
Affective Disturbances After Acute, Severe Traumatic Brain Injury as Measured by the BNI Screen for Higher Cerebral Functions
Susan R. Borgaro, PhD
George P. Prigatano, PhD
Division of Neurology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
This study investigated affective functioning in patients with severe traumatic brain injury (TBI) during the first 60 days after their injury using the BNI Screen for Higher Cerebral Functions (BNIS). The performances of 21 adult patients with severe TBIs, as determined by admitting Glasgow Coma Scale score and neuroimaging findings, were compared to those of 21 normal controls. The Affect subtest of the BNIS measures the ability to express affect in tone of voice, to perceive facial affect, to control affect, and to generate spontaneous affect. To control for educational differences between the groups, an analysis of covariance was computed for each item. The controls performed significantly better on affect perception and spontaneity than the patients with severe TBIs (p<0.001). The affect perception and spontaneity items were entered into a stepwise discriminant function analysis. Both were discriminant in classifying the groups 88% of the time. These findings suggest that the abilities to perceive and generate affect may be especially impaired soon after a severe TBI and can be measured by the BNIS.
Key Words: affect, brain injury, neuropsychology
The BNI Screen for Higher Cerebral Functions (BNIS) was developed to sample a wide range of disturbances in brain dysfunctional patients in a brief but reliable (and valid) manner. Since the BNIS was developed in 1991,[5] a series of studies have documented its validity[5,7,9,13,14] and reliability.[12] Studies also have focused on the clinical utility of assessing both the qualitative and quantitative information the BNIS provides when patients with dementia of the Alzheimer’s type[10] and patients who present with specific neuropsychological disturbances such as constructional dyspraxia and hemi-inattention are assessed.[1]
A unique feature of the BNIS is that it directly assesses disturbances in affect. Prigatano and Wong[11] demonstrated that both cognitive and affective disturbances as measured by the BNIS related to the achievement of rehabilitation goals during the acute stages after a traumatic brain injury (TBI) or cerebrovascular accident. In a heterogenous group of acute brain dysfunctional patients, 40% demonstrated difficulties in perceiving facial affect on the BNIS compared to only 3% of normal controls.[15]
The present investigation analyzes the performance of severely impaired patients with a TBI on the four dimensions of affect measured by the BNIS: the ability to generate affect in one’s tone of voice upon request, the ability to perceive facial affect, the ability to demonstrate spontaneous affect when confronted with an incongruent stimulus, and the ability to control affective responses when examined. All patients were studied during the acute stage of their injury and were able to participate in this basic screening test.
Method
Subjects
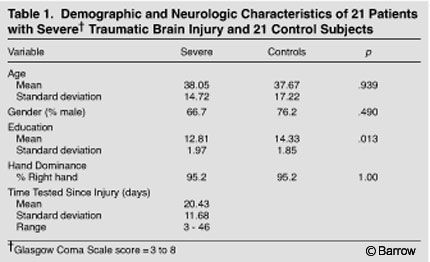 The experimental subjects were 21 patients with an admitting diagnosis of cerebral concussion or contusion at St. Joseph’s Hospital and Medical Center in Phoenix, Arizona (Table 1). These 21 patients were classified as having a severe injury based on their diagnostic imaging studies and admitting Glasgow Coma Scale (GCS) scores,[2] which were obtained in the emergency room. All patients were referred for a clinical neuropsychological evaluation as a part of their neurological and rehabilitation evaluation. The five inclusion criteria were as follows: Participants were within 60 days of their documented TBI. Their admitting GCS score and neuroimaging studies qualified the severity of their injury. Participants were able to take a brief screening test as part of their clinical evaluation.
The experimental subjects were 21 patients with an admitting diagnosis of cerebral concussion or contusion at St. Joseph’s Hospital and Medical Center in Phoenix, Arizona (Table 1). These 21 patients were classified as having a severe injury based on their diagnostic imaging studies and admitting Glasgow Coma Scale (GCS) scores,[2] which were obtained in the emergency room. All patients were referred for a clinical neuropsychological evaluation as a part of their neurological and rehabilitation evaluation. The five inclusion criteria were as follows: Participants were within 60 days of their documented TBI. Their admitting GCS score and neuroimaging studies qualified the severity of their injury. Participants were able to take a brief screening test as part of their clinical evaluation.
Participants had no history of prior TBI or significant psychiatric illness. Participants spoke English as their primary language. Of the 21 patients with severe TBIs, 14 (67%) had space-occupying lesions as reflected by computed tomography (CT) and magnetic resonance (MR) imaging (which were recorded but not deemed necessary to classify patients as having sustained a severe TBI).
Of these 21 patients, 21% had bilateral lesions, 21% had left hemisphere lesions, and 29% had right hemisphere lesions. Another 29% had a space-occupying lesion in the brain stem, cerebellum, or unspecified location. The most common lesions were contusions (61%), hemorrhages (26%), and hematomas (13%).
Twenty-one control subjects, who were initially recruited as a part of the standardization study of the BNIS,[8] also were administered the BNIS (Table 1). Control subjects reported no history of psychiatric or neurologic disease.
Procedures
Patients were evaluated within the first 60 days of their injury. All subjects were interviewed and administered the BNIS by either a staff clinical neuropsychologist or a postdoctoral resident in clinical neuropsychology. Performance on the Affect subtest was examined.
Neuropsychological Instrument
The BNIS requires 10 to 25 minutes to administer and yields a total score (range of raw scores=3 to 50 points) and seven subtest scores. The subtests are Speech/Language, Orientation, Attention/Concentration, Visuospatial and Visual Problem Solving, Memory, Affect, and Awareness vs Performance.[8] In addition to the seven subtest items, three prescreening items (level of consciousness/alertness, basic language, level of cooperation) that contribute to the BNIS total score but not to any of the subtest scores are administered. The BNIS is not administered if the patient’s alertness, language ability, or cooperation on the prescreening items is inadequate. Total raw scores are converted to age-corrected standard (T) scores using the norms of Prigatano and colleagues.[8]
The Affect subtest used in this study consists of four items. Each item is scored “1” if the patient performs the item correctly. Higher scores indicate a higher level of functioning. The first item requires the subject to read aloud the sentence, “They won the game.” Patients are then asked to read the sentence in a happy tone of voice followed by an angry tone of voice. They receive a score of 1 if they are able to generate happy and angry tones in their voice when reading the sentence.
The second item assesses the ability to perceive facial affect. Patients are shown a card with three types of facial expressions. The first picture depicts a feeling of anger. The second picture depicts a feeling of happiness/joy. The third picture depicts a feeling of either fear or surprise. The subject is asked to identify verbally the emotion expressed by each face. They receive full credit if they correctly identify all three facial expressions.
The third item is designed to assess the ability to control affect. This item is assessed throughout the evaluation. A score of 0 is assigned if the subject demonstrates an inability to inhibit inappropriate comments (e.g., swears, rapidly gets angry) or has trouble controlling tone or volume when expressing affect. This item is judged by the examiner as normal or abnormal with a score of 1 for the former and 0 for the latter.
The fourth item assesses the ability to generate affect spontaneously. Patients are shown a picture of a “no smoking” sign and asked what it means. They are then shown a picture of a “no lemon” sign and asked what it means. They receive a score of 1 if they laugh, giggle, or reflexively smile in response to this item; otherwise, they receive a score of 0.
Results
The TBI and control groups did not differ in terms of age (t=-0.08, p=0.939) or gender (c2=0.47, p=0.490). The control group, however, was significantly more educated than the TBI group (t=2.59, p=0.013; Table 1). Consequently, analyses of covariance (ANCOVAs) were performed to control for the potential effect of education on affect performance.

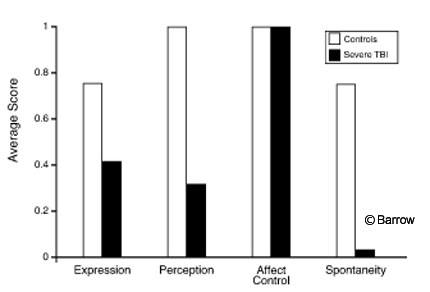
The performance of the group with severe TBIs was significantly worse than that of the controls on the Total Affect subtest score [F(1,40)=74.91, p=0.000, Table 2]. The difference in Total Affect scores remained robust when education was controlled statistically [F(1,40)=60.21, p=0.000]. The group with severe TBIs performed significantly worse than controls on affect perception [F(1,40)=41.57, p=0.000] and spontaneity [F(1,40)=33.58, p=0.000]. The performance of these acute and severely injured patients did not differ significantly from that of the controls on the affect expression or control items (Fig. 1).
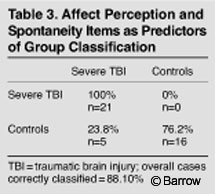 A stepwise discriminant function analysis was performed to determine how well affect perception and spontaneity differentiated the TBI group from the control group.
A stepwise discriminant function analysis was performed to determine how well affect perception and spontaneity differentiated the TBI group from the control group.
Wilks lambda yielded 88% correct classification with the affect perception and spontaneity items distinguishing the two groups. The spontaneity item entered the equation first (F=45.0, p=0.000) followed by the affect perception item (F=39.0, p=0.000, Table 3).
Discussion
Although aspontaneity has been reported after severe bifrontal brain injuries for many years,[4] it has been difficult to study this phenomenon experimentally. Luria[3] noted that many patients with bilateral frontal lobe injuries did not report “mental tension” and believed it was the basis of some of the difficulties patients had with problem solving. The present study demonstrates that normal subjects easily show spontaneous affect when exposed to an incongruent stimulus. In contrast, patients with severe TBI often fail to do so. The BNIS attempts to directly elicit this behavior (or behavioral failure), which is often observed while patients are on rehabilitation units.
Interestingly, however, these severely impaired TBI patients were only marginally different from normal controls in terms of generating happy and angry tones of voice when reading a sentence. Thus, they specifically appeared to have problems in the spontaneous generation of affect but not necessarily in demonstrating affect when requested.
In the present study, TBI patients had notable difficulties in the perception of facial affect. This finding replicates an earlier observation.[15] Many of the judgment and perceptual problems that brain-injured patients demonstrate relate to failures in both cognitive and affective functioning. This component needs to be addressed to understand patients’ phenomenological experience and how to approach them in a rehabilitation setting.[6]
Somewhat paradoxically, the present group of severely brain-injured patients showed no difficulties in controlling emotion while undergoing testing. Clinical experience shows that many severely impaired TBI patients are agitated and disinhibited during testing. The present finding is therefore unexpected. In this study, patients tolerated being examined with a brief screening test. Hence, they do not represent all TBI patients seen in our inpatient rehabilitation or acute neurosurgical and neurological wards. The patients included in this study were those who could tolerate such testing. Clinically, these patients often appear to show flatness of affect and are cooperative, most likely because of widespread cognitive impairment. If these patients were studied serially over time, many of them might exhibit disinhibited behavior and their performance on the control items might differ from that of normal individuals.
The BNIS attempts to sample dimensions that are important for adaptation, rehabilitation, and discharge planning. Unlike other screening tests, the BNIS specifically includes dimensions of affective functioning as well as cognitive functioning. Not only is this dimension important for rehabilitation outcomes,[11] it is important to obtaining a differential diagnoses.[13]
References
- Hahn S, Prigatano G: Errors copying a simple pattern on the BNI Screen for Higher Cerebral Functions. BNI Quarterly 16:28-31, 2000
- Jennett B, Teasdale G: Management of Head Injuries. Philadelphia: F.A. Davis, 1974
- Luria A: Restoration of Function After Brain Trauma (in Russian). Moscow and Pergamon, London: Academy of Medical Science, 1948
- Ota Y: Psychiatric studies on civilian head injuries, in Walker A, Caveness W, Critchley M (eds): The Late Effects of Head Injury. Springfield, IL: Charles C Thomas, 1969, pp 110-119
- Prigatano G: BNI Screen for Higher Cerebral Functions: Rationale and initial validation. BNI Quarterly 7:2-9, 1991
- Prigatano G: Principles of Neuropsychological Rehabilitation. New York: Oxford University, 1999
- Prigatano G, Amin K, Rosenstein L: Validity studies on the BNI Screen for Higher Cerebral Functions. BNI Quarterly 9:2-9, 1992
- Prigatano G, Amin K, Rosenstein L: Administration and Scoring Manual for the BNI Screen for Higher Cerebral Functions. Phoenix, AZ: Barrow Neurological Institute, 1995
- Prigatano G, Palencia B: Halstead Impairment Index, Wechsler IQ, and the BNI Screen for Higher Cerebral Functions. BNI Quarterly 16:22-27, 2000
- Prigatano G, Smason I: Assessment of Alzheimer’s patients with the BNI Screen for Higher Cerebral Functions. BNI Quarterly 10:30-35, 1994
- Prigatano G, Wong J: Cognitive and affective improvement in brain dysfunctional patients who achieve rehabilitaion goals. Arch Phys Med Rehabil 80:77-84, 1999
- Rosenstein L, Prigatano G, Amin K: Reliability studies for the BNI Screen for Higher Cerebral Functions. BNI Quarterly 8:24-28, 1992
- Rosenstein L, Prigatano G, Nayak M: Differentiating patients with higher cerebral dysfunction from patients with psychiatric or acute medical illness using the BNI Screen for Higher Cerebral Functions. Neuropsychiatry Neuropsychol Behav Neurol 10:113-119, 1997
- Wass P: An analysis of the construct-related and ecological validity of the Barrow Neurological Institute Screen for Higher Cerebral Functions. Doctoral Dissertation. 1997, University of Windsor, Ontario, Canada
- Zigler L, Prigatano G: Perception of facial affect after acute brain injury. BNI Quarterly 16:32-36, 2000
