
What Causes Normal Pressure Hydrocephalus?
Harold L. Rekate, MD
Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
Originally described in the mid1960s, normal pressure hydrocephalus (NPH) is a condition found mainly in elderly patients. Enlargement of the cerebral ventricles results in insidious deterioration of gait, bladder control, and cognitive function without overt signs of increased intracranial pressure. The cause of the condition has been the subject of considerable speculation, and a consensus remains elusive. This article evaluates the relevant clinical information and basic science to develop a unifying theory of the cause of NPH. The findings suggest that a long-standing increase in and concomitant prolonged compensation for resistance to the flow of cerebrospinal fluid from its point of production to its points of absorption may be the mechanism underlying NPH. The normal process of aging can lead to softening of the brain or to a decrease in brain turgor, and this chronic compensated condition likely leads to decompensation and symptomatic deterioration.
Key Words: cerebrospinal fluid, dementia, intracranial pressure, mathematical modeling, normal pressure hydrocephalus, pathophysiology, pseudotumor cerebri
In 1965 Hakim and Adams originally described normal pressure hydrocephalus (NPH) as a surgically treatable cause of dementia.[1,16] Elderly patients with dementia and enlarged ventricles on pneumoencephalograms were subjected to the placement of ventriculoatrial shunts. The cognitive performance of some of these patients improved dramatically, but the size of their cerebral ventricles changed in only a few. In these early studies, none of the patients showed any signs of increased intracranial pressure (ICP), and the name NPH was coined to describe this condition. Since then a large volume of literature has attempted to explain this enigmatic condition and to improve the ability to select patients for treatment. Applying relevant information obtained from mathematical modeling, basic science research and neurophysiology, neuropathology, and neuroradiology, this review proposes a unifying theory about the underlying cause or causes of NPH.
Mathematical Modeling
In collaboration with the engineering faculty at Case Western Reserve University, Cleveland, Ohio, I developed a mathematical model of the dynamics of cerebrospinal fluid (CSF) to explain a variety of enigmatic conditions related to the volume of the cerebral ventricles as a function of resistance to outflow of CSF.[35] This model used mathematical equations derived from the engineering principals of hydraulics and incorporated methodologies from system and design engineering. From these equations a computer simulation was developed to describe changes in the volumes of the cerebral ventricles as a function of CSF pressure and flow. This work was supported by a grant from the National Institutes of Neurological Diseases and Stroke (R01-NS22901).
To describe the response of the cerebral ventricles to an increase in resistance to CSF outflow without a significant increase in ICP in the condition of NPH and to account for the lack of an increase in ventricular size with increases in outflow resistance and increased ICP in pseudotumor cerebri, it was necessary to include a modifier termed “brain turgor” or Kb.[35] Originally, this term was conceived of as a constant for each person’s brain, and the term Kb was used for “brain constant.” Studies of pseudotumor cerebri, however, indicated that brain turgor is not always a constant. Rather, it can be a rapidly changing variable that depends on cerebral venous volume.[19]
In the case of pseudotumor cerebri, the predictions of the model were validated by measuring pressure in the venous sinuses, and the cause of pseudotumor cerebri was identified as an increase in sagittal sinus pressure. Actually, two different forms of this condition were identified. In the more common case of pseudotumor cerebri associated with obesity in young women, the increased pressure in the sagittal sinus reflected increased right atrial pressure, which responded to weight loss. In nonobese sufferers of pseudotumor cerebri, gradients were identified across partially occluded venous sinuses, even in patients with normal magnetic resonance (MR) venographic studies. Consequently, in these cases, pseudotumor cerebri resulted from an increase in K[b] and a decrease in the absorption of CSF due to high sagittal sinus pressure being transmitted into the parenchyma of the brain.
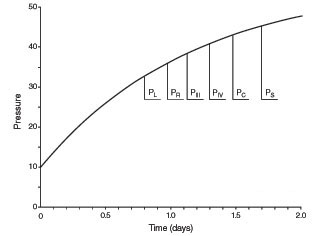
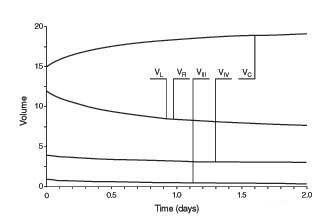
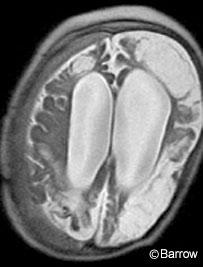
NPH was modeled by making two assumptions. First, turgor of the brain diminishes as a result of the normal process of aging. Second, as suggested by Di Rocco and coworkers in a study discussed in the section on the pathology of NPH,[13] resistance to the flow of CSF between the spinal subarachnoid and the cortical subarachnoid spaces must be increased. Computer simulations of the effect on ventricular volumes (Fig. 1) and pressure (Fig. 2) show an increase in the volumes of the cerebral ventricles, particularly the lateral ventricles, and a very slight increase in ICP that quickly stabilizes with time. The model successfully predicts the behavior of the brain in the case of NPH. Aging is not the only condition that leads to a decrease in brain turgor. Brain turgor also decreases after the brain is injured by stroke, trauma (Fig. 3), and radiation therapy. The NPH in such cases has a distinct cause and is distinct from idiopathic NPH in which the cause is unknown.
For the purposes of illustration and study, blockage between the spinal and cortical subarachnoid spaces was chosen for the computer simulation. A similar outcome is predicted if the point of obstruction is located at the outlet foramina of the fourth ventricle or at the aqueduct of Sylvius. In animal models and in human subjects, it is easy to increase brain turgor and thus to create pseudotumor cerebri by impeding venous return of the jugular veins. Simulation of NPH, however, is more difficult and takes considerably longer. The study of NPH therefore demands analysis of naturally occurring examples of the condition. The remainder of this review analyzes the available clinical and basic science information as it affects the model described here.
Animal Models
Since the early work of Dandy, chronic hydrocephalus has been produced in a variety of animal models. There are also a number of naturally occurring examples of congenital hydrocephalus in inbred species of rats and mice.[10,11,18,24,28,30] When ICP is measured in these animals during the chronic phase of hydrocephalus, it is usually within a normal range. In a canine model, hydrocephalus is produced by the intracisternal injection of a slurry of clay called kaolin. During the first few days after the kaolin is injected, ICP increases greatly. However, ICP rapidly returns to normal with the marked expansion of the ventricles.[26] The ventricles of the naturally occurring H-Tx rat expand. These rats fail maze testing without overt signs of increased ICP, thus simulating NPH. Of rats whose ventricles maintain the ability to decrease, their function on motor testing improves until their performance is indistinguishable from that of their nonhydrocephalic littermates.[29,37]
Pathological Studies
The histological appearance of biopsies from patients with NPH shows no significant differences compared to postmortem samples from age-matched controls.[3,9,12,21-23] On MR imaging and pathological examination, periventricular vacuolization appears to be an incidental finding that increases in severity and extent with aging.[2] These findings reflect the sponge-like quality of the brain in later decades of life. This sponge-like quality leads to a decrease in the turgor of the brain with age.
A number of articles have focused on the parenchymal changes in the brains of patients diagnosed with NPH. However, I found only one article in which postmortem material was studied in an attempt to determine the point of obstruction to the flow of CSF. Di Rocco and colleagues studied the brains of three patients shown to have NPH. These specimens revealed significant leptomeningeal fibrosis, suggesting that the point of obstruction could be the basal cisterns between the spinal and cortical subarachnoid spaces.[13] These findings are consistent with the association of NPH with head injury or aneurysmal subarachnoid hemorrhage.
In summary, the findings from available pathologic studies on the parenchyma of the brain itself suggest only the presence of the hallmarks of senescence. There are no findings specific for NPH. There is some pathologic basis for the thought that as we age our brains become softer. Finally, shunt responsiveness seems to be associated with fibrosis and therefore increased resistance to the flow of CSF in the basal cisterns.
Clinical Evaluation
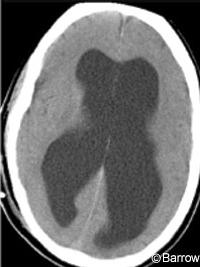
Insight into the origin of NPH often can be gained from the history and physical examination of afflicted patients. A significant but unknown number of patients who develop the classical findings of NPH late in life have a history of always having been aware that their heads were larger than normal (Fig. 4). Vanneste and Hyman have shown that a significant proportion of patients with NPH has an obstruction at the level of the aqueduct (noncommunicating hydrocephalus). This finding strengthens the argument that patients with NPH have long-standing chronic compensated hydrocephalus from which they then decompensate as they age.[39]
Clinical Physiologic Studies
Chronic monitoring of ICP reveals that NPH is actually a misnomer. During ICP monitoring, patients who eventually respond to shunting show intermittent elevations of ICP. Their baseline ICP recordings are normal to high normal. These patients also have plateau waves in greater frequency and duration than normal individuals or than patients who have brain atrophy. [15,32-34,36,40] These studies support the contention that these patients have higher ICP than would be normal for patients of their same age with the same brain viscoelastic properties.
Infusion studies that measure the resistance to outflow of CSF (Rout) are purported to improve the diagnostic accuracy of the clinical and radiological examinations of these patients.[4-8,14,20] These studies involve the infusion of artificial CSF while recording ICP. This test can be performed in a variety of ways, but 18 ml/mm Hg/min seems to be an established threshold for treatment. Patients whose Rout is lower than that level are less likely to respond to treatment than patients with a Rout higher than that level. This test confirms the obstructive nature of NPH and that the point of obstruction is distal to the spinal subarachnoid space. The point of obstruction therefore must be between the spinal and cortical subarachnoid spaces, or at the level of the arachnoid villi. This test also has been advocated to distinguish between “communicating” and “noncommunicating” hydrocephalus. Patients with the latter are potential candidates for endoscopic third ventriculostomy.[27]
Radiographic Studies
A large number of radiographic studies have been purported to aid in the selection of patients for shunting in the context of NPH. There is general agreement that none of these studies alone is sensitive or specific enough to allow the outcome of shunting to be predicted with an acceptable level of confidence. Two studies, however, cast significant light on the pathophysiologic mechanisms involved in NPH. The first reflects that the degree of ventricular distention should be significantly greater than the amount of CSF in the cortical subarachnoid spaces. This relationship suggests that the brain has been pushed outward to fill the volume of the subarachnoid space. It implies that there is an obstruction to CSF flow between the ventricular system and the cortical subarachnoid space.[25]
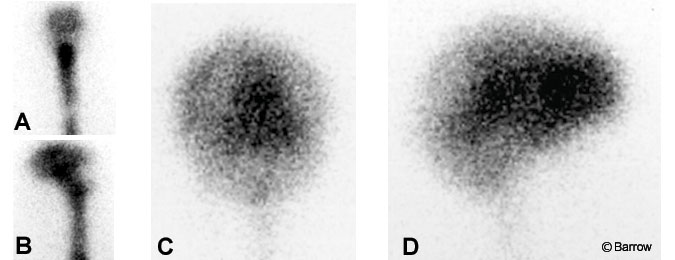
Second, for many years, radionuclide cisternography has been recommended for the diagnosis of NPH. Various protein-bound tracers have been used, including RI131SA and indium-111.[17,38] The technique is the same for both tracers. A lumbar puncture is performed, and the tracer is injected into the spinal subarachnoid space. Scintigraphic scans are obtained at intervals from 3 hours to 72 hours after injection of the tracer. In a positive study (Fig. 5), the dye quickly travels into the ventricular system, remains there for prolonged periods, and clears slowly. A negative study suggests that the patient is unlikely to respond to shunting. In this case, the dye enters the ventricle rather quickly and quickly flows into the cortical subarachnoid space over the convexity of the brain, but it clears within 48 to 72 hours. A positive test implies that a partial block to CSF flow is present between the spinal and cortical subarachnoid spaces. This study is consistent with the pathologic findings of the Di Rocco study described earlier.[13] This test helps illuminate the condition as a form of obstruction to CSF flow. However, its use has almost been abandoned because it does not appear to enhance the selection of patients for treatment beyond that provided by the combination of the patient’s clinical picture and imaging studies.[8]
Conclusion
Based on the studies discussed above, NPH can be understood as a problem that occurs in two phases. In the first phase a pathologic condition develops that impairs the absorption of CSF at some point between its point of origin within the ventricular system and its point of absorption at the level of the arachnoid villi. During this phase, the ventricles may or may not enlarge. If the pathology develops before the cranial sutures fuse, significant macrocephaly may result. The patient compensates fully for this CSF absorptive problem and remains without overt symptoms for a long period, possibly many decades. The point of obstruction can be at the level of the aqueduct of Sylvius, outlet foramen of the fourth ventricle, or basal cisterns.
At some point, patients begin to deteriorate. In young or middle-aged adults, the deterioration often follows a minor head injury.[31] Such patients usually become symptomatic with a mixed picture of high ICP and the characteristic findings of NPH. In addresses to conventions of the Hydrocephalus Association, Williams has referred to this condition as SHYMA (symptomatic hydrocephalus of young and middle-aged adults; Williams MA, personal communication, 1999, 2001). If deterioration does not occur at this stage, patients will begin to show subtle signs of deterioration as they reach senescence. This phase may begin as early as age 60. This phase results from the new expansion of the ventricles as the brain softens, an inevitable part of the normal process of aging.
References
- Adams RD, Fisher CM, Hakim S, et al: Symptomatic occult hydrocephalus with “normal” cerebrospinal fluid pressure. A treatable syndrome. N Eng J Med 273:117-126, 1965
- Awad IA, Spetzler RF, Hodak JA, et al: Incidental lesions noted on magnetic resonance imaging of the brain: Prevalence and clinical significance in various age groups. Neurosurgery 20:222-227, 1987
- Bech RA, Juhler M, Waldemar G, et al: Frontal brain and leptomeningeal biopsy specimens correlated with cerebrospinal fluid outflow resistance and B-wave activity in patients suspected of normal-pressure hydrocephalus. Neurosurgery 40:497-502, 1997
- Boon AJ, Tans JT, Delwel EJ, et al: Does CSF outflow resistance predict the response to shunting in patients with normal pressure hydrocephalus? Acta Neurochir Suppl (Wien) 71:331-333, 1998
- Borgesen SE: Conductance to outflow of CSF in normal pressure hydrocephalus. Acta Neurochir (Wien) 71:1-45, 1984
- Borgesen SE, Gjerris F: The predictive value of conductance to outflow of CSF in normal pressure hydrocephalus. Brain 105:65-86, 1982
- Borgesen SE, Gjerris F: Relationships between intracranial pressure, ventricular size, and resistance to CSF outflow. J Neurosurg 67:535-539, 1987
- Borgesen SE, Westergard L, Gjerris F: Isotope cisternography and conductance to outflow of CSF in normal pressure hydrocephalus. Acta Neurochir (Wien) 57:67-73, 1981
- Casmiro M, Cacciatore FM, D’Alessandro R: The pathogenesis of idiopathic normal pressure hydrocephalus: An open problem. Funct Neurol 4:403-410, 1989
- Dandy WE, Blackfan KD: An experimental and clinical study of internal hydrocephalus. JAMA 61:2216-2217, 1913
- Dandy WE, Blackfan KD: Internal hydrocephalus. An experimental, clinical and pathological study. Am J Dis Child 8:406-482, 1914
- Del Bigio MR, Cardoso ER, Halliday WC: Neuropathological changes in chronic adult hydrocephalus: Cortical biopsies and autopsy findings. Can J Neurol Sci 24:121-126, 1997
- Di Rocco C, Di Trapani G, Maira G, et al: Anatomo-clinical correlations in normotensive hydrocephalus. Reports on three cases. J Neurol Sci 33:437-452, 1977
- Gjerris F, Borgesen SE, Sorensen PS, et al: Resistance to cerebrospinal fluid outflow and intracranial pressure in patients with hydrocephalus after subarachnoid haemorrhage. Acta Neurochir (Wien) 88:79-86, 1987
- Gucer G, Viernstein L, Walker AE: Continuous intracranial pressure recording in adult hydrocephalus. Surg Neurol 13:323-328, 1980
- Hakim S, Adams RD: The special clinical problem of symptomatic hydrocephalus with normal cerebrospinal fluid pressure. Observations on cerebrospinal fluid hydrodynamics. J Neurol Sci 2:307-327, 1965
- Harbert JC, McCullough DC, Schellinger D: Computed cranial tomography and radionuclide cisternography in hydrocephalus. Semin Nucl Med 7:197-200, 1977
- Harris NG, McAllister JP 2nd, Conaughty JM, et al: The effect of inherited hydrocephalus and shunt treatment on cortical pyramidal cell dendrites in the infant H-Tx rat. Exp Neurol 141: 269-279, 1996
- Karahalios DG, Rekate HL, Khayata MH, et al: Elevated intracranial venous pressure as a universal mechanism in pseudotumor cerebri of varying etiologies. Neurology 46:198-202, 1996
- Kosteljanetz M, Nehen AM, Kaalund J: Cerebrospinal fluid outflow resistance measurements in the selection of patients for shunt surgery in the normal pressure hydrocephalus syndrome. A controlled trial. Acta Neurochir (Wien) 104:48-53, 1990
- Krauss JK, Regel JP, Vach W, et al: White matter lesions in patients with idiopathic normal pressure hydrocephalus and in an age-matched control group: A comparative study. Neurosurgery 40:491-496, 1997
- Kristensen B, Malm J, Fagerland M, et al: Regional cerebral blood flow, white matter abnormalities, and cerebrospinal fluid hydrodynamics in patients with idiopathic adult hydrocephalus syndrome. J Neurol Neurosurg Psychiatry 60:282-288, 1996
- Long DM: Aging in the nervous system. Neurosurgery 17:348-354, 1985
- Lovely TJ, McAllister JP 2nd, Miller DW, et al: Effects of hydrocephalus and surgical decompression on cortical norepinephrine levels in neonatal cats. Neurosurgery 24:43-52, 1989
- Matsumae M, Kikinis R, Morocz I, et al: Intracranial compartment volumes in patients with enlarged ventricles assessed by magnetic resonance-based image processing. J Neurosurg 84:972-981, 1996
- McCormick JM, Yamada K, Rekate HL, et al: Time course of intraventricular pressure change in a canine model of hydrocephalus: Its relationship to sagittal sinus elastance. Pediatr Neurosurg 18:127-133, 1992
- Meier U, Zeilinger FS, Schonherr B: Endoscopic ventriculostomy versus shunt operation in normal pressure hydrocephalus: Diagnostics and indication. Minim Invasive Neurosurg 43:87-90, 2000
- Miyajima M, Sato K, Arai H: Choline acetyltransferase, nerve growth factor and cytokine levels are changed in congenitally hydrocephalic HTX rats. Pediatr Neurosurg 24:1-4, 1996
- Miyazawa T, Sato K: Learning disability and impairment of synaptogenesis in HTX-rats with arrested shunt-dependent hydrocephalus. Childs Nerv Syst 7:121-128, 1991
- Miyazawa T, Sato K, Ikeda Y, et al: A rat model of spontaneously arrested hydrocephalus. A behavioural study. Childs Nerv Syst 13:189-193, 1997
- Oi S, Shimoda M, Shibata M, et al: Pathophysiology of long-standing overt ventriculomegaly in adults. J Neurosurg 92:933-940, 2000
- Poca MA, Sahuquillo J, Busto M, et al: Agreement between CSF flow dynamics in MRI and ICP monitoring in the diagnosis of normal pressure hydrocephalus. Sensitivity and specificity of CSF dynamics to predict outcome. Acta Neurochir Suppl 81:7-10, 2002
- Raftopoulos C, Chaskis C, Delecluse F, et al: Morphological quantitative analysis of intracranial pressure waves in normal pressure hydrocephalus. Neurol Res 14:389-396, 1992
- Raftopoulos C, Deleval J, Chaskis C, et al: Cognitive recovery in idiopathic normal pressure hydrocephalus: A prospective study. Neurosurgery 35:397-405, 1994
- Rekate HL, Brodkey JA, Chizeck HJ, et al: Ventricular volume regulation: A mathematical model and computer simulation. Pediatr Neurosci 14:77-84, 1988
- Sahuquillo J, Rubio E, Codina A, et al: Reappraisal of the intracranial pressure and cerebrospinal fluid dynamics in patients with the so-called “normal pressure hydrocephalus” syndrome. Acta Neurochir (Wien) 112:50-61, 1991
- Suda K, Sato K, Takeda N, et al: Early ventriculoperitoneal shunt—effects on learning ability and synaptogenesis of the brain in congenitally hydrocephalic HTX rats Childs Nerv Syst 10: 19-23, 1994
- Vanneste J, Augustijn P, Davies GA, et al: Normal-pressure hydrocephalus. Is cisternography still useful in selecting patients for a shunt? Arch Neurol 49:366-370, 1992
- Vanneste J, Hyman R: Non-tumoural aqueduct stenosis and normal pressure hydrocephalus in the elderly. J Neurol Neurosurg Psychiatry 49:529-535, 1986
- Williams MA, Razumovsky AY, Hanley DF: Comparison of Pcsf monitoring and controlled CSF drainage diagnose normal pressure hydrocephalus. Acta Neurochir Suppl (Wien) 71:328-330, 1998
