
The Infratemporal Fossa: An Anatomic Review
A. Giancarlo Vishteh, MD†
Ernesto Coscarella, MD
Nicholas Theodore, MD
Robert F. Spetzler, MD
Joseph M. Zabramski, MD
Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Current Address: †Quantum Neurological Surgery, Phoenix, Arizona
Abstract
The infratemporal fossa is an anatomic space of great importance to neurological surgeons specializing in skull base surgery. It is a significant crossroads for neurovascular structures that traverse to and from the brain and brain stem. Eight cadaveric sides were dissected to study this regional and adjacent anatomy, which is reviewed here.
Key Words: infratemporal fossa, neuroanatomy, skull base approaches
Multiple neural and vascular structures enter and exit the infratemporal fossa via foramina in the skull base. Knowledge of these calrelationships is extremely important to neurological surgeons, neuro-otologists, craniofacial, and head and neck surgeons.
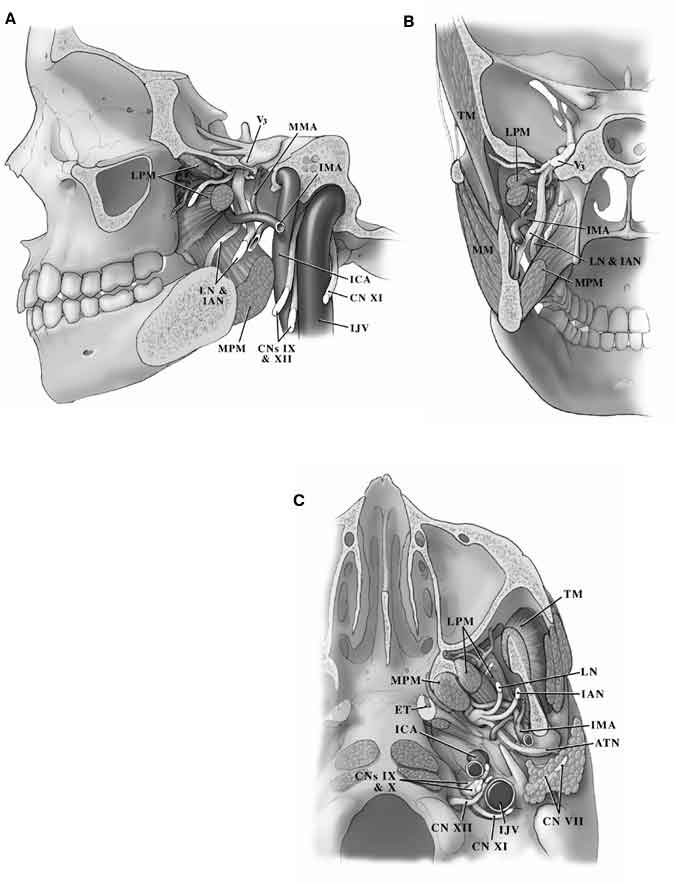
Boundaries of the Infratemporal Fossa
Although controversy exists about the exact boundaries of the infratemporal fossa, we define it as the area bounded anteriorly by the posterolateral aspect of the maxilla and the inferior orbital fissure (Fig. 1).1,3-6,9 The posterior boundary is formed by the tympanic and mastoid portions of the temporal bone. The squamous portion of the temporal bone and the greater wing of the sphenoid form the roof of the infratemporal fossa while the medial boundary is formed by the pterygoid process of the sphenoid bone, the lower surface of the petrous apex, and the lateral portion of the clivus. The lateral boundary of this space is formed by the zygomatic arch and the body of the mandible. The inferior boundary of the infratemporal fossa is along the insertion of the medial pterygoid muscle.
Cadaveric Dissection
The complex relationships among the structures of the infratemporal fossa are reviewed pictorially through detailed cadaveric dissections. The text is organized around the muscles, nerves, and vessels encountered in the region. The cadaveric dissections, however, proceed from superficial to deep. The dissections do not entirely correspond to current surgical approaches to this region; rather, they are intended to demonstrate the anatomy.
The infratemporal fossa is usually exposed through either a retro- or preauricular incision. Ultimately, the incision extends inferiorly to the medial border of the sternocleidomastoid muscle to provide vascular control of the internal carotid artery (ICA), external carotid artery (ECA), and internal jugular vein (Fig. 2).
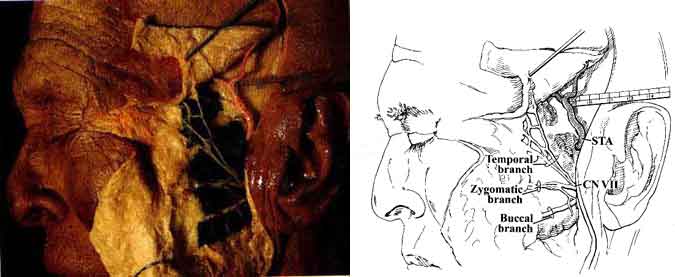
In our dissection, the parotid gland was dissected in the subcutaneous plane. The superficial vessels and their relationship to the frontal branch of the facial nerve (CN VII) then became visible (Fig. 3).
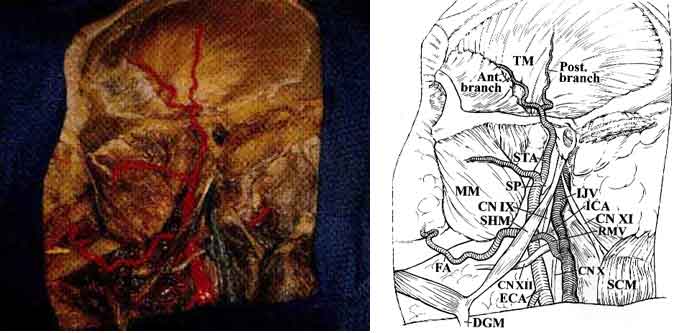
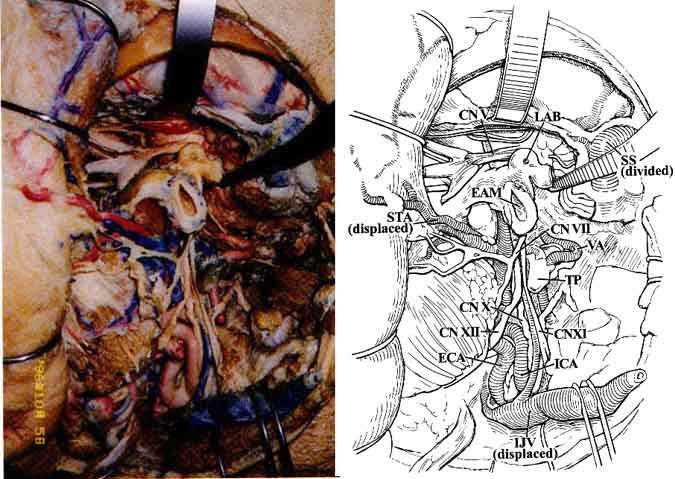
As the anatomic dissection proceeded deeper, the deep portion of CN VII (at its exit point from the stylomastoid foramen) and its relationship to the jugular vein and carotid artery became visible (Fig. 4).
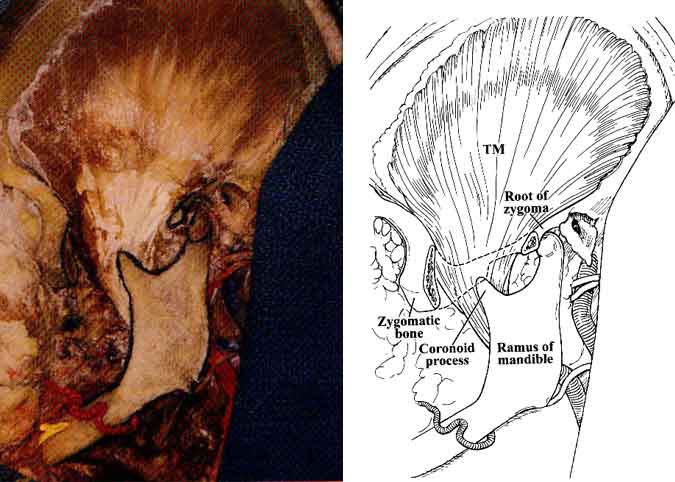
The temporalis fascia was incised behind the frontal division of CN VII, a maneuver performed during surgery to avoid injury to the nerve. The zygoma was exposed and resected from its root to the frontal process. Next, the temporalis muscle became visible from its origin to its insertion points along the coronoid process of the mandible (Fig. 5).
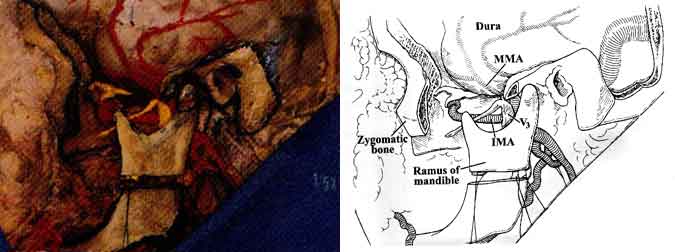
The mandibular condyle was dislocated from the glenoid fossa, and osteotomies were performed along the ramus of the mandible to provide final entry into the infratemporal fossa (Figs. 6 and 7).
Muscle Relationships
The temporalis muscle, along with its deep temporal vessels, passes beneath the zygomatic arch and attaches to the coronoid process of the mandible (Fig. 5).1,3-6,9 The temporalis muscle has two fasciae. The superficial layer attaches to the zygomatic arch superficially while the deep layer attaches to the undersurface of the zygomatic arch.
The masseter muscle consists of two superimposed layers. The superficial layer is attached to the zygomatic process of the maxilla and the lower border of the zygomatic arch. It extends to the temporomandibular joint and inserts inferiorly to the angle of the mandible (Fig. 3). The deep layer of the masseter muscle originates from the medial aspect of the entire zygomatic arch and similarly inserts into the ramus of the mandible inferiorly. The masseter muscle is an important landmark because the parotid gland, parotid duct, and individual branches of the facial nerve are located superficial to it.
After perforating the stylohyoid muscle, the digastric muscle inserts onto the hyoid bone. The anterior belly of the digastric muscle is innervated by the trigeminal nerve (CN V), and the posterior belly is innervated by CN VII. The posterior belly originates in the digastric groove of the mastoid bone, just lateral to the occipital groove.
The upper (infratemporal) head of the lateral pterygoid muscle arises from the infratemporal planum and spine of the greater wing of the sphenoid, and the lower head arises from the lateral pterygoid plate (Fig. 1). Both heads insert onto the neck of the mandible and the anterior aspect of the capsule and disc of the temporomandibular joint. The medial pterygoid muscle originates from the medial pterygoid plate, the pyramidal process of the palatine bone, and the tuberosity of the maxilla. This muscle inserts along the posterior and inferior portions of the medial aspect of the ramus and angle of the mandible. In patients harboring large trigeminal schwannomas, the pterygoid muscles may atrophy and cause the jaw to deviate.
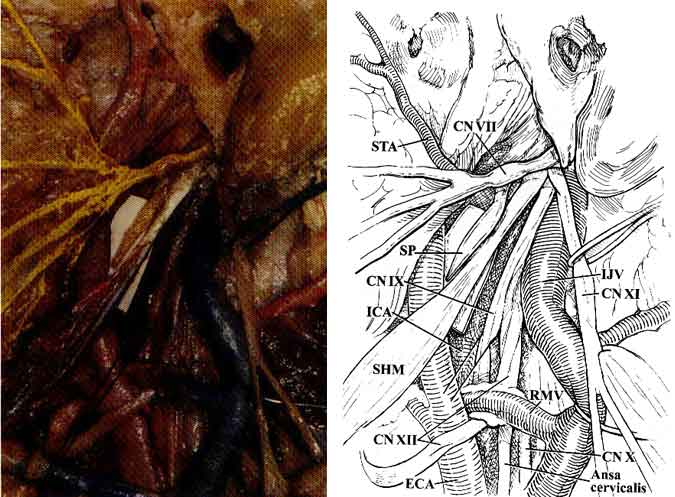
The styloid process is a bony spike emanating from the tympanic portion of the temporal bone and serves as the origin of the styloglossus, the stylohyoid, and the stylopharyngeal muscles (Fig. 4). When these muscles are dissected from the styloid process, the high cervical portion of the ICA is exposed just before it enters the carotid canal. Because of its proximity to the ICA, the styloid process is thought to be the cause of high cervical ICA dissections after blunt trauma or violent and sudden head motions. Because of its proximity to the extracranial portion of the lower cranial nerves and the phrenic nerve, the styloid process can also injure these neural structures.
The tensor veli palatini muscles arise from the scaphoid fossa of the pterygoid process, medial to the foramen ovale, and from the spine of the sphenoid. They merge with the lateral aspect of the cartilage of the eustachian tube. Their point of insertion is along the palatine aponeurosis and the horizontal plate of the palatine bone. The eustachian tube, which is involved in maintaining pressure equilibrium, connects the nasal portion of the pharynx with the tympanic cavity.
Cranial Nerves
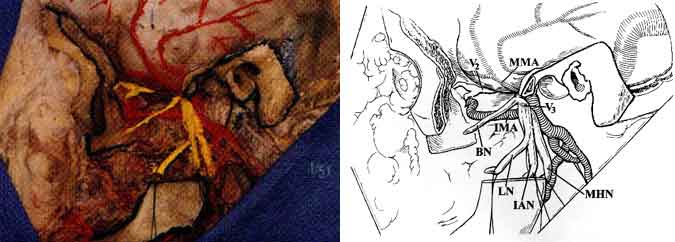
CN VII 1,3-6,9 exits the skull through the stylomastoid foramen and immediately divides into the temporofacial and cervicofacial divisions in the substance of the parotid gland. These branches further subdivide into the temporal, zygomatic, buccal, marginal mandibular, and cervical rami (Figs. 2 and 4). Temporal and zygomatic branches cross the zygomatic arch, coursing over the superficial fascia of the temporalis muscle. The chorda tympani nerve arises from the mastoid portion of CN VII, runs superiorly into the tympanic cavity, and passes the medial aspect of the handle of the malleus. It continues anteriorly and enters the chorda tympani canaliculus along the medial aspect of the petrotympanic fissure. It then descends vertically just medial to the sphenoid spine and lateral pterygoid muscle to join the lingual nerve.
The mandibular branch (V3) of CN V passes through the foramen ovale at the base of the greater wing of the sphenoid bone just anterior and medial to the foramen spinosum. After leaving the foramen, V3 lies between the tensor veli palatini muscle medially and the pterygoid muscle laterally (Figs. 1 and 7). A meningeal branch of V3 enters the skull through the foramen spinosum. The buccal, inferior alveolar, lingual, and auriculotemporal branches also arise from V3. The mylohyoid nerve, another important branch of V3, travels with the inferior alveolar nerve and branches from it before the inferior alveolar nerve enters the mandibular canal. It ultimately innervates the anterior belly of the digastric muscle as well as the mylohyoid muscle. The maxillary nerve (V2) exits through the foramen rotundum and proceeds anteromedially to the inferior orbital fissure and reaches the floor of the orbit where it continues as the infraorbital nerve.
The glossopharyngeal nerve (CN IX) exits the skull through the pars nervosa of the jugular foramen, anterior to the vagus (CN X) and accessory nerves (CN XI). CN IX then continues caudally and medially to the styloid process in proximity to the lateral surface of the carotid artery just before the ICA enters the carotid canal. CN X leaves the skull through the anterior medial edge of the pars venosa of the jugular foramen and courses deep within the carotid sheath between the carotid artery and the jugular vein. CN XI exits through the pars venosa of the jugular foramen, coursing posterior and lateral to the jugular vein and medial to the styloid process and posterior belly of the digastric muscle to innervate the sternocleidomastoid muscle (Fig. 4).
The hypoglossal nerve (CN XII) exits the hypoglossal canal and lies deep to the jugular vein and the nerves emerging from the jugular foramen. Subsequently, it courses caudally between the carotid artery and the jugular vein. It becomes more superficial at the level of the angle of the jaw where it crosses the ICA and ECA just above the bifurcation of the common carotid artery (CCA) to innervate the muscles of the tongue (Fig. 4).
Arteries
The CCA bifurcates into the ICA and ECA at the upper border of the thyroid cartilage.1,3-6,9 Initially, the ICA ascends fairly superficially to the carotid triangle of the neck, but it assumes a much deeper position after passing medial to the posterior belly of the digastric muscle. Below the level of digastric muscle, CN XII, the ansa cervicalis, and the lingual and facial veins cross the ICA. At the level of the digastric muscle, the stylohyoid muscle and the occipital auricular arteries course across the ICA. Superior to the digastric muscle, the ICA is separated from the ECA by the styloid process and the muscles that originate from it (Fig. 4). At its entrance to the carotid canal, the ICA is invested by a dense sheet of connective tissue. It is separated from the internal jugular vein by CN XII and the other nerves that exit the jugular foramen. The ICA has no branches in the neck.
Initially, the ECA lies anterior and medial to the ICA and carotid triangle of the neck. At this level, CN XII as well as the facial and lingual veins cross the ECA. The artery then ascends on the posteromedial margin of the parotid gland, medial to the digastric and stylohyoid muscles (Fig. 3). The internal maxillary artery arises from the ECA deep to the neck of the mandible. It courses anteriorly between the neck of the mandible and the sphenomandibular ligament, along the inferior border of the lateral pterygoid muscle. It then courses obliquely to the pterygopalatine fossa (Figs. 1 and 7). The internal maxillary artery gives rise to the middle meningeal artery, which in turn passes between the lateral pterygoid and tensor veli palatini muscles to enter the skull base through the foramen spinosum of the sphenoid bone.
The superficial temporal artery (STA) arises from the ECA along the substance of the parotid gland above the neck of the mandible. The temporal and zygomatic branches of CN VII cross the STA at this level (Fig. 4). The STA ascends over the posterior root of the zygoma and divides into anterior and posterior branches that course with the superficial temporal veins and the auriculotemporal nerve over the superficial layer of the fascia of the temporalis muscle (Fig. 3).
Veins
The internal jugular vein reaches the infratemporal fossa via the jugular foramen.1,3-6,9 Along with drainage from the inferior petrosal sinus, the internal jugular vein receives multiple tributaries from the face (transverse facial, lingual, retromandibular veins), head, and neck (external jugular vein).
The pterygoid plexus consists of multiple venous channels situated in the region of the lateral and medial pterygoid muscles. The plexus is connected to the cavernous sinus through veins that traverse the foramen ovale, the sphenoid emissary foramen, and the venous plexus that courses inside the carotid canal and surrounds the ICA.
Surgical Approaches
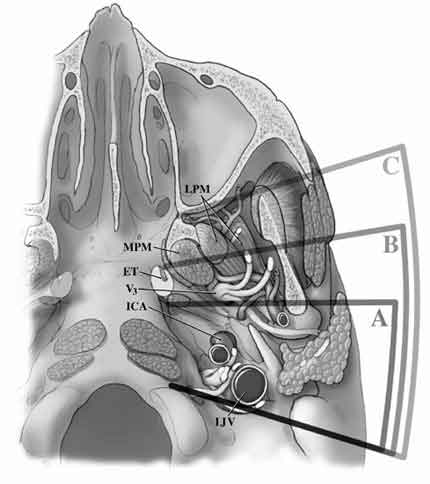
Surgical approaches to the region of the infratemporal fossa were developed for managing not only tumors arising directly from this fossa but also those extending to this locale.2-8 Such tumors included skull base adenocarcinomas, nasopharyngeal carcinomas, adenoid cystic carcinomas, glomus jugulare tumors, and large intra- and extracranial trigeminal schwannomas, among others. With the emergence of multidisciplinary skull base teams composed of neurosurgeons, neuro-otologists, head and neck surgeons, and craniofacial surgeons, a number of these lesions can now be resected more safely and efficaciously than ever before. The division of infratemporal approaches into subtypes is based on the work of Fisch et al.,3,4,6 who classified infratemporal fossa approaches as Type A, Type B, and Type C (Fig. 8).
The Type A approach is suitable for lesions of the jugular foramen and of the posterior infratemporal and mandibular fossae.3,4,6 This approach is an extension of traditional jugular foramen approaches (Fig. 9) but offers additional exposure of the vertical portion of the petrous ICA. Similar to a transcochlear approach, the auditory canal is transected and sewn over, the contents of the middle ear are removed, and the facial nerve is drilled out of its canal and rerouted. However, most of the mastoidectomy, which is intended to provide access to and vascular control of the sigmoid sinus and petrous ICA, can be achieved through an infralabyrinthine exposure or by partially drilling the semicircular canals.
In the Type B approach, a zygomatic osteotomy permits more extensive drilling of the horizontal portion of the petrous ICA so that the petrous apex and clivus can be approached extradurally.3,4,6 This approach requires resection of the glenoid fossa, section of the V3 branch of the trigeminal nerve, and sacrifice of the middle meningeal artery. CN VII need not be rerouted completely, but tethering points at the level of the greater superficial petrosal nerve (GSPN) may require its sacrifice to avoid traction that could cause paresis of CN VII.6,8 Although suited for a number of deeply situated, purely intrapetrous lesions, extradural or combination intra- and extradural (neurosurgical) approaches to this region (i.e., petrous apex and clivus) are much less destructive.
The Type C approach is an anterior extension of the Type B approach.3,4,6 It is useful for gaining exposure of the more medial infratemporal fossa, pterygopalatine fossa, and nasopharynx. Yet more aggressive bony resection along the horizontal segment of the petrous ICA exposes the preclival region and sphenoid sinus.3,4,6
Minor variations have been described for tailoring each of these three approaches to individual lesions. The defect created with some of these approaches may be quite large and may require reconstruction with a free muscle flap. Overall, knowledge of these approaches increases the available options when neurosurgical skull base surgeons plan to resect skull base lesions with or without extensions into the infratemporal fossa.
References
- Bejjani GK, Sullivan B, Salas-Lopez E, et al: Surgical anatomy of the infratemporal fossa: The styloid diaphragm revisited. Neurosurgery 43:842-853, 1998
- Brackmann DE: The facial nerve in the infratemporal fossa approach. Otolaryngol Head Neck Surg 97:15-17, 1987
- Fisch U: Infratemporal fossa approach for glomus tumors of the temporal bone. Ann Otol Rhinol Laryngol 91:474, 1982
- Fisch U, Mattox D: Microsurgery of the Skull Base. New York, Thieme: 1988
- Fournier HD, Mercier P, Velut S, et al: Surgical anatomy and dissection of the petrous and peripetrous area. Anatomic basis of the lateral approaches to the skull base. Surg Radiol Anat 16:143-148, 1994
- Jackler RK: Infratemporal Fossa Approaches. St. Louis, MO, Mosby: 1996
- Leonetti JP, Brackmann DE, Prass R: Improved preservation of facial nerve function in the infratemporal approach to the skull base. Otolaryngol Head Neck Surg 101:74-78, 1989
- Martin C, Prades JM: Removal of selected infralabyrinthine lesions without facial nerve mobilization. Skull Base Surg 2:220-226, 1992
- Warfel JH: The Head, Neck, and Trunk. Philadelphia: Lea & Febiger, 1985
