Postoperative Spinal Epidural Hematomas: Longitudinal Review of 12,000 Spinal Operations
Randall W. Porter, MD
Paul W. Detwiler, MS, MD
Michael T. Lawton, MD†
Volker K.H. Sonntag, MD
Curtis A. Dickman, MD
Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
†Current Address: University of California, San Francisco, California
Abstract
A retrospective review of 12,000 patients who underwent spinal surgery from 1979 to 1996 revealed 17 patients who developed postoperative spinal epidural hematomas that required evacuation. We attempted to determine if surgical timing, rate of symptom progression, or preoperative Frankel grade influenced outcome. New deficits were present immediately after surgery in seven patients and presented in a delayed fashion in 10 patients. The mean interval from symptom onset to surgery was 17.7 hours (range, 1.7 to 132 hours). Postoperative motor and sensory deficits developed from C4 to S2 and patients deteriorated a mean 1.2 grades. Twelve patients were evaluated with CT myelography or MR imaging, one with plain radiographs, and four on clinical suspicion alone. Postoperative symptoms were immediate in seven and delayed in 10 patients. Coagulopathy and vascular lesions were the most common causes for postoperative spinal epidural hematomas. At late follow-up (mean length, 31.6 months), 10 patients had returned to their baseline, 5 had improved, and 2 were worse. Patients who had their hematomas evacuated in less and more than 6 hours improved a mean of 2.1 and 0.6 Frankel grades, respectively. Four patients deteriorated to Frankel A grades but still improved a mean of 1.75 grades after evacuation. Rapid evacuation of postoperative spinal epidural hematomas may maximize neurological outcomes, and even patients with complete sensorimotor deficits may improve after evacuation.
Key Words: complications, epidural hematoma, spinal cord injury
Spinal epidural hematomas are a rare cause of neurologic deficits and spinal cord compression. First described by Jackson in 1869,[14] spinal epidural hematomas result from anticoagulation,[13,17,18,24] hemophilia,[15] pregnancy,[5] trauma,[17,22,23] garlic ingestion,[10] a complication of anterior cervical discectomy,[33] aspirin,[19] hypertension,[24] epidural anesthesia,[5,17] Paget’s disease,[18] hemangioma,[15,24] arteriovenous malformation (AVM),[6,17] and epidural varicose veins.[5] They also may occur spontaneously[2,10,17,22,23] with no obvious etiology.
Considering the large number of spinal operations performed each year, spinal epidural hematomas are surprisingly rare after spinal surgery.[3,8,16,33] Nonetheless, spinal surgeons should be aware of this clinical entity and its predisposing factors. Previously, we reported 30 patients with spinal epidural hematomas from all causes, 12 of which occurred postoperatively.[17] This article adds five more cases and more closely examines this subpopulation of patients.
Patient Population
Between 1979 and 1996, 17 (14 males and 3 females) patients undergoing spinal surgery at our institution developed spinal epidural hematomas that needed evacuation. Their mean age was 51 years (range, 6 months to 78 years). Patients’ medical records, radiographs, operative reports, and pathology reports were reviewed retrospectively. Postoperative spinal epidural hematomas were confirmed by magnetic resonance (MR) imaging or computed tomography (CT)-myelography, by the surgeon intraoperatively, by the pathologist histologically, or by all three.
The subjects underwent a variety of surgical procedures throughout the spinal axis. Anterior cervical discectomy with and without fusion accounted for the largest group (n =6), two of whom underwent plating procedures with Synthes (Synthes® Spine, Paoli, PA) plates. One patient underwent a multilevel cervical corpectomy. Three patients underwent laminectomies for spinal epidural hematomas. Three patients had spinal AVMs—one cervical (the patient presented with a spinal epidural hematoma), one cervicothoracic, and one thoracolumbar. One patient underwent a thoracic laminectomy for spinal cord compression due to nonHodgkin’s lymphoma.
Two patients underwent posterior thoracolumbar decompression with instrumentation. Three patients underwent lumbar laminectomies for spinal stenosis (Table 1). Patients 8 and 15 had drains placed at surgery for excessive oozing. Both had coagulopathies. Patient 8 was a 6 month old who had 425 ml of output in 24 hours. Patient 15 was an adult who had 100 ml of output in the 12 hours after the first surgery.
Patient Evaluation
The Frankel grading system[9] was used to grade each patient’s neurologic status: A=complete motor and sensory loss; B=complete motor, incomplete sensory loss; C=incomplete motor loss, nonfunctional; D=incomplete motor loss, functional; and E=no neurological deficits. The patients’ neurologic status was defined at four points in time: before the initial surgical procedure, after the initial procedure (at the time of deterioration), after evacuation of the postoperative spinal epidural hematoma, and at late follow-up. The patient’s urologic function was also ascertained before surgery, at the time the postoperative spinal epidural hematoma occurred (when available), and at follow-up.
Results
Clinical Features
During the 17-year study period, approximately 12,000 spine operations were performed at our institution. Seventeen patients developed postoperative spinal epidural hematomas, an incidence of 0.14%. Before the initial surgery, four patients were Frankel grade E, seven were grade D, three were grade C, and three were grade B (Table 1). Bladder function was normal in nine patients and abnormal in six. Bladder function could not be determined in the two patients who were younger than 1 year.
After the initial surgical procedure, four patients were Frankel grade A, three were grade B, seven were grade C, and three were grade D. Postoperative motor and sensory deficits developed from C4 to S2. Three patients presented with weakness, two with weakness and numbness, four with paraplegia, one with quadriplegia, one with paresthesia, one with an anterior cord syndrome, two with sepsis, one with dysphagia and a wound hematoma, one with radiculopathy, and one with cauda equina syndrome. Bladder function was normal in four and abnormal in six. Bladder function could not be documented in three patients because immediate postoperative motor deficits necessitated urgent evacuation. Two patients had indwelling catheters after surgery. A clinical urologic evaluation was not possible in the two infants.
After the initial surgical procedure, the patients deteriorated a mean of 1.2 Frankel grades. The Frankel grades of five patients did not change. The latter group complained of increased pain, radiculopathy, paresthesia, sensory loss, or mild changes in their motor examination, but overall their status did not change enough to manifest as a change in grade. Two of the five patients (Patients 8 and 10) failed to improve as expected; they remained at Frankel grade B, prompting immediate postoperative studies (Table 1).
Radiographic Evaluation
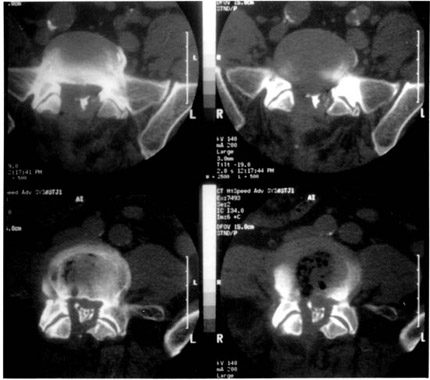
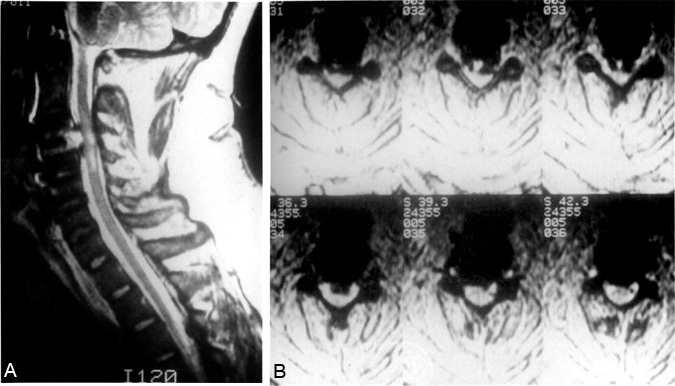
Thirteen patients underwent radiographic evaluation after their initial surgical procedure. Four patients were taken to the operating room on clinical suspicion alone, without a diagnostic study. One patient was evaluated by plain radiography of the cervical spine to rule out a malpositioned fibular strut graft. The patient became symptomatic with a wound hematoma, neck swelling, and dysphagia. Although the graft’s position was satisfactory, swelling of the prevertebral soft tissue was significant. Six patients were evaluated with CT-myelography, four with MR imaging, and two with both (Figs. 1 and 2). Epidural hematomas were observed in all regions of the spinal axis: eight cervical, three thoracic, three lumbar, two thoracolumbar (T8-L2), and one cervicothoracic. Six were anterior, 10 were posterior, and 1 was anterior and posterior. Twelve were primarily in the midline; two were eccentric to the right and three to the left. All anterior spinal epidural hematomas developed after cervical corpectomies or discectomies while all posterior spinal epidural hematomas developed after laminectomies. The hematoma that was both anterior and posterior to the spinal cord developed after an anterior cervical discectomy and fusion.
Surgical Timing
The onset of postoperative deficits upon recovering from anesthesia was immediate in 7 patients and delayed in 10. Eight patients were returned to the operating room emergently (<6 hours). Nine patients were returned to the operating room urgently (>6 hours). The hematomas were evacuated through the previous surgical approach in 12 patients. The remaining five patients underwent evacuation of the spinal epidural hematoma in addition to the following procedures. One patient underwent a right-sided microdiscectomy. One patient’s laminectomy was widened to evacuate a recurrent thoracic epidural hematoma. One patient’s laminectomy was extended to evacuate a recurrent spinal epidural hematoma and to resect an unrecognized cervical AVM. Harrington rods were adjusted in one patient. One patient required a thoracolumbar laminectomy and manipulation of Cotrel-Dubousset instrumentation.
Progression of Symptoms
The mean interval from initial symptoms to maximal deficit was 4.8 hours (range, 0 to 24 hours). Eight patients deteriorated a mean of one Frankel grade to maximal deficit in more than 1 hour. Nine patients deteriorated a mean of 1.3 grades to maximal deficit in less than 1 hour. The symptoms of patients who ultimately recovered completely (Frankel E) progressed to maximal deficit over a wide range of values (0 to 18 hours).
The mean interval from symptom onset to surgery was 17.7 hours (range, 1.7 to 132 hours). This interval was 7.1 hours for patients who made complete neurological recoveries (Frankel E, Patients 2, 8, 12, 13, 17; Table 1) compared with 22.4 hours for the 12 patients with less than complete recoveries (Frankel A, B, C, or D). The eight patients whose symptoms-to-surgery interval was less than 6 hours improved a mean of 2.1 grades while the nine patients whose symptoms-to-surgery interval was longer than 6 hours improved only 0.6 grades (Table 2). One patient, however, improved two grades after experiencing deficits for 20 hours, and one patient improved one grade after 24 hours of deficit. The mean interval from maximal deficit to surgery was 12.8 hours (range, 0.3 to 108 hours). Eight patients whose maximal deficit lasted less than 6 hours improved a mean of 2.1 grades. Five patients whose maximal deficit to surgery lasted more than 6 hours did not improve.
Etiology
There was no obvious cause for the postoperative spinal epidural hematoma in five patients. Potential risk factors for postoperative bleeding were identified in 12 patients. Three patients had spinal AVMs (one also had factor VIII deficiency and one was on aspirin). Two patients had liver disease with coagulopathy. Three patients were on aspirin or nonsteroidal anti-inflammatory medications. One patient bled heavily beneath the posterior longitudinal ligament. One patient presented with a wound hematoma that caused dysphagia 1 week after the initial operation. Bleeding from an esophageal artery was discovered at surgery. One patient had a vascular metastatic tumor in the thoracic spine (nonHodgkin’s lymphoma).
Three of these patients (Nos. 8, 9, 10) also presented with recurrent spinal epidural hematomas (one from epidural veins and two from spinal AVMs). In other words, the initial surgical procedure was to evacuate a spinal epidural hematoma, which then recurred, necessitating a second evacuation (Table 1). A 6-month-old male with a history of factor VIII deficiency who underwent a cervical laminectomy for a spinal epidural hematoma caused by a type I spinal AVM remained quadriplegic and became hemodynamically unstable. MR imaging revealed that the hematoma had recurred. Subsequently, it was evacuated. At a 90-month follow-up examination, the child had no neurological deficits.
A 1-year-old male who presented with spontaneous quadriplegia (Patient 9) underwent C3-T2 and T5-T7 laminectomies and evacuation of a spinal epidural hematoma. Postoperatively, he was still quadriplegic. Repeat myelography showed a C6 block and he underwent a second evacuation and an additional laminectomy at C2. A Type I spinal AVM (dorsal intradural fistula) was also found, and the fistula was resected. At a 26-month follow-up examination, he was ambulating independently but his lower extremities were spastic.
A 33-year-old male (Patient 10) presented with interscapular and leg pain, an anterior cord syndrome, and incontinence. CT-myelography showed a complete block at T5-T6. He underwent a T3-T7 laminectomy with evacuation of a posterior spinal epidural hematoma. Postoperatively, he was still paraplegic. Myelography showed a complete block at T5-T6 consistent with a recurrence, which was emergently evacuated. At a 1-month follow-up examination, he was a Frankel grade D.
The mean length of follow-up was 31.6 months (range, 22 days to 90 months). After evacuation of the postoperative spinal epidural hematoma, Frankel grades improved in 13 patients: four improved one grade, six improved two grades, and three improved three grades. Frankel grade did not change in four patients. Overall, the mean improvement was 1.5 Frankel grades. No patients were worse after evacuation. Comparing the patients’ Frankel grades before the initial surgery and after evacuation of the spinal epidural hematoma, 10 patients were at their baseline, 5 had improved (three improved one Frankel grade, one improved two grades, and one improved three grades), and two patients were worse. At the latest follow-up, two patients were Frankel grade A, one was Frankel grade B, nine were Frankel grade D, and five were Frankel grade E. Bladder function was normal in 10 patients and abnormal in seven. Compared with their baseline bladder function, two patients deteriorated, two improved, and 11 were unchanged. Again, bladder function could not be determined in the two infants.
Fourteen patients had postoperative spinal epidural hematomas on the spinal cord. Their final Frankel grades were A=1, B=1, D=8, and E=4. After evacuation (at last follow-up), eight were unchanged compared to their baseline, one improved three grades, one improved two grades, and two improved one grade. One patient each deteriorated two and one Frankel grade, respectively. One patient had a lesion that involved the conus medullaris and cauda equina. At last follow-up, he had improved one grade to Frankel C. Two patients with lesions of the cauda equina only had returned to baseline at last follow-up (one grade E, one grade C). Of the four patients who were Frankel grade A after the initial surgery, one improved to grade C initially. Two weeks later, however, he developed sepsis and disseminated intravascular coagulation and again deteriorated to grade A. His unstable medical condition and irreversible coagulopathy precluded further surgery. The other three grade A patients improved a mean of 2.3 grades. The patient who remained a Frankel B had a spinal AVM. He awoke with an anterior cord syndrome, and his condition never improved despite evacuation of the postoperative spinal epidural hematoma.
By late follow-up four patients had died: one from metastatic disease 2 years later, one from transfusion-related auto-immune deficiency syndrome 8 years later, one from multisystem organ failure 22 days after surgery, and one at the age of 79 from unrelated causes. Three patients were using walkers: One had returned to baseline, one had not walked preoperatively, and the third patient’s (mentioned above) metastatic disease progressed. One patient was disabled because of hepatitis C and one was unemployed because of alcoholism.
Discussion
It has been estimated that the etiology of 41% of spontaneous cases of spinal epidural hematoma is unknown.7 Epidural hematomas of the spinal canal have been reported after invasive procedures such as epidural anesthesia, especially when anticoagulant or thrombolytic therapy was employed,1,4 but they are rare after spinal surgery. In Foo and Rossier’s8 review of 158 cases of spinal epidural hematomas from the world literature, no cases had developed after surgery. U and Wilson33 reported spinal epidural hematomas after 3% of their anterior cervical discectomies. Laursen et al.16 reported a patient who developed a spinal epidural hematoma 1 week after lumbar microdiscectomy. Di Lauro et al.3 reported that 2 of 990 patients developed spinal epidural hematomas after lumbar discectomy. At our institution the overall incidence of postoperative spinal epidural hematoma was 0.14%. The exact incidence within each category cannot be determined from our computer database, but we recently reviewed our spinal AVM experience and found 70 surgically treated cases. Thus, our incidence of spinal epidural hematoma after surgery for spinal AVM is 5.7% (4/70). Two of these patients, however, had a spinal epidural hematoma as their presenting symptoms. None of the patients had residual AVM, but this seems to be a high-risk subpopulation at our institution.
Diagnosis
A patient with new deficits after spinal surgery should be evaluated quickly. Physical and neurological examinations in the recovery room are an essential part of immediate postoperative care. If the deficits are profound (i.e., new complete motor or sensory loss), the patient should be returned to surgery without delay. Epidural hematomas, bone fragments, intervertebral disc fragments, or a foreign body may be compressing the spinal cord or nerve roots.
Obtaining plain radiographs in the recovery room can help exclude bone fragments, allograft compression, or malpositioned hardware as a cause of spinal epidural hematoma. The diagnostic modality of choice for a spinal epidural hematoma is MR imaging. If it is unavailable or contraindicated, CT-myelography can be used. An extradural defect immediately after surgery suggests the presence of a spinal epidural hematoma. If the onset of the deficit is delayed by days or weeks, the possibility of a spinal epidural abscess should also be considered.
Management and Treatment
Spontaneous recovery without surgery is rare but has been observed in one patient who developed a spinal epidural hematoma after anticoagulation therapy[13] and in one patient with hemophilia.[27] However, the management of a symptomatic postoperative spinal epidural hematoma typically requires emergency surgical evacuation. The previous surgical site should be reopened and the epidural space inspected. Occasionally, the exposure is extended. The entire clot should be evacuated and the surgical bed should be inspected for epidural bleeding, which tends to arise from the epidural venous plexus. Arterial bleeding may cause epidural hemorrhage, which in the cervical spine can originate from the arterial plexus between the posterior aspect of the vertebral body and the posterior longitudinal ligament.[33]
An unrecognized Type I spinal AVM provided an arterial source of epidural hemorrhage in two of our patients (Patients 8 and 9). This malformation is distinguished by an arteriovenous fistula at the dural root sleeve. The exact site of the fistula is controversial. However, epidural, subdural, and subarachnoid hemorrhages have been reported.[12] These lesions should be suspected in spontaneous cases of epidural hemorrhage. Angiography often demonstrates the lesion but false negatives are common. Thus, we recommend intradural exploration if the source or cause of bleeding is not readily apparent.
Surgical Timing
Surgery should proceed without delay. Previous authors have also substantiated that surgical timing and preoperative neurological condition are related to neurological outcome.[2,7,11,17,20,25,28] In the present study, patients who underwent surgery within 6 hours improved more than twice as many Frankel grades as those who underwent surgery after 6 hours. The goal is to decompress the neural elements before the spinal arterial supply is compromised.[21] In laboratory studies with a canine model, neurological recovery after spinal cord compression with epidural balloons has depended on the force and duration of compression and on the rapidity of the onset of motor deficits.[29-32] In this study, however, the rapidity of onset of symptoms did not seem to correlate with outcome, as evidenced by the five patients who recovered completely. Their symptoms progressed over the wide interval between zero and 18 hours. Further, those whose symptoms progressed to maximal deficit in less than 1 hour deteriorated a mean of 1.3 grades. Those whose symptoms progressed to maximal deficit over a period longer than 1 hour deteriorated one grade.
Reasonable neurological recovery has been observed even when neurological deficits have persisted as long as 2 weeks after symptom onset,[16] especially if the symptoms developed slowly.[21] Even patients with complete motor and/or sensory deficits can improve neurologically.[15,22,25] Lougheed and Hoffman[20] reported that a patient who had been paraplegic for 3 days improved neurologically after evacuation of a spinal epidural hematoma. In their review of 158 spinal epidural hematomas, Foo and Rossier[7] found that postoperative motor function was present in 45.3% of the Frankel A patients, 11% of whom recovered completely. This tenet is supported by the mean improvement of 1.75 grades in the four patients in our series who initially had deteriorated to Frankel grade A. We therefore advocate urgent decompression of the neural elements once spinal epidural hematoma is diagnosed even in the presence of a complete motor/ sensory deficit.
Prevention
When confronted with a complication after surgery, the surgeon must determine how to avoid the complication in the future. Preoperatively, a family or medical history of bleeding diatheses should be recognized and treated. Use of anticoagulants or other platelet-inhibitory medications, heavy alcohol use, or liver disease also should be identified. Intraoperatively, attention to hemostasis should be meticulous, especially at closure.
The use of drains, especially after extensive thoracolumbar laminectomies with instrumentation, is common at our institution. However, hemostasis was deemed to be adequate in Patients 12 and 13, and drains were not placed. These patients had no other predisposing factors for developing spinal epidural hematomas; consequently, epidural drains might have prevented their hematomas from developing. The output of Patient 8 was far greater (425 ml/24 hours) than would be expected in a 6-month-old child, and the drain provided a clue to the amount of ongoing epidural bleeding. Patient 15 had a coagulopathy (and thus intraoperative oozing) related to alcoholic liver disease and renal failure. The drain, however, did not show excessive output in the first 12 hours after surgery.
Three patients who underwent anterior cervical discectomy developed postoperative spinal epidural hematomas. One patient had significant intraoperative bleeding from under the posterior longitudinal ligament at the time of the first surgery. One patient was on aspirin and had a lumboperitoneal shunt in place that may have created a negative pressure in the epidural space. The third patient had no predisposing factors. During anterior cervical discectomy, U and Wilson[33] have suggested placing a nerve hook or angled dissector in the cervical spinal canal underneath the posterior longitudinal ligament and applying direct pressure to the ligament against the posterior surface of the vertebral body. To aid in hemostasis, the cut surface of the posterior longitudinal ligament can be coagulated with bipolar cauterization.
Postoperatively, the patient should be kept in a calm environment and prevented from excessive straining or coughing. The epidural venous plexus has no venous valves[26] and anastomoses via the segmental veins with the inferior vena cava, azygos, and hemizygos veins. Thus, a Valsalva maneuver transmits the visceral cavity pressure directly to the epidural venous plexus and may thereby precipitate epidural bleeding. In their review, Markham et al.[22] found that spinal epidural hematomas were associated with straining at stool, micturition, lifting, sneezing, and shoveling coal. Finally, some authors have recommended observing patients in the recovery room for 3 hours after anterior cervical discectomy.[33] We observe patients for an hour or long enough to allow them to awaken for a detailed neurological examination. The most important step in prevention of spinal epidural hematoma, however, is to suspect it in the differential diagnosis of postoperative patients with new deficits.
References
- Anonymous: Major complications in continuous epidural anesthesia. Chin Med J 93:194-200, 1980
- Banerjee T, Meagher JN: Spontaneous spinal epidural hemorrhage. South Med J 67:858-861, 1974
- Di Lauro L, Poli R, Bortoluzzi M, et al: Paresthesias after lumbar disc removal and their relationship to epidural hematoma. Report of two cases. J Neurosurg 57:135-136, 1982
- Dickman CA, Shedd SA, Spetzler RF, et al: Spinal epidural hematoma associated with epidural anesthesia: Complications of systemic heparinization in patients receiving peripheral vascular thrombolytic therapy. Anesthesiology 72:947-950, 1990
- Dickman CA, Zabramski JM, Sonntag VKH, et al: Myelopathy due to epidural varicose veins of the cervicothoracic junction. Case report. J Neurosurg 69:940-941, 1988
- Foo D, Chang YC, Rossier AB: Spontaneous cervical epidural hemorrhage, anterior cord syndrome, and familial vascular malformation: Case report. Neurology 30:308-311, 1980
- Foo D, Rossier AB: Preoperative neurological status in predicting surgical outcome of spinal epidural hematomas. Surg Neurol 15:389-401, 1981
- Foo D, Rossier AB: Post-traumatic spinal epidural hematoma. Neurosurgery 11:25-32, 1982
- Frankel HL, Hancock DO, Hyslop G, et al: The value of postural reduction in the initial management of closed injuries of the spine with paraplegia and tetraplegia. Part I. Paraplegia 7:179-192, 1969
- Gold ME: Spontaneous spinal epidural hematoma. Radiology 80:823-828, 1963
- Grollmus J, Hoff J: Spontaneous spinal epidural haemorrhage: Good results after early treatment. J Neurol Neurosurg Psychiatry 38:89-90, 1975
- Han P, Theodore N, Porter RW, et al: Subdural hematoma from a type I spinal arteriovenous malformation. Case report. J Neurosurg (Spine 2) 90:255-257, 1999
- Harik SI, Raichle ME, Reis DJ: Spontaneously remitting spinal epidural hematoma in a patient on anticoagulants. N Engl J Med 284:1355-1357, 1971
- Jackson R: Case of spinal apoplexy. Lancet 2:5-6, 1869
- Kosary IZ, Braham J, Shacked I, et al: Spinal epidural hematoma due to hemangioma of vertebra. Surg Neurol 7:61-62, 1977
- Laursen J, Fode K, Dahlerup B: Spinal epidural hematomas. Clin Neurol Neurosurg 89:247-253, 1987
- Lawton MT, Porter RW, Heiserman JE, et al: Surgical management of spinal epidural hematoma: Relationship between surgical timing and neurological outcome. J Neurosurg 83:1-7, 1995
- Lee KS, McWhorter JM, Angelo JN: Spinal epidural hematoma associated with Paget’s disease. Surg Neurol 30:131-134, 1988
- Locke GE, Giorgio AJ, Biggers SL, Jr., et al: Acute spinal epidural hematoma secondary to aspirin-induced prolonged bleeding. Surg Neurol 5:293-296, 1976
- Lougheed WM, Hoffman HJ: Spontaneous spinal extradural hematoma. Neurology 10:1059-1063, 1960
- Lowrey JJ: Spinal epidural hematomas. Experiences with three patients. J Neurosurg 16:508-513, 1959
- Markham JW, Lynge HN, Stahlman GE: The syndrome on spontaneous spinal epidural hematoma. Report of three cases. J Neurosurg 26:334-342, 1967
- Matsumae M, Shimoda M, Shibuya N, et al: Spontaneous cervical epidural hematoma. Surg Neurol 28:381-384, 1987
- Mayer JA: Extradural spinal hemorrhage. Can Med Assoc J 89:1034-1037, 1963
- McQuarrie IG: Recovery from paraplegia caused by spontaneous spinal epidural hematoma. Neurology 28:224-228, 1978
- Norgore M: Clinical anatomy of the vertebral veins. Surgery 17:606-614, 1945
- Priest WM: Epidural hemorrhage due to hemophilia causing compression of the spinal cord. Lancet 2:1289-1291, 1935
- Scott BB, Quisling RG, Miller CA, et al: Spinal epidural hematoma. JAMA 235:513-515, 1976
- Tarlov IM: Spinal cord compression studies. III. Time limits for recovery after gradual compression in dogs. Arch Neurol Psychiatry 71:588-597, 1954
- Tarlov IM, Herz E: Spinal cord compression studies. IV. Outlook with complete paralysis in man. Arch Neurol Psychiatry 72:43-59, 1954
- Tarlov IM, Klinger H: Spinal cord compression studies. II. Time limits for recovery after acute compression in dogs. Arch Neurol Psychiatry 71:271-290, 1954
- Tarlov IM, Klinger H, Vitale S: Spinal compression studies. I. Experimental techniques to produce acute and gradual compression. Arch Neurol Psychiatry 70:813-819, 1953
- U HS, Wilson CB: Postoperative epidural hematoma as a complication of anterior cervical discectomy. Report of three cases. J Neurosurg 49:288-291, 1978