Cranial Dural Arteriovenous Malformations
Paul W. Detwiler, MS, MD,
Cesar de Paula Lucas, MD†
Joseph M. Zabramski, MD
Cameron G. McDougall, MD
Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
†Division of Neurological Surgery, Hospital Orthopedico de Goiania, Goiania, Brazil
Abstract
Dural arteriovenous malformations represent 10 to 15% of intracranial vascular malformations. They are characterized by the presence of an abnormal arteriovenous fistula or nidus located in the dura. Common nidus locations include the transverse, sigmoid, and cavernous sinuses as well as the falx and floor of the frontal fossa. The natural history of these lesions varies with location and pattern of venous drainage. Certain patterns of venous drainage, especially retrograde leptomeningeal venous drainage, are associated with a high risk of hemorrhage. Therapeutic options for the management of these lesions include transarterial and/or transvenous embolization, direct surgical obliteration, or a combination of the two. This review examines the natural history of these lesions, the indications for treatment, and the selection of approaches for their management.
Key Words: arteriovenous malformation, dural fistula, embolization, resection
Intracranial dural arteriovenous malformations (DAVMs) consist of an abnormal arteriovenous fistula or nidus within the dura. They occur about one-tenth as frequently as their intraparenchymal counterparts. Most DAVMs behave benignly, occasionally even involuting on their own. However, they also can become symptomatic with an aggressive neurological course that includes seizures, focal neurological deficits, and hemorrhage.[5,8,9,17] To manage these lesions effectively, one must be able to identify the risk factors associated with a poor natural history.
The idea that the natural history of these lesions could be predicted by the pattern of venous drainage was first suggested by Houser et al. in 1972.[14] They concluded that intracranial hemorrhage occurred when venous drainage of the lesions was limited to the pial (leptomeningeal) veins. These findings were confirmed and expanded by others.[3-6,8-10,16,19,20] Lesions that drain freely into a dural sinus tend to have a benign course, whereas those with evidence of cortical (leptomeningeal) venous drainage are associated with hemorrhage and neurologic deficits and symptoms.
In certain locations, such as the floor of the anterior cranial fossa and the tentorium cerebelli, DAVMs appear to be associated with an aggressive course. Lesions in these locations, however, are commonly associated with extensive retrograde pial drainage as an independent risk factor. No location is immune to the risk of hemorrhage and neurological deficits. Location only raises the index of suspicion for the presence of dangerous venous anatomy.
Treatment options for the management of DAVMs include endovascular embolization by transarterial or transvenous routes, surgical obliteration, or a combination of these two. Location and the pattern of venous drainage determine the best approach to an individual lesion.[18] The purpose of this review is to examine the natural history of these lesions, their indications for treatment, and the selection of therapeutic options.
Etiology
DAVMs may be either congenital or acquired lesions.[2,7,12,13,15] Patients with a presumably acquired dural AV fistula often have a history of previous trauma, craniotomy, or venous sinus thrombosis.[15]
Two mechanisms have been proposed to account for the formation of acquired dural AV fistulae. The first is based on the proximity of dural arteries and veins, which presumably predisposes to direct connections after trauma.[11] The second model assumes that inflammation (e.g., secondary to trauma, infection, surgery, tumor) or spontaneous thrombosis leads to occlusion of a sinus with the opening of preexisting, minute AV fistulae that exist in the walls of the sinus.[2,13,15] This theory has been tested in a rat model. Dural AV fistulae were produced in rats subjected to a combination of venous sinus thrombosis and increased venous sinus pressure.[12]
Diagnosis
Despite advances in noninvasive neuroimaging, angiography remains the “gold standard” for the diagnosis of DAVMs. Evaluation should include selective, bilateral injections of the internal (ICA) and external carotid (ECA) arteries as well as the vertebral artery because lesions are often fed by multiple branches on both sides regardless of anatomic location. The ability to define both the magnitude and direction of blood flow highlights the value of angiography compared to computed tomography or magnetic resonance imaging.
The successful treatment of DAVMs requires a thorough understanding of both arterial and venous anatomy. The location of the lesion and the venous characteristics are the most important features determining the approach to treatment.
Natural History and Classification
The clinical presentation of patients with DAVMs is quite variable. Their course can be benign with spontaneous involution of the fistula or frankly aggressive, manifesting with seizures, focal neurological deficits, or intracranial hemorrhage. The clinical presentation and natural history of these lesions are related to their venous anatomy.
Pulsatile tinnitus is a common initial symptom of DAVMs draining into the transverse and sigmoid sinuses.[3,8] The bruit itself is benign but may be disabling to patients. A significant change in the character of the bruit requires reassessment. A decrease in the intensity of a bruit can reflect involution of the DAVM but can also result from obstruction of the draining dural sinus and diversion of outflow into the leptomeningeal venous system.
Neurological symptoms other than tinnitus can be caused by three physiopathological mechanisms: (1) direct compression of neural structures by a dysplastic vein(s);[8] (2) venous hypertension;[3,8,16] or (3) an intracerebral, subarachnoid, or subdural hemorrhage of ruptured leptomeningeal draining veins.[5,17] Nonspecific headache related to venous hypertension can be the only symptom of a DAVM and should be explored if the patient has a history of cranial trauma or paranasal sinus infection. Ocular pain associated with chemosis (red eye syndrome), with or without pulsatile exophthalmos, suggests a DAVM in the cavernous sinus.[18] Atypical facial pain or trigeminal neuralgia can be caused by direct compression of the respective cranial nerves by dilated venous channels. Seizure activity and hydrocephalus or papilledema are less common clinical manifestations of DAVMs related to the effects of local and generalized venous hypertension, respectively.
A number of classification schemes have been proposed in an attempt to organize DAVMs into various risk and treatment groups. As early as 1973, Aminoff[1] separated these lesions by anatomical location into an anteroinferior and posterosuperior group, reporting that the latter had a more dangerous course. Other authors have expanded this work and confirmed that DAVMs in some locations are more likely to manifest with intracerebral hemorrhage or nonhemorrhagic neurologic deficits.[3,8] Today, the most widely used anatomical schema classifies these lesions into six categories: anterior fossa, superconvexity sagittal sinus, cavernous sinus middle fossa, transverse-sigmoid sinus, tentorium, and foramen magnum. Combining their experience and a meta-analysis of the published literature, Awad and coworkers[3] reported that an aggressive presentation was most likely to occur with tentorial (92% to 97%) and anterior cranial fossa (68% to 88%) lesions, but they emphasized that no site was immune to such behavior. In addition to anatomical location, certain patterns of venous anatomy were also associated with an increased risk of neurologic complications.
Retrograde cortical venous drainage and venous ectasia, first correlated with intracranial hemorrhage by Houser et al.,[14] were confirmed and expanded by others.[3-6,8-10,16,19,20] In 1978, Djindjian and Merland[10] proposed classifying DAVMs into four categories based on venous anatomy. Cognard and colleagues[8] suggested dividing lesions into seven categories, while Borden et al.[4] suggested a simplified classification scheme with three categories (Table 1). A recent review of the experience of Toronto University with DAVMs in 98 consecutive patients reported an excellent correlation (p < 0.0001) between clinical presentation and both the Cognard and Borden classification schemes.[19] Because of its simplicity, we prefer the Borden classification system (Table 1).
Patient Selection and Treatment Strategies
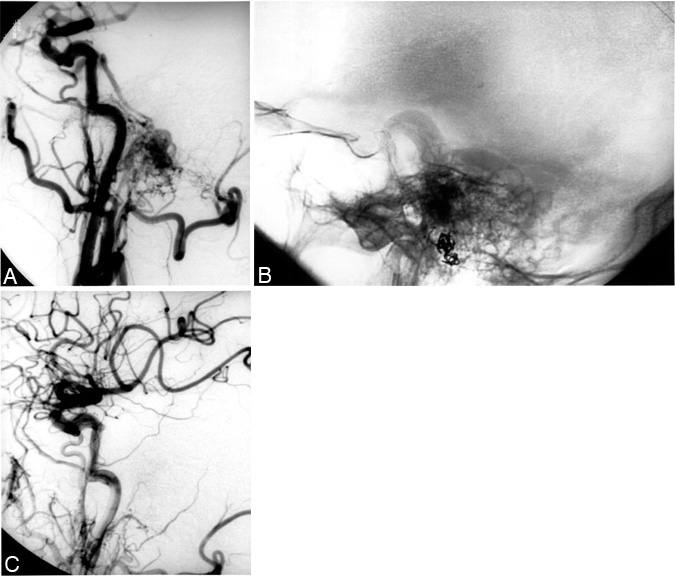
The selection of patients with DAVMs for treatment is based on clinical presentation and knowledge of their natural history. A thorough angiographic assessment is required. Patients who present with a bruit and have DAVMs with no evidence of retrograde venous drainage (Fig. 1) are at little risk of hemorrhage and should be followed. Intolerance for the bruit produced by the lesion occasionally necessitates intervention.
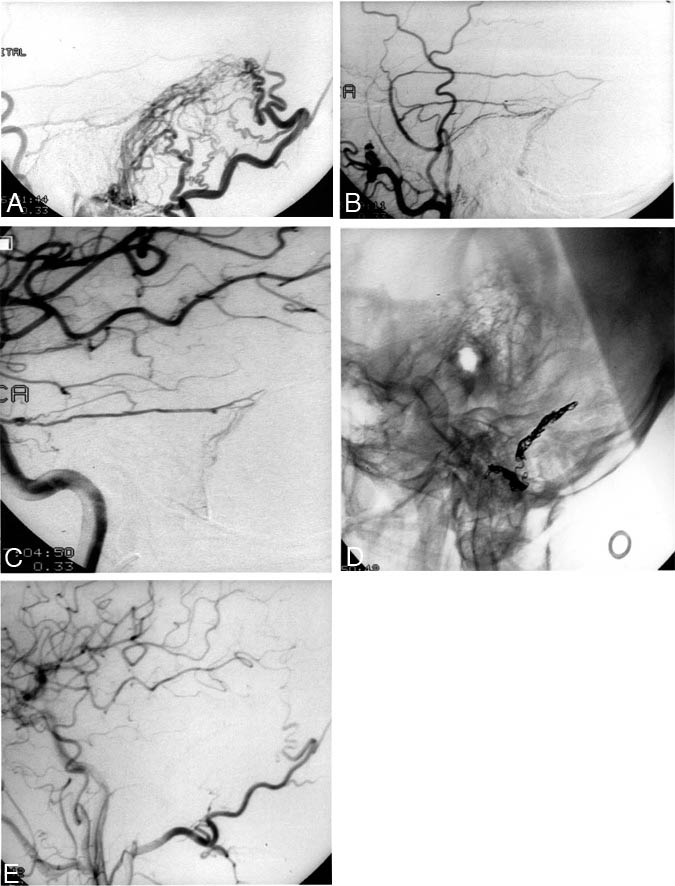
In contrast, DAVMs with retrograde venous drainage are at increased risk of hemorrhage (Figs. 2 and 3). The risk is greatest for lesions with isolated retrograde drainage into the cortical (leptomeningeal) venous system (Fig. 4). In fact, many authors consider such lesions to be neurosurgical emergencies that require urgent intervention because of their high risk of hemorrhage.[3,8,17,18]
[full_width]
Figure 2. A 71-year-old man presented with proptosis and chemosis of the left eye, raising the suspicion of a left cavernous-carotid fistula. Magnetic resonance imaging studies were unrevealing. Angiography demonstrated the presence of a dural arteriovenous (AV) fistula at the level of the foramen magnum adjacent to the marginal sinus. Retrograde venous embolization of the fistulous connection resulted in its elimination. The proptosis and chemosis resolved promptly. (A) A lateral projection of a common carotid artery injection demonstrates an AV shunt at the skull base with retrograde venous filling of the inferior petrosal sinus and anterograde filling to the internal jugular vein. (B) Lateral radiograph of the skull demonstrates the transvenously placed coils at the point of the fistula. (C) Lateral projection of a common carotid artery angiogram reveals no residual AV fistula.
[/full_width]
[full_width]
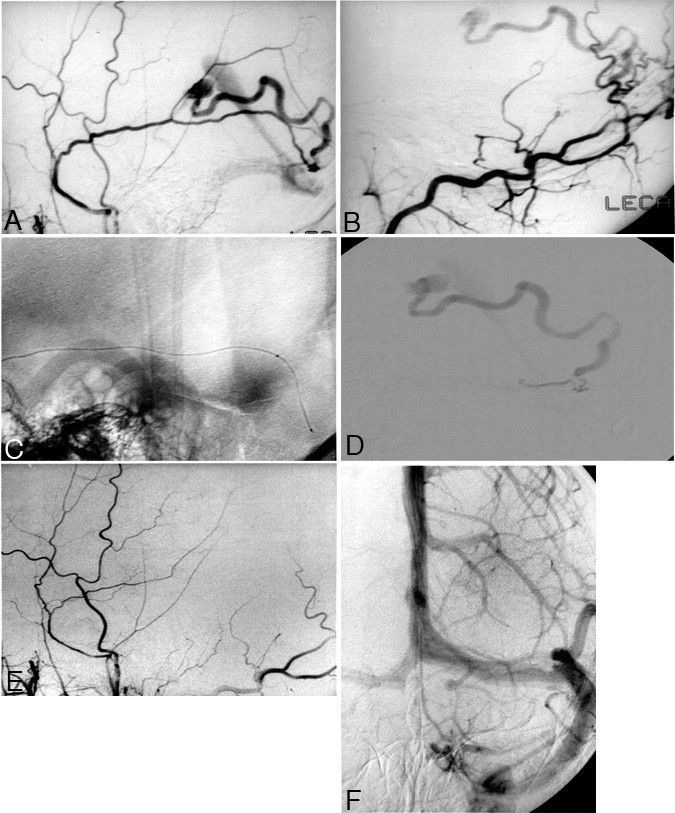
Figure 3. A 31-year-old man presented with headaches. Computed tomography demonstrated abnormal vessels, which were subsequently revealed to be part of the draining vein of a dural arteriovenous fistula. (A) Left-sided external carotid artery angiogram shows the middle meningeal artery with arteriovenous shunting into a tortuous cortical vein that drains ultimately to the straight sinus. (B) Lateral superselective injection into the occipital artery shows additional supply to this fistula through perforating branches of the occipital artery. (C) Scout image showing a microcatheter placed through the left middle meningeal artery to the site of the fistula. (D) Injection of contrast via the microcatheter shows filling of the draining vein with contrast passing through the straight sinus, then through the transverse sinus, demonstrating the location of the fistula adjacent to the wall of the left transverse sinus. (E) After liquid adhesive embolization at the fistula site, injection of the left external carotid artery shows no residual arteriovenous (AV) shunting. (F) A left internal carotid artery injection demonstrates preservation of the left transverse sinus after occlusion of the AV fistula.
[/full_width]
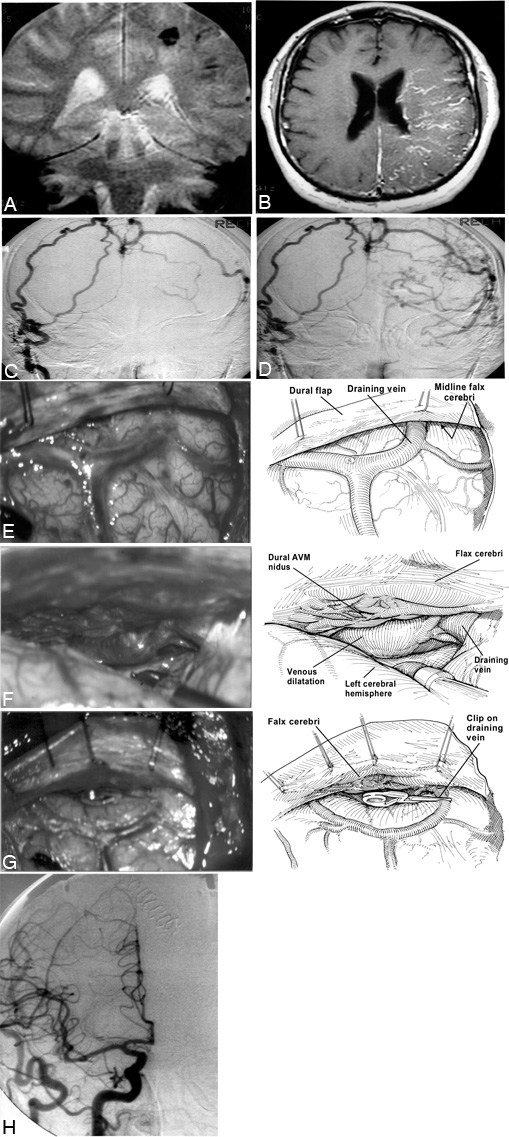
Figure 4. A previously well 63-year-old man presented with intracerebral hemorrhage and seizures. Following direct surgical obliteration of a midline arteriovenous (AV) fistula, the patient recovered completely. (A) Coronal magnetic resonance (MR) image showing a small intracerebral hemorrhage on the left. (B) Contrast-enhanced T1-weighted axial MR image showing the prominent veins of the left hemisphere caused by venous hypertension. (C) A frontal view of a right external carotid artery angiogram demonstrates the midline AV fistula with an early draining vein over the left hemisphere. (D) A later phase of this angiogram demonstrates filling of the retrograde cortical veins shown in Figure B. The patient was taken to the operating room where he underwent a bilateral parietal-occipital craniotomy. The bone flap was extended across the midline to allow exposure of the falx without the need for brain retraction. The dura was opened carefully to avoid injuring the enlarged bridging veins. The nidus of the fistula had been localized during a previous angiogram avoiding the need for stereotactic localization. (E) When the dura was opened, the arterialized bridging vein that drained the fistula was readily visualized. (F) The brain was gently retracted, allowing the nidus of the dural fistula to be visualized. Bipolar coagulation was applied to the surface of the fistula, and the draining bridging vein was occluded with temporary aneurysm clips. After confirming that the patient could tolerate occlusion, the draining vein was coagulated and divided. (G) The leptomeningeal system collapsed and normal venous coloration returned. The patient tolerated the procedure well and developed no new deficits. (H) Angiographic frontal view of right common carotid artery injection confirming there is no residual AV shunting after the fistula was obliterated surgically.
Clinical and angiographic cures, the primary objectives of treatment,[18] can only be achieved by complete obliteration of the lesion including its venous drainage. If obliteration is not achieved, new arterial supplies can be recruited by the fistula, a situation associated with a delayed risk of hemorrhage.[17,18] Incomplete obliteration also carries the risk of a low-grade lesion converting to a higher grade with a concomitant increased risk of hemorrhage.
Because the nidus of a DAVM is contained within the dural folds, the lesion can be attacked not only from the arterial side but also from the venous side without the risk of swelling and hemorrhage associated with venous occlusion in parenchymal AVMs. Dysplastic venous structures remote from the fistula nidus do not require direct treatment; obliteration of the nidus alone will promote thrombosis or the natural involution of these lesions.
The literature offers a variety of treatment strategies for DAVMs, including ligature of the feeding arteries, coagulation and/or excision of the dural nidus, coagulation of venous outflow, transarterial or transvenous embolization, or combinations thereof. To clarify the best treatment for DAVMs, we recently reviewed our experience with these lesions and that reported in the English language literature to determine the success rates of different treatment strategies in obliterating DAVMs.[18] Success was defined as angiographic documentation of complete obliteration. A total of 223 reports contained sufficient information on the treatment of 258 lesions to be included in the analysis. For DAVMs of the transverse-sigmoid sinus (n=64), combined therapy (endovascular plus surgical treatment) proved significantly more effective than either therapy alone (p < 0.01). For lesions of the tentorial incisura (n = 66), combined therapy and surgical obliteration alone proved superior to endovascular embolization (p < 0.001).
DAVMs of the cavernous sinus (n = 67) were treated primarily by endovascular approaches with success rates of 62% to 78% for transarterial and transvenous embolization, respectively. Surgery for cavernous sinus DAVMs was reserved for lesions in which embolization was unsuccessful. In the anterior fossa (n=23), surgical obliteration proved highly effective with a success rate of 95%. The small number of cases in both the superior sagittal sinus (n = 28) and middle fossa (n = 10) regions precluded statistical analysis. Finally, simple ligature of feeding vessels produced success rates of only 0 to 8% and can no longer be recommended.
The results of this review emphasize that there is no ideal treatment for obliterating DAVMs. The management of each case is best individualized. The location of the lesion and the venous anatomy are the deciding factors in whether a surgical, endovascular, or combined approach is most appropriate.
In this regard, the simplest lesions to treat are those with isolated retrograde leptomeningeal venous drainage: Borden Type III, Djindjian Type III, and Cognard Types III and IV DAVMs (Table 1). Effective treatment requires only the obliteration of the draining veins, which eliminates the risk of hemorrhage and leads to thrombotic obliteration of the dural fistula (Fig. 4). Such lesions are most often located in the frontal fossa or along the sagittal sinus, but they can occur throughout the cranium. Success rates for surgical intervention approach 100% for this group of lesions. [18] Endovascular treatment by transarterial routes can reduce blood flow through the lesion but is unlikely to lead to a long-term cure because new arterial collaterals are rapidly recruited.Transarterial embolization followed by surgery or surgery alone is the best approach for lesions whose drainage into the dural sinuses is associated with a variable degree of retrograde drainage into the leptomeningeal venous system (Borden Type II, Cognard Type IIB, and Djindjian Type II). Typically, such lesions are located along the tentorium (Fig. 2). When venous access is available, transvenous occlusion of the involved dural sinus is another alternative for management (Fig. 3). Care must be taken to ensure complete obliteration of the lesion occlusion of the sinus with continued retrograde leptomeningeal venous drainage could increase the risk of hemorrhage, neurological deficits, or both.
Patients with DAVMs that show no evidence of retrograde leptomeningeal venous drainage (Borden Type I, Cognard Type I & IIA, Djindjian Type I) should be considered for observation. Indications for treatment include an intolerable bruit and signs of increased intracranial pressure related to venous hypertension. Transarterial embolization can be used to reduce blood flow and thus to ameliorate or eliminate the bruit. The recruitment of dural collaterals, however, often leads to the recurrence of symptoms. The role of stereotactic radiosurgery in the management of these lesions has yet to be defined, but preliminary reports suggest that it may lead to effective obliteration when the delay associated with this treatment is acceptable. We have treated two patients with Type I lesions and intolerable bruits with a combination of endovascular embolization and the Gamma Knife, but long-term follow-up data are not yet available.
DAVMs that involve the cavernous sinus are considered separately because of their threat to visual function. Retrograde drainage from the cavernous sinus into leptomeningeal veins is rare and thus the risk of hemorrhage is small. These lesions most often become symptomatic with ocular pain and chemosis (red eye syndrome). Pulsatile exophthalmos is also a common manifestation. Transarterial and transvenous embolization of DAVMs that drain into the cavernous sinus is successful in 60% and 80% of cases, respectively. Direct surgical treatment is a rare therapeutic necessity. It is important to establish the correct differential diagnosis between this lesion and a carotid-cavernous fistula because each requires treatment with a distinct endovascular method. Carotid-cavernous fistulae are treated with detachable balloons while DAVMs in this region are best treated by transvenous obliteration of the involved sinus.
Conclusion
The results of this review emphasize that no single treatment is ideal for the obliteration of DAVMs. Management requires a team approach that must be individualized for each patient. The location of the lesion and venous anatomy are the key factors in selecting a surgical, endovascular, or combined approach.
References
- Aminoff MJ: Vascular anomalies in the intracranial dura mater. Brain 96:601-612, 1973
- Awad IA: Intracranial dural arteriovenous malformations, in Wilkins RH, Rengachary SS (eds): Neurosurgery. New York: McGraw-Hill, 1996, pp 2519-2527
- Awad IA, Little JR, Akarawi WP, et al: Intracranial dural arteriovenous malformations: Factors predisposing to an aggressive neurological course. J Neurosurg 72:839-850, 1990
- Borden JA, Wu JK, Shucart WA: A proposed classification for spinal and cranial dural arteriovenous fistulous malformations and implications for treatment. J Neurosurg 82:166-179, 1995
- Brown RD Jr, Wiebers DO, Nichols DA: Intracranial dural arteriovenous fistulae: Angiographic predictors of an intracranial hemorrhage and clinical outcome in nonsurgical patients. J Neurosurg 81:531-538, 1994
- Castaigne B, Bories J, Brunet P, et al: Les fistules artério-veineuses méningées pures a drainage veineux cortical. Rev Neurol (Paris) 132:169-181, 1976
- Challa VR, Moody DM, Brown WR: Vascular malformations of the central nervous system. J Neuropathol Exp Neurol 54:609-621, 1995
- Cognard C, Gobin YP, Pierot L, et al: Cerebral dural arteriovenous fistulas: Clinical and angiographic correlation with a revised classification of venous drainage. Radiology 194:671-680, 1995
- Davies MA, TerBrugge K, Willinsky R, et al: The validity of classification for the clinical presentation of intracranial dural arteriovenous fistulas. J Neurosurg 85:830-837, 1996
- Djindjian R, Merland JJ: Superselective Arteriography of the External Carotid Artery. New York: Springer-Verlag, 1978
- Garcia H, Anderson ML: Circulatory disorders and their effect on the brain, in Davis RL, Robertson DM (eds): Textbook of Neuropathology. Baltimore: Williams & Wilkins, 1997, pp 715-822
- Herman JM, Spetzler RF, Bederson JB, et al: Genesis of a dural arteriovenous malformation in a rat model. J Neurosurg 83:539-545, 1995
- Hoang TA, Hasso AN: Intracranial vascular malformations. Neuroimaging Clin N Am 4:823-842, 199414.
- Houser OW, Baker HL, Jr, Rhoton AL Jr: Intracranial dural arteriovenous malformations. Radiology 105:55-64, 1972
- Houser OW, Campbell JK, Campbell RJ, et al: Arteriovenous malformation affecting the transverse dural sinus—an acquired lesion. Mayo Clin Proc 54:651-661, 1979
- Lasjaunias P, Chiu M, ter Brugge K, et al: Neurological manifestations of intracranial dural arteriovenous malformations. J Neurosurg 64:724-730, 1986
- Lucas CP, de Oliveira E, Tedeschi H, et al: Sinus skeletonization: A treatment for dural arteriovenous malformations of the tentorial apex. Report of two cases. J Neurosurg 84:514-517, 1996
- Lucas CP, Zabramski JM, Spetzler RF, et al: Treatment for intracranial dural arteriovenous malformations: A meta-analysis from the English language literature. Neurosurgery 40:1119-1130, 1997
- Malik GM, Pearce JE, Ausman JI, et al: Dural arteriovenous malformation and intracranial hemorrhage. Neurosurgery 15:332-339, 1984
- Obrador S, Soto M, Silvela J: Clinical syndromes of arteriovenous malformations of the transverse-sigmoid sinus. J Neurol Neurosurg Psychiatry 38:436-451, 1975