from the posterior rim of the vertebral body and 1 cm from the edge of the distal end plates. A contoured plate guide is used to position a bone awl; the awl is used to create a proximal pilot hole 10 to 15 mm deep in the bone.
The self-tapping bolts are placed parallel to the vertebral end plates and are angled 10Þ away from the spinal canal. The bolts are placed on a hex screwdriver and inserted so that the shoulders of the bolts are positioned flush with the surface of the vertebrae. Before the plate is inserted over the bolts, the vertebral body reconstruction graft is inserted. The plate is inserted, grasped with a plate holder inside the thoracic cavity, and positioned onto the two bolts. The graft can be compressed with a compression tool applied to the wrench screwdrivers over the surface of the chest. Locking nuts are applied to secure the plate to the bolts. The nuts are applied with a hexagonal wrench that is an integrated sleeve on the shaft of the screwdriver. After the nuts are applied to lock the plate to the bolts, screws are placed in the ventral slot and the ventral hole on the plate. The screws are 5 mm longer than the bolts to accommodate the height of the plate. The screws are angled 10Þ toward the spinal canal and are positioned more ventrally and proximally to the vertebrectomy defect compared to the bolts. Unlike the bolts, the screws do not lock to the plate. The screws have a bicortical purchase to anchor them into position.
Wound Closure and Postoperative Management
Upon completion of the dissection, all tissue debris and blood are thoroughly irrigated from the thoracic cavity. The contents of the thoracic cavity are inspected with the thoracoscope. The fan retractors are removed, the surface of the lung is inspected, and the lung is inflated to insure that there is no air leak. Two chest tubes are placed through different, preexisting portal incisions while the insertion is visualized directly with the thoracoscope to ensure that the chest tubes are positioned properly. An apical chest tube is used to reinflate the lung. A posteroinferior chest tube is used to drain fluid and blood from the thorax postoperatively.
The wounds are closed with a subcuticular skin closure. The chest tubes are placed to 20 cm H2O of suction. The skin entry sites for the chest tubes are sealed with an occlusive dressing. Postoperative chest and spine radiographs are obtained to ensure that no postoperative complications have occurred.
Contraindications
Contraindications to a thoracoscopic approach may consist of medical reasons that would prohibit surgery (i.e., uncontrollable coagulopathy, irrecoverable terminal illness, severe cardiac or pulmonary disease). Preoperatively, patients should be evaluated by a pulmonologist or an internist. Patients must have enough pulmonary reserve to tolerate temporary ventilation of one lung during surgery. Preoperatively, spirometry, blood gases, and pulmonary function studies are needed. Patients with conditions such as prior chest trauma, a prior thoracotomy, emphysema, or a hemothorax may have extensive adhesions that can prohibit thoracoscopic access. Extensive scar tissue from a previous operation at the site of the spinal pathology also could preclude thoracoscopy.
When thoracoscopy is not possible, other operative options include thoracotomy, sternotomy, or costotransversectomy, depending on the location of the pathology.
The thoracoscopic approach is only able to access the anterior and anterolateral vertebrae (i.e., the vertebral bodies, ipsilateral pedicles, and transverse processes). This exposure can access neither the posterior spinal elements (i.e., the laminae, spinous processes, facets) nor the contralateral pedicle and transverse process.
The biomechanical advantages achieved with anterior reconstruction of the loadbearing capabilities of the spine are significant. However, multisegmental vertebral fixation and a strong leverage effect, characteristics of posterior hook-rod instrumentation systems, cannot yet be obtained with this thoracoscopic approach. The multisegmental torsional, distractive, and compressive loads that are required during the reduction of severe spinal deformities mandate that fixation be achieved from a posterior approach for complex spinal deformities.
Complications
Potential intraoperative complications are related to the anatomical dissection performed. Naturally, any of the contents of the mediastinum or thorax are at risk during thoracoscopy, thoracotomy, or posterolateral approaches to the spine. Cardiac arrhythmias are prevented by avoiding monopolar cauterization near the heart. Pulmonary lacerations are avoided by minimizing or avoiding lung retraction and by using blunt fan retractors carefully. The fan retractors should only be opened or closed under direct visualization with the endoscope after they have been removed from the surface of the lung. Neurological and spinal complications are prevented by meticulous dissection techniques under direct visualization. The spinal cord and nerve roots can be protected by prioritizing the visualization of the neural structures during the spinal exposure.
Potential postoperative complications include pneumothorax, hemothorax, chylothorax, atelectasis, pneumonia, neurological injury, intercostal neuralgia, infection, spinal instability, and hardware- and fixation-related complications among others. Post-operative atelectasis and pneumonia can be minimized by temporarily reinflating the lung intraoperatively. The lung is ventilated for 10 minutes every 2 hours of surgical time. Postoperatively, patients are placed routinely on aggressive pulmonary physiotherapy regimens. If no intraoperative pulmonary complications occur, patients usually can be extubated immediately after surgery. The chest tubes can be removed if there is no air leak and when fluid drainage diminishes.
Conclusion
Thoracoscopic techniques provide an advantageous way to perform extensive spinal decompression and reconstruction procedures, yet minimize the disruption of the superficial soft tissues. Previously, the operative procedures for anterior thoracic spinal pathology have been performed exclusively through large incisions. The basic anatomy and the dissection techniques are familiar to most spine surgeons. However, significant adaptations of the surgical techniques using longer tools, restricted access, and new methods of perceiving, visualizing, illuminating, and magnifying the operative site create distinct technical challenges. Technological improvements in endoscopic visualization capabilities and in the development of new tools have facilitated the application of this technique to the treatment of a broad spectrum of spinal disorders.
Neural decompression, interbody fusion, vertebral body reconstruction, and internal fixation can be achieved using these minimally incisional thoracoscopic techniques. Thoracoscopic spinal surgery has tremendous potential for improving patient comfort, improving cosmetic outcomes, and for shortening recovery time. The ultimate analysis of benefit, whether clinical or economic, needs to be addressed by a prospective outcome study. Such a study has already been initiated by the authors in collaboration with spinal surgeons throughout the country.
Adapted from Dickman et al: Thoracic vertebrectomy and reconstruction using a microsurgical thoracoscopic approach. Neurosurgery 38(2): 279-293, 1996. With permission.
References
- Coltharp WH, Arnold JH, Alford WC Jr, et al: Videothoracoscopy: Improved technique and expanded indications. Ann Thorac Surg 53:776-779, 1992
- Dickman CA, Mican CA: Multilevel anterior thoracic discectomies and anterior interbody fusion using a microsurgical thoracoscopic approach. J Neurosurg 84: 104-109, 1996.
- Dickman CA, Rosenthal D, Karahalios DG, et al: Thoracic vertebrectomy and reconstruction using a microsurgical thoracoscopic approach. Neurosurgery 38(2): 279-293, 1996.
- Errico TJ, Cooper PR: A new method of thoracic and lumbar body replacement for spinal tumors: Technical note. Neurosurgery 32:678-681, 1993
- Kaiser LR: Video-assisted thoracic surgery. Current state of the art. Ann Surg 220:720-734, 1994
- Landreneau RJ, Mack MJ, Hazelrigg SR, et al: Video-assisted thoracic surgery: Basic technical concepts and intercostal approach strategies. Ann Thorac Surg 54:800-807, 1992
- Mack MJ, Aronoff RJ, Acuff TE, et al: Present role of thoracoscopy in the diagnosis and treatment of diseases of the chest. Ann Thorac Surg 54:403-409, 1992
- Mack MJ, Regan JJ, Bobechko WP, et al: Application of thoracoscopy for diseases of the spine. Ann Thorac Surg 56:736-738, 1993
- Williams MP, Cherryman GR, Husband JE: Significance of thoracic disk herniation demonstrated by MR imaging. J Comput Assist Tomogr 13:211-214, 1989
- Zdeblick TA: Z-plate anterior thoracolumbar instrumentation, in Hitchon PW, Traynelis VC, Rengachary SS (eds): Techniques in Spinal Fusion and Stabilization. New York: Thieme Medical, 1995, pp 279-289
