
Rehabilitation of Higher Cerebral Functions and the Patient’s Personality†
George P. Prigatano, PhD
Division of Neurology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
†Adapted from Prigatano GP: Rehabilitation of higher cerebral functions and the patient’s personality. Japanese Neuropsychological Journal (in press). With permission from the Japanese Neuropsychological Journal.
Abstract: The rehabilitation of higher cerebral functions after various brain lesions often requires remediation of both cognitive and personality disturbances during the acute and postacute phases of injury. Luria’s original insights regarding restoration of higher cerebral functions are supported by contemporary neuroscientific investigations. However, teaching brain dysfunctional patients social skills and helping them with their personal losses are equally important tasks. Higher cerebral functions can recover maximally only when heteromodal regions of the brain (which integrate thinking and feeling) regain their functional integrity.
Key Words: higher cerebral functions, personality, rehabilitation
The rehabilitation of patients with disturbances in their higher cerebral functions caused by various brain lesions requires that both cognitive and personality disturbances be addressed. Failure to do so, particularly in patients with a moderate to severe traumatic brain injury (TBI), leads to glib or erroneous conclusions 4,43 about the efficacy of cognitive rehabilitation as practiced within the broad context of neuropsychological rehabilitation.31,33,35
Neuropsychology, as the scientific study of brain-behavior relationships, has been preoccupied with studying cognitive deficits and has excluded a thorough analysis of patients’ emotional and motivational characteristics. Without studying the interaction of personality and cognitive disturbances as they emerge in patients at various stages after brain injury, only a partial view of how various brain lesions affect behavior is obtained and approaches to rehabilitation will remain limited.
A 1998 conference sponsored by the National Institutes for Health on the rehabilitation of persons with TBI noted that both “cognitive exercises” and psychotherapy appeared to hold promise as important components of neurorehabilitation.27 The link, however, between therapeutic interventions for rehabilitation of higher cerebral functions (after various brain injuries) and the personality of patients is often missing. Yet, poignant reports such as Luria’s Man with a Shattered World22 show that this interaction is powerful. Personality influences how patients cope emotionally with residual cognitive deficits and how they approach neurorehabilitation.
Perhaps of more interest to academic neuropsychologists is the propo sition that the underlying cognitive deficits of patients influence the emergence of personality disturbances after brain injury. Conversely, the underlying personality characteristics of patients may influence the expression of cognitive deficits. Research on personality characteristics of dyslexic adults suggests that the underlying brain abnormality may influence both phonological-linguistic development and patterns of interpersonal relationships.13,42 This type of research, often lacking in neuropsychology, is badly needed. This article attempts to demonstrate the importance of working with both cognitive and personality disturbances after brain injury and how such work has led to certain theoretical positions concerning adaptation and recovery phenomena after brain injury.
Consequences of Not Providing Rehabilitation for Cognitive and Personality Disturbances After Brain Injury
Initially, patients with a moderate to severe TBI often show partial recovery of their higher brain functions, only to be left with permanent impairments. As a group, they have difficulty maintaining employment2 and eventually become socially isolated.18 Many brain-injured children show a decline in academic performance and behavioral adaptation over time (Rivara, unpublished data, 1994). Cognitive, behavioral, and social functioning often deteriorates. Patients with good premorbid and psycho social adjustment may show specific cognitive deficits after sustaining a focal brain lesion, but certain emotional reactions and personality changes are predictable.17,39 A long-term outcome study suggests that a portion of brain dysfunctional patients may show an unexpected cognitive decline as they age.5
The inability to control emotions, particularly angry outbursts and depression, has been related to poor rehabilitation outcomes1 and failure to maintain employment.2 It therefore becomes crucial to understand and to treat both cognitive and personality disturbances in brain dysfunctional patients.
Personality and the Symptom Picture After Brain Damage
A model for conceptualizing personality disturbances after brain injury has been presented elsewhere.33,37 A few examples given here highlight how personality factors influence or contribute to the resulting symptom picture.
During acute confusional states, how brain dysfunctional patients explain themselves in their environment varies. After a severe TBI, a young woman concerned about getting pregnant before marriage attributed her hospitalization to her being in the hospital to have a baby. A young man who always sought attention and relished himself as having a personality similar to that of John Wayne explained his stay in an inpatient neurorehabilitation unit as being on a M*A*S*H movie set. Some actors were playing doctors and other actors were playing patients; he was playing the role of a patient.
Even beyond the acute confusional state, patients can still confabulate. Confabulation is statistically related to the presence of frontal lesions,7 but the content of the confabulation varies greatly across patients. Weinstein et al.46 reported that a high-ranking officer who suffered from dysarthria and dysgraphia confabulated that he had written a speech for the President of the United States to deliver.
Months and years after a brain injury, patients exhibit different preoccupations that are compatible with their premorbid concerns. For example, a successful businessman known for marginally legal dealings worried that he did not have enough money in his bank account after he experienced cerebral anoxia during cardiac arrest. Despite repeated reassurances from his wife, who showed him their monthly financial statements, his perseverative tendencies persisted. He repeatedly asked to review their financial statements and expressed concerns that they did not have enough money despite his wife’s reassurances. A patient with dementia of the Alzheimer’s type, who had a history of inappropriate sexual behavior including sexual abuse of a stepdaughter, repeatedly asked for sexual contact with his wife and next door neighbor despite the inappropriateness of the request and his sexual impotence. Numerous other examples could be given of how pres ent and past personality characteristics influence a person’s behavior after brain injury and how these characteristics make their rehabilitation a relatively positive or negative experience for those involved in their care.
Implications for Cognitive Rehabilitation
Cognitive rehabilitation has been defined in several ways.33 I define it as a training experience intended to restore higher cerebral functions and to teach patients to manage their dysfunctions in daily life.
Traditionally, there have been three approaches to the remediation of cognitive impairments: (1) drill and practice exercises to improve an impairment, (2) teaching strategies to circumvent a theoretically defined component of a cognitive process considered to produce the impairments, and (3) a “broad-band” approach that attempts to facilitate recovery in a wide variety of theoretically defined functions (i.e., arousal, alertness, attention, memory, mental energy).34 These three approaches have been applied to the treatment of underlying impairments. When the goal is to treat the disability, compensatory training and psychotherapy have often been employed.15,39
Patients exhibit a wide variety of reactions to cognitive rehabilitation. Many resist it and believe it to be useless. Sometimes they are right. Patients, however, must be engaged in the rehabilitation process to determine to what degree function can be restored. As patients become more aware of their residual cognitive strengths and limitations, they can be taught compensatory strategies to help them gain control and mastery in some areas of function. This process can lead to greater social reintegration. Examples of this process within the context of a neuropsychological rehabilitation program can be found elsewhere.33,37
Two studies provide evidence that such rehabilitation helps brain dysfunctional patients to return to work and that it may foster modest improvement in their cognitive functioning while helping them to obtain greater emotional control and to improve their social relationships.36,38 Changing patients’ attitude about themselves and their capacity to regain a productive lifestyle seems crucial to this process. Failure to deal adequately with both the cognitive and personality disturbances of such patients is associated with less promising outcomes.44
The work of Merzenich and colleagues provides an example of how advances in the cognitive neurosciences improve approaches to cognitive retraining.23,45 They described a new technology that can be used to teach children with language-based learning impairments to make finer and finer phonemic discriminations, which improve their language function:
Cognitive neuroscience studies also reveal the most effective strategies for driving brain change. The subject must be attentive and motivated. The training must be progressive and adaptive to each training subject. The training schedule must be repetitious and the training schedule intense (p. 5).24
Despite the sophistication of these statements, they were made several years ago by individuals who took seriously the need to attempt to rehabilitate brain dysfunctional patients.
Historical Insights and Contemporary Findings
Slightly more than 75 years ago, Shepherd Ivory Franz9 made a number of observations that are pertinent to contemporary efforts at cognitive rehabilitation. In describing the “re-education” process of three young men who were aphasic after they contracted “cerebral-spinal syphilis,” he made the following points:
(1) Each retraining effort must be individualized to patients’ cognitive impairments and to their personality (i.e., their interest patterns, capacity to inhibit, fatigue level, and so on; see p. 351).
(2) The goal of all such reeducation is to enhance social reintegration (see p. 355).
(3) During the reeducation exercises, performance often varies within and across subjects. Some days patients do better than others. This variability must be understood and incorporated into any form of cognitive rehabilitation.
(4) The process by which aphasic patients relearned names was puzzling. They showed “great difficulty in learning some things and ease at the learning of others,” and the difference could not be easily explained by the therapists (p. 406). “It was impossible to determine the presence of any kind of repression or complex in the psychoanalytic sense” that could explain these differences (p. 406). Thus, Franz was interested in understanding how emotional factors may or may not have influenced the process of re-education.
(5) Although certain cerebral pathways important for performing higher cerebral functions may be “opened up” as the effects of diaschisis are reduced, they may not become functional unless individuals have the “opportunity to use them” (p. 428). This latter insight may be especially important during early stages of cognitive rehabilitation.
Karl Lashley19 reflected on his collaboration with Shepherd Ivory Franz and highlighted the importance of motivation in the learning process after brain injury. He reported an aphasic patient who could not learn the alphabet after brain injury using the clinical methods at hand. Lashley then wagered the patient 100 cigarettes that he could not learn the alphabet; apparently, the patient did so within a few trials. Motivation was an extremely important variable that influenced the patient’s performance. This clinical example and Lashley’s earlier work with animals suggested that organisms may develop “a passive attitude” toward learning after brain injury, perhaps because they are easily frustrated and confused by the learning process.33 By overcoming this “passive attitude,” the higher cerebral functioning of patients or animals may seem to “have improved.” In fact, this scenario reflects the activation of abilities that are already present but, for one reason or another, not utilized in the course of normal problem solving.
Certain clinical findings could be interpreted along these same lines.37 Some brain dysfunctional patients seem to perform better once their attitude about the impact of their brain injury on their life changes and they are taught compensatory problem-solving strategies. In this context, the development of a therapeutic alliance with patients (and family) clearly relates to patients’ ability to achieve a productive lifestyle after brain injury. This finding was first reported in 199438 and has been replicated with a larger group of patients.16
Goldstein’s Insights
Kurt Goldstein’s insights are especially useful to contemporary neuropsychologists and neurorehabilitationists because he recognized the complexity of higher cerebral symptoms and the need to define the symptoms being rehabilitated when trying to determine the efficacy of cognitive rehabilitation. The lack of such precise categorization of symptoms leads to erroneous conclusions about cognitive rehabilitation.
Goldstein10 was impressed that brain injury often affects patients’ ability to develop abstract concepts. After injury their behavior was often described as “concrete.” According to Goldstein,10 some of the symptoms that patients demonstrate reflect the loss of the abstract attitude. He was aware, however, that other symptoms were a direct result of specific brain impairments. For example, memory impairment was considered to be a direct effect of brain injury.
Most important to neurorehabilitationists, Goldstein distinguished symptoms directly caused by brain injury from those indirectly caused by brain injury. Within this latter category, he made a further distinction. Some symptoms reflected the struggle to adapt to a cognitive deficit (perhaps fatigue and irritability) while other deficits reflected a tendency to avoid this struggle (i.e., social isolation). He eloquently described that working with brain dysfunctional patients involved far more than working purely with their cognitive impairments:
Now, let us observe one of our patients. Here is a man with a lesion of the frontal lobe, to whom we present a problem in simple arithmetic. He is unable to solve it. But simply noting and recording the fact that he is unable to perform a simple multiplication would be an exceedingly inadequate account of the patient’s reaction. Just looking at him, we can see a great deal more than this arithmetical failure. He looks dazed, changes color, becomes agitated, anxious, starts to fumble, his pulse becomes irregular; a moment before amiable, he is now sullen, evasive, exhibits temper, or even become (sic) aggressive. It takes some time before it is possible to continue the examination. Because the patient is so disturbed in his whole behavior, we call situations of this kind catastrophic situations (p. 71).10
Clinical work with brain dysfunctional patients often involves helping patients and staff to manage such catastrophic reactions successfully. Managing patients’ emotional and motivational disturbances often determines if they can resume a productive lifestyle.
A. R. Luria’s Contribution
A. R. Luria articulated the need to establish a scientific basis for the rehabilitation of higher cerebral functions. In the introduction to his book, Restoration of Function After Brain Trauma, Luria21 stated the following:
The material for this book, which is now being published in the English language, was collected during and after the Second World War. In this period Soviet neurologists and psychologists, by their joint efforts, devised a rational system of treatment of patients with brain injuries and a scientifically based system for the restoration of brain functions deranged by these injuries (p. ix).
He emphasized that “the program of rehabilitation of patients with injuries to the brain must rest on a firm scientific basis. The loss of function associated with brain injury in various situations needs precise definitions; the primary disturbances resulting directly from localized injuries had to be distinguished from the secondary results of these primary disturbances” (p. ix).
Luria21 articulated three mechanisms that might contribute to partial recovery: the resolution of diaschisis, substitution (whereby “certain functions normally sustained by a particular region of the brain can be transferred to new areas” [p. xiii]), and radical reorganization of the destroyed activity “by means of entirely different neural structures, unaffected by the trauma” (p. xiii) via systematic retraining.
Subsequent research using positron emission tomography (PET) and functional magnetic resonance (fMR) imaging seems to support these insightful observations. Contemporary neuropsychological rehabilitationists must incorporate these insights into their methods. The acute onset of right cerebral hemorrhagic damage can produce bilateral cerebral metabolic depression.6 In patients with acute left hemispatial neglect who later improve, the hypometabolic activity may resolve in the so-called “unaffected” hemisphere and may decrease in the “affected” hemisphere.29 So-called “spontaneous recovery” may indeed reflect the resolution of diaschisis in some patients. No systematic studies, however, have shown that diaschisis can be reduced via cognitive retraining “exercises.”
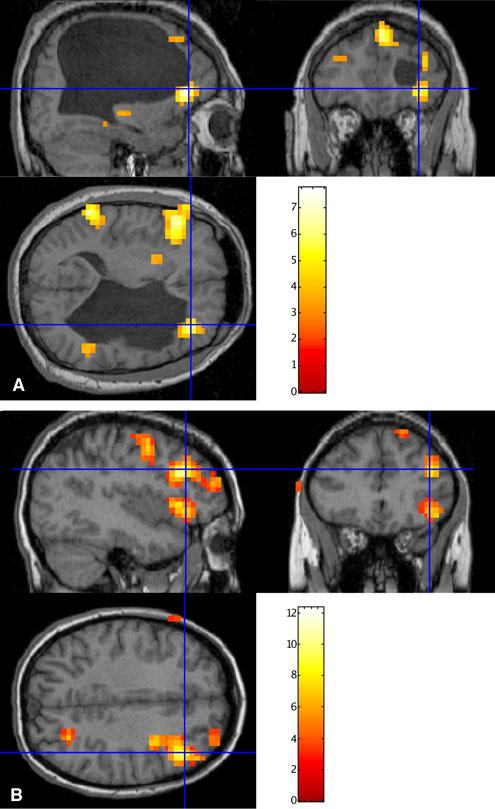
Substitution phenomena are also readily observed after brain injury. Substitution may occur as an individual interacts with the environment; it does not necessarily require specific retraining. My colleague Sterling Johnson recently attempted to map selected language functions in normal and brain dysfunctional patients. He found specific patterns of activation consistent with traditional neuropsychological perspectives. When normal individuals were involved in a word-generation task, anterior regions of the left cerebral hemisphere were activated. In contrast, when a patient with a long-standing left hemisphere porencephalic cyst was involved in such a word-generation task, cerebral blood flow increased bilaterally and posteriorly (as well as anteriorly, Fig. 1). Areas far removed from those normally activated were activated when this brain dysfunctional individual performed the word-generation task. Luria21 noted that his book did not discuss substitution phenomena; rather, it discussed the process of deinhibiting brain activity and helping to restore impaired function through reorganization or retraining.
Evidence is beginning to support the intriguing possibility that cognitive-based training may improve neural reorganization. The effects, however, are modest and a number of caveats apply to the results. A number of investigators have studied the changes in cerebral blood flow associated with partial recovery from aphasia (see Cappa, 2000, for an excellent recent review3). Recently, Musso et al.26 used PET to study short-term changes in the cortical networks involved in language comprehension in four patients with Wernicke’s aphasia. They studied cerebral blood flow changes immediately after brief, intensive language comprehension training and related those changes to performance on a version of the token test. They reported that “The region which best correlated with training-induced improvement in verbal comprehension was the posterior part of the right superior temporal gyrus and the left precuneus” (p. 781). These changes were associated with improved performance on the token test but not with perfect performance on the token test. Thus, training may stimulate specific regions of the brain to perform a function, but the regions may do so imperfectly. Restitution of function may only be possible when the area close to the actual lesion is able to regain its functional “normality.”
Pizzamiglio et al.30 reported the recovery of unilateral neglect in three patients with primarily subcortical lesions. They also used PET to study changes in cerebral blood flow when subjects performed a visuospatial task before and after treatment. The neglect improved substantially in all patients with time, training, or both (there were no control subjects). Based on their PET studies, the recovered patients performed the visuospatial tasks similar to normal controls. Behavioral improvement appeared to be associated with the return of normal cerebral activation in the right hemisphere, particularly the parietal lobe. However, bilateral activation, particularly involving the left inferior frontal gyrus, was present. Their work again suggests that the region of the brain that normally performs a function regains its normal metabolic activity in patients who recover completely or almost completely. Normal activation of the parietal lobe may be especially important for the complete resolution of neglect or hemi-inattention.
An animal study by Nudo et al.28 directly and experimentally demonstrates the role of retraining in reorganizing a damaged brain region. Four squirrel monkeys underwent daily training on a pellet removal task. The task required careful manipulation of their digits. The exact area of the primary motor cortex involved in performing these movements was recorded by intracortical microstimulation (ICMS) techniques. A very small infarct was then produced in each monkey. The predictable behavioral deficit followed. Three of the four monkeys underwent a retraining program similar to their prelesion training program, and the results of ICMS were again obtained. After the retraining, intact cortical tissue surrounding the infarct was represented when the monkeys were able to move their digits more normally. The authors concluded that ” . . . our present data suggest that rehabilitation training resulted in the prevention of the loss of spared hand area in the adjacent, intact cortex” (p. 1793).
Contemporary Efforts at Holistic Neuropsychological Rehabilitation
Contemporary efforts at holistic neuropsychological rehabilitation began in the 1970s with Yehuda Ben-Yishay and Leonard Diller’s work with Israeli brain-injured soldiers.33 Their work was influenced by the early observations of Franz, Lashley, Goldstein, Luria, and others. It, however, emphasized two new points. The first, primarily Dr. Ben-Yishay’s contribution, was that a combination of individual and small group cognitive retraining exercises embedded in a milieu-oriented rehabilitation program would substantially improve the social integration of some severely brain-injured patients. Such work helped the injured soldiers to improve their social skills. Second, as the patients participated in both individual and small group activities, the impact of their personality characteristics on others became obvious. The patients’ personal suffering about their brain injury became the focus of discussion, particularly in some group settings. As psychotherapeutic interventions helped patients deal with what I have previously referred to as the problem of lost normality,33 the role of such interventions after brain injury became more clear.
These efforts at psychotherapeutic intervention with postacute brain-injured patients who appeared to “deny” their cognitive disabilities reopened the question of whether a defensive mechanism in the psychoanalytic sense or an underlying brain disorder representing the residuals of an earlier anosognosia was involved.33 In turn, the study of the correlates of impaired self-awareness after TBI led to a re-evaluation of recovery and deterioration phenomena associated with so-called static brain lesions. The following section considers one of these earlier findings as it relates to dealing with both cognitive and affective disturbances in brain dysfunctional patients.
Data from an Intensive Neuropsychological Rehabilitation Program
Early efforts at cognitive rehabilitation emphasized exercise to improve patients’ level of arousal and “mental energy.” In turn, speed of information processing, a dimension notoriously impaired after severe TBI,20 might be facilitated.
Interesting observations were made in patients who daily practiced speeded cognitive retraining tasks.37 Postacute TBI patients who underwent an intensive neuropsychological rehabilitation program showed small but reliable improvements on the Wechsler Adult Intelligence Scale (WAIS) Performance intelligence quotient (IQ) measure (F=9.74, df=21; p<.005), the Block Design Subtest score (F=22.81, df=26; p<.001), and the old Wechsler Memory Scale Memory Quotient (F=5,27, df=1.27; p=.03). Treated TBI patients tended to perform timed psychometric tasks faster than TBI patients who did not undergo such treatment. After training, treated patients’ performance on the Digit Symbol Subtest of the WAIS also improved slightly (F=3.48, df=1,25; p=.07) as did the time they needed to complete the Tactual Performance Test with the nondominant hand (F=4.73, df=1,9; p=.06). Typically, patients showed greater changes on these measures before and after treatment com pared to untreated TBI patients. Speed of finger tapping tended to be higher in treated than in untreated TBI patients.
Some treated patients showed sustained improvement in speed and memory scores while others declined to the same level they were before they entered the neuropsychological rehabilitation program (Prigatano GP, unpublished data). The performance of a few patients as well as some of the control TBI patients actually deteriorated with time. Our overall impression was that several hours of repetitive cognitive exercises did not improve underlying higher cerebral functioning so much as it helped patients to use their residual abilities several years after their injury.
Patients’ emotional status, how ever, was substantially improved by the combination of activities conducted in the intensive neuropsychological rehabilitation program. Relatives, for example, reported that the attitude of treated patients was less negative than it had been before treatment. Their behavior was more reliable and the overall signs of their psychopathology declined.37 TBI patients who later were employed improved more on these various measures than unemployed TBI patients. Their scores on the WAIS Digit Symbol Subtest (F=10.39; df=1,15; p=.0006) especially improved as did their psychiatric status. The findings appeared to be reliable. Postacute patients with a history of moderate to severe TBI who were unemployed until they had undergone an intensive neuropsychological rehabilitation program often demonstrate improvements in mental energy and speed of information processing, and a reduction of psychopathological characteristics.
A later follow-up study with a larger group of brain dysfunctional patients treated in a similar intense neuropsychological rehabilitation program showed that patients who later returned to work were able to sustain a good working alliance with their therapists.38 To do so, it was important for patients to show reasonable judgment; to be able to control their emotional responses, particularly negative responses; and to be able to sustain their efforts when working in the real world albeit at a lower level than their preinjury job. Interestingly, a good working relationship with patients’ family strongly related to patients’ long-term outcomes.38
Impaired Self-Awareness and Finger Tapping
These clinical and research observations highlight two points. Many postacute TBI patients seem either to deny their disturbances in higher cerebral functioning or they are unaware of those disturbances, presumably because their higher cerebral functions are impaired.36 Second, the Halstead Finger Tapping scores of TBI patients with impaired self-awareness tend to be slow.33
Subsequent work has shown that speed of finger tapping is related to the severity of a TBI8,11 and correlates with patients’ capacity to return to part-time and full-time employment after injury.12 Speed of finger tapping has even been related to the achievement of inpatient neurorehabilitation goals after TBI and stroke.40
When normal individuals are asked to perform the Halstead Finger Tapping test, bilateral cerebral activation occurs as might be expected.14 Interestingly, however, as patients fatigue while performing this task (i.e., their tapping slows), bilateral activation of anterior brain structures increases. These structures correspond to what has been referred to as the “heteromodal cortex.”25 Patients with impaired self-awareness may have disturbances in heteromodal cortex.32,33
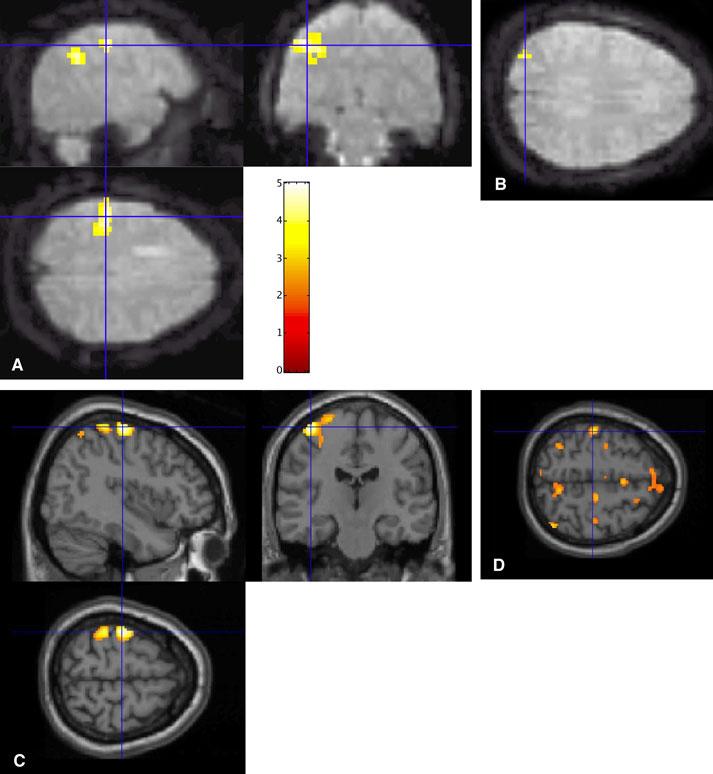
For example, in at least one patient with chronic impaired self-awareness, reduced speed of finger tapping was associated with decreased cerebral activation in anterior regions of the brain (Fig. 2). This patient, who sustained a TBI 12 years earlier, continues to show poor awareness of his cognitive and behavioral limitations, particularly in social situations. The speed of finger tapping with his left hand is slow, and MR imaging suggests the presence of bilateral cerebral contusions, particularly in the right frontal region. His pattern of activation while performing the Halstead Finger Tapping task is atypical compared to normal individuals. As predicted, he failed to show bilateral anterior cerebral activation.33
The anterior regions of the brain, especially the heteromodal regions thought to integrate “thinking” and “feeling,”31 have multiple connections with limbic inputs.25 This patient’s failure to show normal patterns of anterior cerebral activation while performing the Halstead Finger Tapping test may reflect disruption of the frontal-limbic connections. In particular, the disturbance of heteromodal regions of the brain may inevitably disturb both “cognition” and “personality.” Consequently, brain damage often produces disturbances in both dimensions, and recovery is marked by improvements in both cognitive and affective realms.
Improvement in Cognitive and Affective Functioning After Inpatient Neurorehabilitation
Cognitive and affective disturbances are not only important in postacute neuropsychological rehabilitation, they are also important in the rehabilitation of acute brain injuries. Prigatano and Wong41 compared the cognitive and affective disturbances of patients who achieved their rehabilitation goals within the first 30 days of a cerebrovascular accident or TBI to those who did not. Improvements in problem-solving skills were related to the achievement of rehabilitation goals but so were improvements in such dimensions as the ability to show affect spontaneously and to perceive facial affect accurately. Patients’ ability to predict their memory performance accurately, even on a brief screening test, correlated highly with the achievement of rehabilitation goals.
These findings suggest that early efforts at cognitive rehabilitation should focus not only on the traditional goals of improving cognitive functions like attention, memory, and language but should help patients to predict their performance more realistically and to respond to affective stimuli in a normal fashion. For example, patients could be helped to generate affect in their tone of voice, to perceive facial affect accurately, and to respond to humorous material in a manner similar to daily functioning. Collectively, the acute and postacute studies with brain dysfunctional patients indicate that working with both cognitive and affective functions is important to rehabilitation outcomes.
Conclusions
Collectively, the various studies and clinical experiences reported here lead to certain conclusions regarding the rehabilitation of patients with higher cerebral dysfunction. First, restoration of higher cerebral functions via retraining exercises, particularly after a severe TBI, is a slow, laborious process typically associated with marginal results. Dramatic improvements may primarily reflect compensatory strategies. Second, a patient’s personality not only influences the symptom picture after brain injury but also the degree that social integration may be possible. Third, a combination of individual and group rehabilitation experiences aimed at both cognitive and personality disorders is often necessary to optimize recovery and rehabilitation outcomes. Finally, focusing purely on cognitive deficits is often a mistake. This approach can lead to an inadequate understanding of the rehabilitation needs of patients with disturbances in higher cerebral functioning during both the acute and postacute stages after brain injury.
PET and fMR imaging may improve our understanding of recovery and deterioration phenomena after brain injury. Such advances are of considerable importance because successful neuropsychological rehabilitation is often a re-educational process that needs to consider individual patterns of cognition and personality.9 Consequently, individuals must be evaluated to determine what they can learn and how to present educational materials to maximize their learning experience. This process is often slow, and individuals must maintain their motivation to learn maximally. This educational process attempts to integrate insights from the neurosciences with insights obtained from the history of psychotherapy of individuals who have struggled with major losses in their life. Ultimately, the combination of these two bodies of knowledge will enhance understanding of the nature of the higher cerebral functions, how these functions change with time, and what may be done to facilitate their recovery.
References
- Ben-Yishay Y, Prigatano GP: Cognitive remediation, in Griffith E, Rosenthal M, Bond MR, et al (eds): Rehabilitation of the Adult and Child with Traumatic Brain Injury. Philadelphia: F.A. Davis, 1990, pp 393-409
- Brooks N, McKinlay W, Symington C, et al: Return to work within the first seven years of severe head injury. Brain Inj 1:5-19, 1987
- Cappa SF: Neuroimaging of recovery from aphasia. Neuropsychological Rehabilitation 10:365-376, 2000
- Carney N, Chesnut RM, Maynard H, et al: Effect of cognitive rehabilitation on outcomes for persons with traumatic brain injury: A systematic review. J Head Trauma Rehabil 14:277-307, 1999
- Corkin S, Rosen TJ, Sullivan EV, et al: Penetrating head injury in young adulthood exacerbates cognitive decline in later years. J Neurosci 9:3876-3883, 1989
- Cramer SC, Bastings EP: Mapping clinically relevant plasticity after stroke. Neuropharmacology 39:842-851, 2000
- DeLuca J, Cicerone KD: Confabulation following aneurysm of the anterior communicating artery. Cortex 27:417-423, 1991
- Dikmen SS, Machamer JE, Winn HR, et al: Neuropsychological outcome at 1-year post head injury. Neuropsychology 9:80-90, 1995
- Franz SI: Studies in re-education: The aphasias. Comparative Psychology 4:349-429, 1924
- Goldstein K: Aftereffects of Brain Injury in War. New York: Grune and Stratton, 1942
- Haaland KY, Temkin N, Randahl G, et al: Recovery of simple motor skills after head injury. J Clin Exp Neuropsychol 16:448-456, 1994
- Heaton RK, Chelune GJ, Lehman RA: Using neuropsychological and personality tests to assess the likelihood of patient employment. J Nerv Ment Dis 166:408-416, 1978
- Jensen J, Lindgren M, Meurling AW, et al: Dyslexia among Swedish prison inmates in relation to neuropsychology and personality. J Int Neuropsychol Soc 5:452-461, 1999
- Johnson SC, Prigatano GP: Functional MR imaging during finger tapping. BNI Quarterly 16(3):37-41, 2000
- Kime SK, Lamb DG, Wilson BG: Use of a comprehensive programme of external cueing to enhance procedural memory in a patient with dense amnesia. Brain Inj 10:17-25, 1996
- Klonoff PS, Lang DG, Henderson SW, et al: Outcome assessment after milieu-oriented rehabilitation: New considerations. Arch Phys Med Rehabil 79:684-690, 1998
- Kolb B: Recovery from occipital stroke: A self- report and an inquiry into visual processes. Can J Psychol 44:130-147, 1990
- Kozloff R: Network of social support and the outcome from severe head injury. J Head Trauma Rehabil 2:14-23, 1987
- Lashley KS: Factors limiting recovery after central nervous lesions. J Nerv Ment Dis 88:733-755, 1938
- Levin HS, Gary HE, Jr., Eisenberg HM, et al: Neurobehavioral outcome 1 year after severe head injury. Experience of the Traumatic Coma Data Bank. J Neurosurg 73:699-709, 1990
- Luria AR: Restoration of Function After Brain Trauma. Oxford: Pergamon, 1963
- Luria AR: The Man with a Shattered World. Cambridge, MA: Harvard University, 1972
- Merzenich MM, Jenkins WM, Johnston P, et al: Temporal processing deficits of language-learning impaired children ameliorated by training. Science 271:77-81, 1996
- Merzenich MM, Wright B, Jenkins W, et al: Cortical plasticity underlying perceptual, motor, and cognitive skill development: Implications for neurorehabilitation, in Cold Spring Harbor Symposia on Quantitative Biology (ed): Proceedings of the 1996 Symposium, Function & Dysfunction in the Nervous System. New York: CSHL Press, 1996
- Mesulam M-M: Principles of Behavioral Neurology. Philadelphia: F.A. Davis, 1985
- Musso M, Weiller C, Kiebel S, et al: Training-induced brain plasticity in aphasia. Brain 122:1781-1790, 1999
- NIH Consensus Development Panel on Rehabilitation of Persons with Traumatic Brain Injury: Rehabilitation of persons with traumatic brain injury. JAMA 282:974-983, 1999
- Nudo RJ, Wise BM, SiFuentes F, et al: Neural substrates for the effects of rehabilitative training on motor recovery after ischemic infarct. Science 272:1791-1794, 1996
- Perani D, Vallar G, Paulesu E, et al: Left and right hemisphere contribution to recovery from neglect after right hemisphere damage—an [18F] FDG PET study of two cases. Neuropsychologia 31:115-125, 1993
- Pizzamiglio L, Perano D, Cappa SF, et al: Recovery of neglect after right hemispheric damage: H2(15)O positron emission tomographic activation study. Arch Neurol 55:561-568, 1998
- Prigatano GP: Commentary: Beyond statistics and research design. J Head Trauma Rehabil 14:308-311, 1996
- Prigatano GP: Impaired awareness, finger tapping, and rehabilitation outcome after brain injury. Rehabilitation Psychology 44:145-159, 1999
- Prigatano GP: Principles of Neuropsychological Rehabilitation. New York: Oxford University, 1999
- Prigatano GP: Cognitive rehabilitation: An impairment-oriented approach embedded in a holistic perspective. BNI Quarterly 16(3):4-6, 2000
- Prigatano GP: Rehabilitation for traumatic brain injury. JAMA 284(14):1783, 2000
- Prigatano GP, Fordyce DJ, Zeiner HK, et al: Neuropsychological rehabilitation after closed head injury in young adults. J Neurol Neurosurg Psychiatry 47:505-513, 1984
- Prigatano GP, Fordyce DJ, Zeiner HK, et al: Neuropsychological Rehabilitation After Brain Injury. Baltimore: Johns Hopkins University, 1986
- Prigatano GP, Klonoff PS, O’Brien KP, et al: Productivity after neuropsychologically oriented, milieu rehabilitation. J Head Trauma Rehabil 9:91-102, 1994
- Prigatano GP, Smith KA: The role of psychotherapy in a neurological institute. BNI Quarterly 16:7-11, 2000
- Prigatano GP, Wong JL: Speed of finger tapping and goal attainment after unilateral cerebral vascular accident. Arch Phys Med Rehabil 78:847-852, 1997
- Prigatano GP, Wong JL: Cognitive and affective improvement in brain dysfunctional patients who achieve inpatient rehabilitation goals. Arch Phys Med Rehabil 80:77-84, 1999
- Richardson AJ, Stein JF: Personality characteristics of adult dyslexics, in Wright SF, Groner R (eds): Facets of Dyslexia and Its Remediation. New York: Elsevier Science, 1993, pp 411-424
- Salazar AM, Warden DL, Schwab K, et al: Cognitive rehabilitation for traumatic brain injury: A randomized trial. Defense and Veterans Head Injury Program (DVHIP) Study Group. JAMA 283:3075-3081, 2000
- Scherzer BP: Rehabilitation following severe head trauma: Results of a three-year program. Arch Phys Med Rehabil 67:366-374, 1986
- Tallal P, Miller SL, Bedi G, et al: Language comprehension in language-learning impaired children improved with acoustically modified speech. Science 271:81-84, 1996
- Weinstein EA, Marvin SL, Keller NJ: Amnesia as a language pattern. Arch Gen Psychiat (Chicago) 6:259-270, 1962
