
CT Angiography: A Tool for Managing Cerebral Aneurysms?
Matthew T. Walker, MD*
Jill Blackburn, RT
Cameron N. Nourani
Shahram Partovi, MD
Division of Neuroradiology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
*Current Address: Division of Neuroradiology, Northwestern Memorial Hospital, Chicago, Illinois
Abstract
The diagnosis of cerebral aneurysms has relied on conventional digital subtraction angiography (DSA) and magnetic resonance (MR) angiography. However, recent advances in computed tomographic (CT) angiographic technology now provide an alternative modality for the diagnosis of most aneurysms. This imaging modality has several advantages. The procedure can be performed faster and with less risk than that associated with conventional DSA. Reformatted and three-dimensional data provide morphologic information unavailable from DSA such as the relationship of the aneurysm to the clot, the presence of associated calcification, and bony landmarks. This article reviews the different imaging modalities used to diagnose and manage cerebral aneurysms and presents an evolving imaging algorithm that incorporates CT angiography, MR angiography, and DSA.
Key Words: aneurysms, computed tomographic angiography, digital subtraction angiography, magnetic resonance angiography
Intracranial aneurysms represent a significant health risk. They are reported in 1 to 2% of the general population; however, autopsy series identify aneurysms in more than 5% of the population.[11] With each hemorrhagic event, the morbidity and mortality rate associated with ruptured aneurysms is greater than 50%. More than half of all ruptured aneurysms rehemorrhage, the greatest percentage within the first 24 hours.[18] Diagnosis and management can be further complicated by multiple aneurysms in the same patient; between 20 to 30% of affected patients will have at least one other aneurysm.[17] Patients who do survive may have substantial complications related to vasospasm, hydrocephalus, and stroke.[10,25] Given this grim natural history, appropriate and timely treatment of cerebral aneurysms is crucial and relies heavily on diagnostic imaging.
The cornerstone diagnostic examination for intracranial aneurysms has been digital subtraction angiography (DSA). Magnetic resonance (MR) angiography has played a supportive role in some screening algorithms. Advances in computed tomography (CT) technology have reintroduced a third modality, CT angiography, which had remained largely a curiosity. The exact role of CT angiography in the diagnosis of cerebral aneurysms remains to be defined, but our experience suggests that its role in this diagnostic pathway will be important. This article reviews the angiographic modalities used to evaluate cerebral aneurysms, focusing on new developments in CT angiography. A new diagnostic algorithm for the evaluation of patients with acute nontraumatic subarachnoid hemorrhage (SAH) is presented.
Digital Subtraction Angiography
Much of the pioneering work in cerebral angiography was performed by Egas Moniz, a Portuguese physician best known for his Nobel prize-winning work on prefrontal lobectomy in psychiatric patients. At the Society of Neurology in Paris in July 1927, Moniz presented a series of angiographic experiments involving animals and humans. The patients were chronically ill with the diagnoses of general paralysis of the insane, postencephalitic parkinsonism, and brain tumors. Of the original nine patients, useful images were obtained from only two and one patient died from a complication. However, the potential for the technique was clear.[15]
Since then angiography has evolved considerably, and DSA is now available in most neuroradiology departments. Toxic contrast agents such as Thorotrast have been replaced with nonionic iodinated contrast. Filming capabilities also have improved. In the last 20 years enhancements to x-ray detectors for DSA have substantially improved image quality. DSA image resolution has increased by a factor of 4, from a 512×512 to a 1024×1024 pixel matrix, and the increased resolution permits smaller lesions to be identified. In concert with the increase in resolution, the response time of the x-ray detector–the minimum duration required to capture information–has decreased allowing more rapid filming speeds. Rapid filming is paramount for the dynamic evaluation of the cerebral circulation because it permits the construction of a “movie” that depicts how blood flows intracranially. This attribute is unique to DSA, a key feature that distinguishes DSA from all other forms of angiography, including CT angiography. Recently, three-dimensional (3D) reconstruction has become available; this format can be particularly useful when the neck of an aneurysm is difficult to define.
While DSA is considered the gold standard for the evaluation of the cerebral circulation, it still has limitations and associated risks.[6] These risks prevent the use of such an invasive test in screening algorithms and restrict its use to instances where the pretest probability of finding a lesion is high, such as in the presence of SAH. Even in this setting, for example, the procedure may fail to detect the source of acute SAH in as many as 5 to 15% of patients.[4,19] The false-negative rate of DSA is partially a reflection of its limitations, which include factors such as the presence of a thrombosed aneurysm, adjacent vasospasm, poor vessel opacification, and inadequate angiographic views. As an invasive technique, angiography is associated with complications, most importantly a risk of permanent stroke, which should be less than 1% at performing institutions as recommended by the American Society of Neuroradiology and the American Society of Interventional and Therapeutic Neuroradiology.[1] Complications related to the arterial puncture site and to the administration of iodinated contrast also can occur. Lengthy procedures performed in medically unstable patients outside an intensive care setting can further compromise patients’ safety. Moreover, angiographic examinations require considerable personnel–a radiologist, technologist, and nurse at minimum. In a busy angiography department, these requirements can contribute to significant time delays in completing an examination. Finally, in some geographic locations, deployment of a costly angiography suite and the required personnel may be financially prohibitive.
MR Angiography
Although the effects of flow in MR imaging were recognized as early as 1954, MR angiography did not become a useful clinical tool until the late 1980s. MR angiography now offers several advantages, the most important of which is the absence of inherent procedural risks to an appropriately selected patient population. Patients who have no MR-incompatible intracranial implants or pacemakers, who are independent of an MR-incompatible life support system, who have no intraocular metallic foreign bodies, and who can remain motionless for extended periods can safely enjoy a noninvasive evaluation of their intracranial vasculature to determine whether they have an aneurysm. Meeting these criteria is possible in an otherwise healthy outpatient being screened for aneurysms, such as those with polycystic kidney disease (PCKD), but is unlikely for acutely ill patients with nontraumatic SAH. These patients are likely to violate some if not most of the above prerequisites. Many such patients are intubated and on a respirator, most of which are MR-incompatible. If not intubated, patients may be agitated and unable to remain motionless for the time required to obtain useful images. Typically, they are unable to relate their medical history due to the acuity of their illness, and much needed critical screening information, such as the presence of a pacemaker, intraocular metallic foreign bodies, or any prior intracranial aneurysm clips, cannot be obtained. Patients with aneurysm clips must have the manufacturer’s information to identify their clip and to confirm its safety during an MR examination. Aneurysm clips produced before 1995 may be incompatible with MR imaging, and deaths related to the MR imaging of patients with clips have been reported.[12] Such stringent preimaging requirements hinder the use of MR imaging for the rapid evaluation of the intracranial vasculature in critically ill patients. As discussed below, few prerequisites and imaging speed are key elements that distinguish the utility of CT angiography from MR angiography in the noninvasive assessment of these patients.
CT Angiography
If an initial basic head CT identifies SAH, additional time is needed to transport and prepare the patient for evaluation by DSA. MR angiography is not used to assess patients with acute SAH. If it were, however, delays would occur because the patient would need to be screened for MR compatibility and then transported to the MR suite from the CT department. If intracranial hemorrhage and its source could be identified reliably in one combined test that required 5 minutes or less, the quality of care for patients with SAH would reach a level unparalleled by the current two-step process. Recent profound advances in CT technology approach this threshold. Supporting evidence and refined clinical algorithms, both of which are pursued aggressively at our institution, are now needed.
It is almost a misnomer to refer to current CT scanners by the same acronym used to describe first-generation CT scanners. Other than basic imaging principles, the two devices have little in common. During the last three decades, CT has advanced from a technique that required hours to obtain several slices centimeters in thickness to a study that requires only seconds and that provides submillimeter resolution over the entire brain.
The developments that propelled CT into the angiographic arena include helical (or spiral) scanning, improved x-ray generators, improved computing power, and the critical advent of multidetectors.[9] Each advance alone was insufficient; together, however, they transformed CT angiography into a strong alternative to DSA and MR angiography.[16,24] Multidetector helical CT scanners have increased scanning speed while improving image resolution and accuracy.[7] These technologically advanced machines make it possible to complete a CT angiogram of the brain in less than 30 seconds. Combined with the patient’s preangiographic head CT and any necessary postprocessing, both the brain parenchyma and its vasculature can be visualized completely in less than five minutes. The need for further patient transportation is eliminated as are the risks of an arterial puncture, vascular injury, stroke, and death in the case of DSA or the need for screening policies and precautions, MR-compatible life support equipment, and prolonged patient cooperation in the case of MR angiography. Yet, it would be imprudent to eliminate risk at the cost of compromising diagnostic accuracy. Like any other new modality, the risk-benefit ratio of CT angiography must be evaluated carefully.
Accuracy of CT Angiography
The accuracy of detecting cerebral aneurysms with CT angiography has paralleled advances in CT technology. Meta-analysis suggests that its sensitivity has increased from 61% before 1994 to 96% thereafter.[26] Rapid changes in technology quickly date published studies as new techniques become available. Although the most recent studies on CT angiography use helical CT, no data have yet been published on the use of multidetector helical CT for detecting aneurysms. Based on our prospective comparisons of more than 50 patients with nontraumatic SAH evaluated by both multidetector CT angiography and conventional DSA (unpublished data), multidetector helical CT likely will be found to offer even more sensitivity and specificity than currently reported.
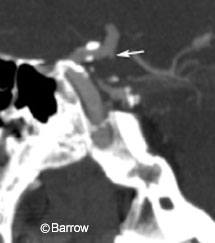
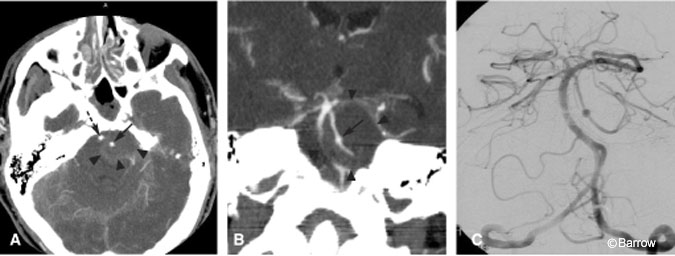
There is some reservation about how well CT angiography detects small aneurysms (Fig. 1).[3,13] This concern may be particularly important when asymptomatic subjects, a third of whom have aneurysms smaller than 5 mm, are screened.[27] Evidence from multiple series suggests that CT angiography is 96% sensitive for detecting aneurysms larger than 3 mm in diameter.[26] A recent study suggests that CT angiography can detect aneurysms as small as 1.7 mm and that prior failures might be explained by suboptimal technical parameters and image reconstruction.[14,24] Undoubtedly, ongoing advances in postprocessing and 3D reconstruction will have further implications for the sensitivity and specificity of CT angiography.[8,20] Such data help reassure surgeons that CT angiography can identify small lesions.
Diagnostic Screening for Aneurysms
MR angiography still plays a primary role in screening asymptomatic patients at risk for cerebral aneurysms such as those with PCKD or close relatives with aneurysms. Unlike MR angiography, which is completely noninvasive, CT angiography is not without risk and should be considered a relatively noninvasive examination. Whether CT angiography adds any significant sensitivity to warrant incurring the additional risks (i.e., the high rate of administration of intravenous contrast and exposure to radiation) in asymptomatic patients has yet to be determined. In this population, CT angiography is unlikely to displace MR angiography as the primary screening modality.
Once, however, an aneurysm is discovered in an asymptomatic patient via MR angiography, the next confirmatory examination may indeed become CT angiography rather than conventional DSA.[5,21] If the aneurysm is confirmed by CT angiography, the need for a DSA is debatable and perhaps should be limited to patients in whom specific information can only be obtained through DSA. After MR angiography, replacing DSA with CT angiography in asymptomatic patients helps to reduce their exposure to the risks associated with DSA. “Wedging” a less invasive test ahead of DSA has been an effective strategy in the past. For example, today MR angiography is performed after carotid ultrasonography instead of DSA as the confirmatory examination for stenosis of the internal carotid artery (ICA) when historically DSA served that purpose. Now, only confusing cases or patients with specific flow-related issues undergo DSA for final analysis of ICA stenosis.
Diagnostic Imaging of Nontraumatic Subarachnoid Hemorrhage
In patients with acute nontraumatic SAH, the pretest likelihood of a lesion is so high that the higher level of invasiveness associated with DSA has been tolerated for years. The new developments in CT technology, however, may alter tolerance for such invasiveness.
If CT angiography supplanted DSA as the primary examination for the evaluation of patients with acute SAH, at least one neurosurgical question would quickly arise: What is the appropriate course of action in the presence of SAH and a negative CT angiogram? In simpler terms, should a neurosurgeon rely on a negative CT angiographic examination to define the absence of an aneurysm? Given the impressive morbidity and mortality rates associated with this disease, the answer lies in the development of sound clinical and imaging algorithms to minimize or avoid false negatives. This issue will likely be debated for years just as it has been with DSA, which also cannot exclude an aneurysm definitively.
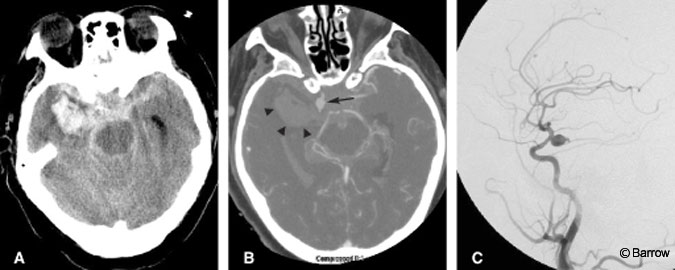
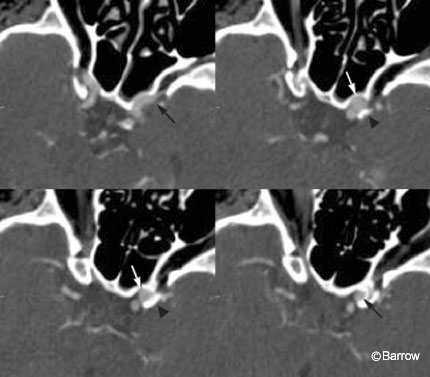
CT angiography should be viewed on the strength of its specificity in a high pretest likelihood population, such as patients with nontraumatic SAH. From this perspective, a patient with SAH but a negative CT angiogram would undergo DSA (a more sensitive test) to confirm the negative finding. In contrast, positive CT angiography that is concordant with a patient’s initial head CT identifying the location of the hemorrhage will likely prove sufficient for diagnosis and operative planning (Fig. 2).[22] In such patients, additional preoperative DSA is unlikely to yield additional information that would alter immediate treatment. Preoperatively, it would be reasonable to rely on multidetector CT angiography when the results are positive but to proceed to DSA if the results are negative.
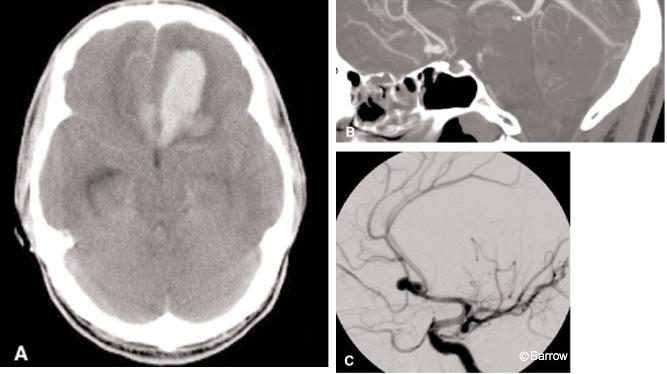
A positive CT angiogram may not necessarily indicate the site of hemorrhage. As many as 30%[17] of patients presenting to the emergency department may have multiple aneurysms. Consequently, an aneurysm identified on CT angiography may not have been the offending lesion. These patients may undergo unnecessary craniotomies while the true lesion remains untreated and at risk of immediate rehemorrhage. To avoid this potential complication, CT angiography should be compared with a patient’s initial noncontrast basic CT of the brain and the location of the SAH noted. The initial location of SAH on the head CT alone is a strong predictor of an aneurysm’s location. If an aneurysm is visualized on the CT angiogram where expected, surgical intervention could be considered with high confidence (Fig. 3). If the results of the CT angiography are incongruent with the basic CT, the patient should undergo DSA to exclude that the ruptured aneurysm was missed by the CT angiography.

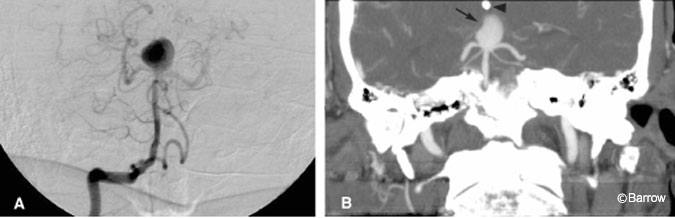
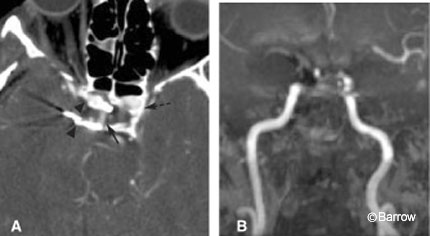
For patients with newly diagnosed aneurysms, CT angiography may offer unique advantages compared with DSA.[24] Its ability to identify the neck of the aneurysm may be useful when deciding between surgical or endovascular treatments. With 3D-CT angiography, an aneurysm can be rotated in multiple planes until the ideal projection is obtained (Fig. 4). Rotational DSA offers some of this information, but the availability of the technique is limited. Furthermore, CT depicts the thrombosed portion of the aneurysm (Fig. 5), mural calcifications, and adjacent clot (Fig. 6), all of which can complicate clip placement. Finally, identifying the relationship between osseous structures and the aneurysm can help to determine whether an aneurysm is intra- or extradural at the level of the cavernous sinus. This process requires that scanning be matched to a tight arterial bolus so that no venous blood in the cavernous sinus is present during scanning. If timed appropriately, paraophthalmic and cavernous sinus aneurysms can be visualized consistently on CT angiography (Fig. 7).
Postoperative Angiographic Evaluation of Aneurysm Clipping
Postoperative angiography may be indicated for aneurysms for a number of reasons. We often confirm the absence of residual aneurysm via DSA immediately after surgery. Possible vasospasm also can lead to an angiographic examination. After an aneurysm has been clipped, coincident disease process, such as cerebral ischemia, may require a vascular evaluation, typically by MR angiography. Either DSA or MR angiography can be used for long-term follow-up. CT angiography will also affect these diagnostic pathways.
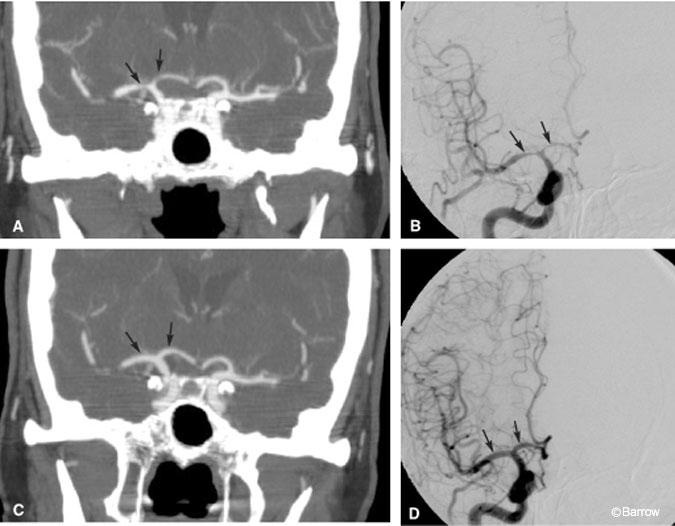
Historically, the presence of an aneurysm clip has been thought to preclude CT angiography because vascular structures could be obscured from the resultant beam-hardening artifact. Certain types of aneurysm clips, however, such as the Spetzler titanium clip, produce insignificant amounts of beam artifact (Fig. 8A). If the vessel of interest is near a titanium clip, CT angiography will better demonstrate the adjacent vascular structures than MR angiography, which is more susceptible to signal loss from metallic artifact (Fig. 8B).
Although MR angiography is the least invasive method for evaluating the cerebral vasculature after an aneurysm has been clipped, the study is immaterial if the area of concern is obscured by artifact. The signal loss associated with clips can extend beyond the clip and obscure vessels “downstream” from the clip itself. On MR angiography, for example, branches of the anterior cerebral artery distal to an anterior communicating artery clip may be attenuated because blood protons are dephased as they pass the metallic clip.
This artifact can simulate distal vasospasm. The risk associated with CT angiography is greater than that associated with MR angiography, but CT angiography is more likely to demonstrate a vessel of interest immediately adjacent to an aneurysm clip. Downstream artifact is not a problem with CT angiography.
CT can play a significant role in the postoperative evaluation for possible vasospasm. Not only can CT angiography help identify proximal vasospasm (Fig. 9) when coupled with CT perfusion imaging, it can identify the status of cerebral blood flow at a microcirculatory level (see Computed Tomography Perfusion Imaging, Illustrative Case 3). Although the ability of CT angiography to detect mild-to-moderate vasospasm of distal branches is uncertain,[2] this shortcoming can be overcome by combining the two techniques. Together, CT angiography and CT perfusion may substantially reduce the need for invasive DSA to assess and follow patients for postoperative vasospasm.
Whether CT angiography can identify residual aneurysms remains to be determined. Early data on the postoperative use of CT angiography to evaluate the placement of aneurysm clips and the presence of residual aneurysm are promising but limited.[23] We have evaluated more than 20 patients after clipping with both DSA and CT angiography, but the analysis is pending. Given that the postclipping probability of a residual aneurysm at our institution is small, it is uncertain whether CT angiography will be sensitive enough to identify residual lesions.
Evolving Diagnostic Protocol for Cerebral Aneurysms
In cooperation with our neurosurgical colleagues, we are reevaluating the diagnostic imaging protocol for patients with acute nontraumatic SAH. Undoubtedly, the recent advances in CT will introduce CT angiography into the diagnostic pathway of patients with cerebral aneurysms, both asymptomatic individuals and those with nontraumatic SAH. The key is to develop a diagnostic algorithm tailored to maintain the high specificity and sensitivity required to identify or exclude these lesions, which otherwise can be associated with devastating outcomes.
In patients with nontraumatic SAH, the classic diagnostic approach has been to obtain a noncontrast CT of the head. If positive, these patients are then screened with DSA. This pathway could be altered to insert CT angiography ahead of DSA. If a basic head CT is positive for SAH and the CT angiographic findings are consistent with location of the hemorrhage, the evidence is sufficient to consider direct operative intervention without obtaining a preoperative DSA. If findings on CT angiography are discordant with the location of the hemorrhage or if the CT angiogram is altogether negative in the presence of SAH, patients should undergo DSA.
Whether the postoperative use of CT angiography to determine the presence or absence of a residual aneurysm will be worthwhile is debatable. We do not advocate this use at this time. Once CT angiography is used as the primary diagnostic modality, a postoperative DSA evaluation for possible residual disease should be a full four-vessel study until more data on the sensitivity of CT angiography have been established. If, however, a relatively noninvasive assessment of the intracranial vasculature is indicated after aneurysm clipping (e.g., to exclude proximal vasospasm or thromboembolic disease), CT angiography is a less invasive choice than DSA. In some instances, CT angiography may offer more advantages than MR angiography even though it is slightly more invasive.
MR angiography continues to be the primary modality for screening asymptomatic patients at risk of cerebral aneurysms. Classically, these patients undergo DSA to confirm a lesion identified on MR angiography. However, a positive MR angiogram could be confirmed with CT angiography instead of DSA. If these two examinations, which rely on completely different imaging principles, are positive, the need for preoperative DSA should be questioned. If the CT angiographic findings are incongruent with the findings on MR angiography, patients should be evaluated by DSA as the gold standard.
Conclusion
In conjunction with DSA and MR angiography, CT angiography offers a quick and accurate means of detecting cerebral aneurysms. After using CT angiography intensively for almost 18 months, we have concluded that CT angiography will never wholly replace DSA or MR angiography. It will, however, affect almost every clinical algorithm used to identify cerebral aneurysms and their complications in at-risk populations and in individuals with nontraumatic SAH. The challenge is to test such new diagnostic paradigms carefully to ensure that quality of care is not compromised by blind enthusiasm for a new imaging modality. As data on the newer multidetector CT scanners emerge, such new diagnostic pathways will likely be increasingly accepted.
References
- American Society of Neuroradiology, American Society of Interventional and Therapeutic Neuroradiology, Society of Cardiovascular and Interventional Radiology: Quality improvement guidelines for adult diagnostic neuroangiography. Cooperative study between the ASNR, ASITN, and the SCVIR. AJNR Am J Neuroradiol 21:146-150, 2000
- Anderson GB, Ashforth R, Steinke DE, et al: CT angiography for the detection of cerebral vasospasm in patients with acute subarachnoid hemorrhage. AJNR Am J Neuroradiol 21:1011-1015, 2000
- Anderson GB, Steinke DE, Petruk KC, et al: Computed tomographic angiography versus digital subtraction angiography for the diagnosis and early treatment of ruptured intracranial aneurysms. Neurosurgery 45:1315-1320, 1999
- Bagley LJ, Hurst RW: Angiographic evaluation of aneurysms affecting the central nervous system. Neuroimaging Clin N Am 7:721-737, 1997
- Harrison MJ, Johnson BA, Gardner GM, et al: Preliminary results on the management of unruptured intracranial aneurysms with magnetic resonance angiography and computed tomographic angiography. Neurosurgery 40:947-957, 1997
- Heiserman JE, Dean BL, Hodak JA, et al: Neurologic complications of cerebral angiography. AJNR Am J Neuroradiol 15:1401-1411, 1994
- Hu H, He HD, Foley WD, et al: Four multidetector-row helical CT: Image quality and volume coverage speed. Radiology 215:55-62, 2000
- Johnson PT, Halpern EJ, Kuszyk BS, et al: Renal artery stenosis: CT angiography—comparison of real-time volume-rendering and maximum intensity projection algorithms. Radiology 211:337-343, 1999
- Kalender WA, Seissler W, Klotz E, et al: Spiral volumetric CT with single-breath-hold technique, continuous transport, and continuous scanner rotation. Radiology 176:181-183, 1990
- Kassell NF, Torner JC, Haley EC Jr, et al: The International Cooperative Study on the Timing of Aneurysm Surgery. Part 1: Overall management results. J Neurosurg 73:18-36, 1990
- King JT Jr: Epidemiology of aneurysmal subarachnoid hemorrhage. Neuroimaging Clin N Am 7:659-668, 1997
- Klucznik RP, Carrier DA, Pyka R, et al: Placement of a ferromagnetic intracerebral aneurysm clip in a magnetic field with a fatal outcome. Radiology 187:855-856, 1993
- Korogi Y, Takahashi M, Katada K, et al: Intracranial aneurysms: Detection with three-dimensional CT angiography with volume rendering—comparison with conventional angiographic and surgical findings. Radiology 211:497-506, 1999
- Liang EY, Chan M, Hsiang JH, et al: Detection and assessment of intracranial aneurysms: Value of CT angiography with shaded-surface display. AJR Am J Roentgenol 165:1497-1502, 1995
- Morris P: Practical Neuroangiography. Philadelphia: Lippincott Williams & Wilkins, 1997
- Napel S, Marks MP, Rubin GD, et al: CT angiography with spiral CT and maximum intensity projection. Radiology 185:607-610, 1992
- Nehls DG, Flom RA, Carter LP, et al: Multiple intracranial aneurysms: Determining the site of rupture. J Neurosurg 63:342-348, 1985
- Pakarinen S: Incidence, aetiology, and prognosis of primary subarachnoid haemorrhage. A study based on 589 cases diagnosed in a defined urban population during a defined period. Acta Neurol Scand 43:1-28, 1967
- Pathirana N, Refsum SE, McKinstry CS, et al: The value of repeat cerebral angiography in subarachnoid haemorrhage. Br J Neurosurg 8:141-146, 1994
- Ruigrok YM, Rinkel GJ, Buskens E, et al: Perimesencephalic hemorrhage and CT angiography: A decision analysis. Stroke 31:2976-2983, 2000
- Schwartz RB, Tice HM, Hooten SM, et al: Evaluation of cerebral aneurysms with helical CT: Correlation with conventional angiography and MR angiography. Radiology 192:717-722, 1994
- Velthuis BK, Van Leeuwen MS, Witkamp TD, et al: Computerized tomography angiography in patients with subarachnoid hemorrhage: From aneurysm detection to treatment without conventional angiography. J Neurosurg 91:761-767, 1999
- Vieco PT, Morin EE 3rd, Gross CE: CT angiography in the examination of patients with aneurysm clips. AJNR Am J Neuroradiol 17:455-457, 1996
- Villablanca JP, Martin N, Jahan R, et al: Volume-rendered helical computerized tomography angiography in the detection and characterization of intracranial aneurysms. J Neurosurg 93:254-264, 2000
- Wardlaw JM, White PM: The detection and management of unruptured intracranial aneurysms. Brain 123:205-221, 2000
- White PM, Wardlaw JM, Easton V: Can noninvasive imaging accurately depict intracranial aneurysms? A systematic review. Radiology 217:361-370, 2000
- Zager EL: Surgical treatment for intracranial aneurysms. Neuroimaging Clin N Am 7:763-782, 1997
