
Treatment of Normal Pressure Hydrocephalus
Harold L. Rekate, MD
Division of Neurological Surgery, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
The treatment of normal pressure hydrocephalus (NPH) is complicated by the lack of certain diagnostic criteria. The risks associated with treating NPH are also higher than for other forms of hydrocephalus. Possible forms of treatment include ventricular shunts, lumbar shunts, and endoscopic third ventriculostomy. The lack of well-constructed, randomized, controlled trials on the treatment of NPH means that all forms of treatment should be considered “options.” Recent technological advances, however, have improved the management of NPH. The risks of treatment are lessened by the use of high-pressure valves with devices that retard siphoning. Better outcomes are associated with lowpressure valves. When NPH is treated with ventriculoperitoneal shunts, choosing a programmable valve with a device that retards siphoning maximizes the efficacy of treatment while minimizing the risks. Lumboperitoneal shunts are effective in the management of NPH but have a higher overall failure rate than ventriculoperitoneal shunts. Furthermore, their continued functioning is difficult to assess. A few patients with NPH have responded well to endoscopic third ventriculostomy, and this form of treatment deserves careful assessment.
Key Words: endoscopic third ventriculostomy, lumboperitoneal shunt, normal pressure hydrocephalus, ventriculoperitoneal shunt
The quality of life of carefully selected patients with normal pressure hydrocephalus (NPH) can be improved significantly by shunting. Because they are programmable, valves with devices that retard siphoning, specifically the Codman Hakim Programmable Valve with Siphonguard, can recreate the normal dynamics of cerebrospinal fluid (CSF), improve the possibility of good outcomes, and minimize the risks of mechanical problems with the shunts. Lumboperitoneal (LP) shunts can manage NPH effectively, but the difficulty of assessing their function and their lack of long-term reliability make their use problematic. The use of endoscopic third ventriculostomy (ETV) has only recently been advocated. No large assessment studies have been performed, but ETV may hold promise for the management of this condition without a permanent internal shunt.
Other articles in this issue consider the pathophysiology of NPH and patient selection. The starting point of this review is a patient who has already been evaluated and found to be a potential candidate for treatment of this condition. What are the treatment options and how does one select among them to maximize the risk-benefit ratio associated with treatment?
Methods
Using computerized search engines, the literature in English was surveyed using the key words, normal pressure hydrocephalus, with the modifier, treatment. Clinical information derived from the author’s practice and theoretical considerations based on the concepts of the biophysics of the condition were also used. Initially, 208 articles met the inclusion criteria and were scanned by title. Of these, 30 articles were read entirely and found useful for this review. Alternative forms of treatment for NPH, including the use of LP shunts and ETV, were also explored.
Results of Literature Review
Unfortunately, no randomized controlled trials have compared patients who have been shunted to matched controls of patients who may have been candidates but were not shunted. Furthermore, no randomized control trials have compared the outcomes of different treatments. Therefore, no Class I data are available on which to base treatment decisions for NPH.
Three articles retrospectively compared the treatment outcomes of patients who had undergone two different types of shunting methods. In two of these studies, low-pressure valves were superior to medium- or highpressure valves.[3,11] In one of these studies, however, the complication rate associated with low-pressure valves was significantly higher than those associated with the other valves. Therefore, the advantage of the low-pressure valve was significantly reduced. Nonetheless, the authors suggested that their data supported the use of low-pressure valves in patients undergoing shunt treatment for NPH.[3]
One article compared the success of treatment and complications rates between differential pressure valves and flow-control valves (Orbis Sigma valve, NMT Corporation, Duluth, GA),[21] but there were no differences in outcomes. The authors concluded that flow-control valves and differential pressure valves are associated with overdrainage, and both can treat the condition adequately.[21] Of their patients, 90% improved. Those with the differential pressure valves did slightly better than the other patients, but the finding was not statistically significant.
Without randomized control trials, the data are insufficient to recommend any form of treatment of NPH as a standard. Retrospective studies of a relatively large number of patients treated with low-pressure valves compared to high- and medium-pressure valves show an increased likelihood of improvement with low-pressure valves.[11] The lack of statistical significance and the higher complication rate associated with low-pressure valves make it impossible to recommend this form of treatment even as a guideline. Therefore, all forms of treatment for NPH must be viewed as options.
Based on this discussion, the ideal valve for use with ventriculoperitoneal (VP) shunting is likely to differ among patients. Such a valve would allow the lowest drainage pressure and therefore the highest likelihood of decreasing ventricular size that could be tolerated by a given patient. There are methods purported to help select such a valve, but none have been validated.[2,23] Valve selection is discussed further after the complications associated with treating patients with NPH and the relationship of shunt biophysics to preventing these complications are explored.
Complications Associated with Treatment of NPH
By consensus, complication rates associated with the treatment of NPH are significantly higher than those associated with the treatment of other forms of hydrocephalus. Three types of complications need to be discussed. The first and most common complication involves the failure of the procedure to lead to improvement in patients or patients who continue to deteriorate after a brief period of improvement. There are two potential explanations for these failures. The first case assumes that if patients fail to improve, they did not actually have NPH and were selected for treatment inappropriately. Williams and colleagues have cast doubt on this explanation and suggest that many of these patients have nonfunctioning shunts and would benefit from a shunt revision.[22] The diagnosis of shunt malfunction is particularly difficult in patients with NPH because marked decreases in ventricular size are unusual and the signs and symptoms of increased intracranial pressure (ICP) are lacking. Before clinicians conclude that patients are not responding to a shunt, Williams et al.[22] believe that it is essential to assure that an adequate shunt is in place. Shunt function should be assessed either clinically or radiographically, or the shunt should be explored surgically and replaced with a shunt with a lower opening pressure.
Infectious complications of shunt systems are a continuous problem in the treatment of hydrocephalus. A recent multicenter trial in children found a baseline rate of shunt infection of 8%.[7] The infection rate in adults is likely somewhat lower, but this issue has not been subjected to multicenter scrutiny. A recent publication with a relatively large number of patients with NPH reported an infection rate of 8.5% in adults.[23] Shunt infection is a serious complication and can be associated with multiple surgical procedures and weeks in the hospital. It rarely, however, causes permanent disability. Furthermore, if patients respond to the treatment, a decision against such treatment would seldom be made.
Mechanical complications are probably common in patients who receive VP shunts for the treatment of NPH. The size of the ventricles seldom decreases. Therefore, obstruction of the ventricular catheter, which is the most common complication associated with ventricular shunting in children, is a very rare complication in adults. Most severe and especially life-threatening complications are related to overdrainage of the shunt. Overdrainage leads to the usually benign but troubling complication of subdural effusions or to the potentially life-threatening complication of brain collapse and the development of subdural hematomas. Subdural effusions are usually associated with headaches that are localized to the side of the effusion. In a Dutch study, they occurred in 71% of patients with low-pressure differential valves but only in 34% of patients with medium-pressure differential valves.[3] Later discussion of this study postulated that the use of devices that retard siphoning (DRS) would improve outcomes in these patients.[6]
A recent review of 44 articles documented a 59% rate of improvement and a 38% complication rate in patients receiving shunts for the treatment of NPH.[9] Of these complications, 6% were considered severe (i.e., death or permanent life-changing neurologic injury). Twenty-two percent of the patients required a second surgical procedure.[9] Some of these repeat surgeries were needed to treat surgical complications (misplacement of a catheter) or infection. Understandably, however, some were related to the valve pressure or configuration. At least half of these repeat procedures could be avoided with the use of properly designed programmable valves that contained an effective DRS. The results reported in a multicenter study likewise showed a poorer response rate, but the complication rates were almost identical: 7% of patients either died or suffered permanent decreased neurologic functioning.[20]
Valve Selection for VP Shunts
Recent advances in the design of valves for shunts have resulted in at least the likelihood of improvement after treatment for NPH. The first DRS, developed by Portnoy and reported in 1973, was the anti-siphon device.[15] For the first time, shunt valves prevented extremely negative ICP when patients assumed the erect position. However, technical difficulties with the device and unpredictable failure modes created significant resistance to its routine use.[10]
In the early 1990s several other devices, including the Siphon Control Device, Delta Valves (Medtronic, PS Medical, Santa Barbara, CA), and the Orbis Sigma Valve (NMT, Duluth, GA), were developed to prevent overdrainage. The use of these devices in the treatment of NPH has not yet been validated with randomized controlled trials. Certainly, however, they have made the treatment of NPH safer and decreased the likelihood that subdural effusions and hematomas will develop.
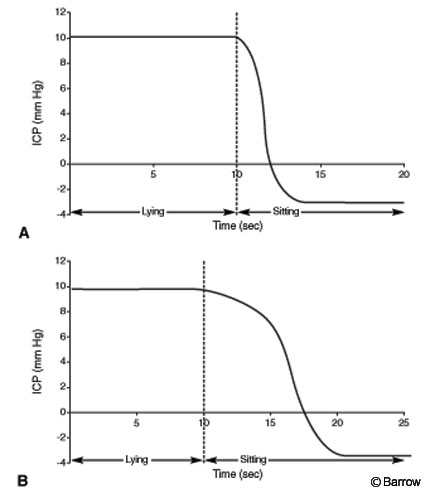
As a balance between valves with a low opening pressure and valves with an effective DRS to prevent significant negative ICP, I previously recommended using VP shunts with a Level I Delta valve for the treatment of NPH.[16] Normal ICP is about 10 mm Hg in the recumbent position and falls slowly to about –3 mm Hg when the erect position is assumed (Fig.1). The Level I Delta valve is associated with almost normal ICP in the recumbent position and also after an erect posture is assumed. It differs from normal only in that it equilibrates to the nadir of ICP much more quickly.
Some caveats, however, must be considered when using Delta valves. First, to function properly, the valve must be placed at the level of the foramen of Monro. For the diaphragm mechanism to work properly, the valve also must be placed under freely moving skin.[8]
The most recent technological advance to improve the risk-benefit ratio associated with the management of NPH is the development of programmable valve mechanisms. The first, the Sophy Valve (Sophysa Corporation), has never been marketed in the United States. The Medos Valve, now called the Codman Hakim Programmable Valve (Codman Corporation, Raynam, MA),was the first programmable valve to be approved by the Food and Drug Administration. It consists of a dial of differential pressure valves with opening pressures ranging from 30 mm H2O to 200 mm H2O in 10-mm steps. The valve pressures are extremely precise and stable over long periods.[14]
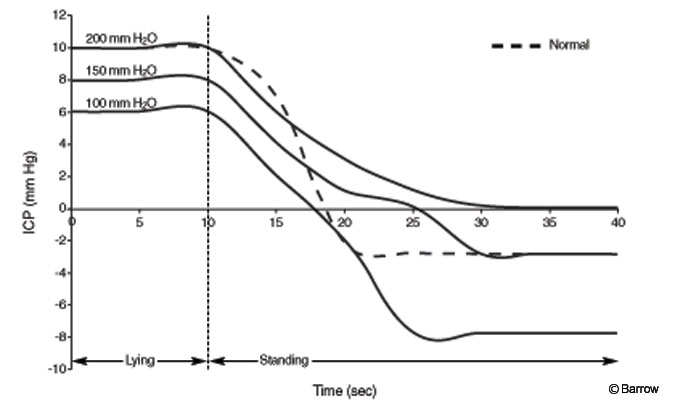
This valve was inappropriate for the treatment of most cases of NPH because the valve mechanism equilibrated rapidly and was associated with extremely negative ICP in the erect position. More recently, the device has been marketed with the addition of a new DRS called Siphonguard (Codman Corporation, Raynam, MA). For the first time, it is possible to recreate normal ICP dynamics in a patient with a VP shunt (Fig. 2). At present, this valve appears to be ideal for the treatment of NPH. I now use this valve system for all patients with NPH who receive a VP shunt.
Treatment with Programmable Valves
Without a compelling reason otherwise, all patients with NPH are treated with the Codman Hakim Programmable valve with Siphonguard originally set at 100 mm H2O. Information from the operating room is rarely available in the hospital or outpatient setting when a patient is next seen. Therefore, it is safe to assume that the valve is set at this level unless the setting has been adjusted in an environment in which the information is available. In patients with a positive outcome from the shunt and with no negative symptoms, there is no reason to change this setting, which is the most likely to recreate the normal dynamics of ICP. Patients who do not improve after surgery or who later deteriorate can be treated with a systematic decrease in the opening pressure of the valve. Patients often respond initially and later show signs of deterioration. Lowering the opening pressure of the valve often recaptures the positive effect of treatment. Williams et al. found this situation to be true of patients who underwent valve replacements with lower pressure valves that required surgical intervention.[22]
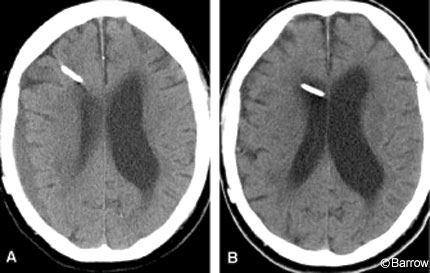
A small number of patients complain of postural headache when they assume the erect position. Many have unilateral headaches and will be found to have subdural effusions on imaging studies. For example, an 83-year-old man had a compelling history and imaging studies that led to the diagnosis of NPH (Fig. 3A). Immediately after surgery, his gait and bladder control improved markedly. A month later he complained of severe right-sided headaches, especially when assuming the erect position. A significant subdural effusion was found. The pressure of his valve was increased to 200 mm H2O, and the subdural effusion resolved within 3 weeks (Fig. 3B). Unfortunately, his gait disturbance returned and he required a walker. The valve pressure was changed to 170 mm H2O. He again was able to walk without the walker and continues to improve.
LP Shunting
LP shunting is a well-established form of treatment for NPH, but little has been published on the use of this technique.[19] High-volume withdrawal of CSF and the use of chronic controlled lumbar drainage in the diagnosis of this condition suggest that the technique would be an excellent option for treating NPH (Marmarou A, personal communication, 2002). Unless hydrocephalus is caused by aqueductal stenosis, LP shunting is an option in the treatment of any form of hydrocephalus. In infants LP shunting can lead to hindbrain herniation that could become symptomatic after a number of years.[4,5] The use of valves in an LP shunt can prevent this complication.[18] No studies have implicated LP shunts as causing hindbrain herniation in patients with NPH or other causes of hydrocephalus in adults.
Compared to VP shunting in patients with NPH,LP shunting has several advantages. That the cranium is not entered is an advantage because intraoperative bleeding is troublesome. However, subdural hematomas can still develop as a result of negative ICP. Published infection rates for LP shunts are significantly lower than those for VP shunts, possibly 10-fold lower.[7,19]
For two interrelated reasons, LP shunts are not ideal forms of treatment for NPH. The size of the ventricles does not decrease routinely after shunting, even when the outcome is excellent. Therefore, the function of the shunt cannot be assessed by a cranial imaging study. ICP does not increase markedly when the shunt has failed, nor are there signs of increased intracranial hypertension. It therefore is difficult to determine whether the shunt is working. If the patient does not respond, it may be impossible to discern whether the patient is a nonresponder or whether the shunt is not working properly.
The second problem is related to the overall lack of confidence in LP shunts for prolonged function. This lack of confidence stems from their use to manage pseudotumor cerebri. The failure rate of LP shunts is quite high, and their long-term function is the exception. Because the risk associated with this procedure is relatively low, LP shunting might be seen as an effective diagnostic test in patients who may be less likely to respond to shunting.
ETV
Most cases of NPH are classified as communicating hydrocephalus. Such patients are not candidates for ETV. The ideal candidate for ETV has aqueductal stenosis. Information based on modeling NPH reflects the likelihood that many, if not most, cases of NPH are caused by an obstruction to CSF flow between the spinal subarachnoid space and the cortical subarachnoid spaces.[17] Patients with this form of communicating hydrocephalus are indeed candidates for ETV.[1]
In two small series, NPH in nine patients was treated successfully with ETV.[12,13] Meier and colleagues selected patients by performing both ventricular and lumbar infusion tests. Patients who showed a high resistance to CSF outflow in the ventricle and low resistance in the lumbar theca were offered ETV. In the selected patients, the floor of the third ventricle was bowed into the interpeduncular cistern, suggesting CSF flow had failed between the third ventricle and cistern (Meier U, personal communication, 2002).
An alternative method for selecting patients for ETV that might allow a greater number of patients to be considered as candidates for the procedure is suggested by the results of isotope cisternography. This technique has been used to select patients with NPH for shunting. Patients were selected in whom the radioactive tracer linked to albumin was injected into the lumbar theca. In a positive response, the dye entered the ventricle quickly but stayed there for prolonged periods. The dye flowed into the ventricle, but its entrance into the cortical subarachnoid space was restricted as suggested by the mathematical model.[17] The blockage is at the skull base between the spinal and cortical subarachnoid spaces; therefore, ETV will be an effective internal bypass. I have performed the procedure in one patient who had an excellent outcome. However, the use of ETV must be evaluated in a larger series before the procedure can be recommended.
Conclusion
NPH is one of the few surgically treatable forms of dementia. In truth, it is one of the few dementing, degenerative conditions for which any hope exists at all. It is frustrating that the diagnosis cannot be made with certainty and that all tests are associated with both false-negative and false-positive results. What choices do these patients have? They can accept their progressive deterioration, or they can take the courageous course to undergo a relatively risky operation that might reverse or stabilize the progress of the disease. It is the responsibility of clinicians to give patients and families the information they need to decide whether the risks are worth taking. The availability of programmable shunts, advances in diagnostic acumen, and the potential of alternative forms of treatment are improving the quality of life for a growing number of older patients.
References
- Baskin JJ, Manwaring KH, Rekate HL: Ventricular shunt removal: The ultimate treatment of the slit ventricle syndrome. J Neurosurg 88:478-484, 1998
- Black PM, Hakim R, Bailey NO: The use of the Codman-Medos Programmable Hakim valve in the management of patients with hydrocephalus: Illustrative cases. Neurosurgery 34:1110-1113, 1994
- Boon AJ, Tans JT, Dewel EJ, et al: Dutch normal-pressure hydrocephalus study: Randomized comparison of low-and medium-pressure shunts. J Neurosurg 88:490-495, 1998
- Chumas PD, Armstrong DC, Drake JM, et al: Tonsillar herniation: The rule rather than the exception after lumboperitoneal shunting in the pediatric population. J Neurosurg 78:568-573, 1993
- Chumas PD, Kulkarni AV, Drake JM, et al: Lumboperitoneal shunting: A retrospective study in the pediatric population. Neurosurgery 32:376-383, 1993
- de Jong DA, Delwel EJ, Avezaat CJ: Hydrostatic and hydrodynamic considerations in shunted normal pressure hydrocephalus. Acta Neurochir (Wien) 142:241-247, 2000
- Drake JM, Kestle JT: Determining the best cerebrospinal fluid shunt valve design: The pediatric valve design trial. Neurosurgery 43:1259-1260, 1998
- Hassan M, Higashi S, Yamashita J: Risks in using siphon-reducing devices in adult patients with normal-pressure hydrocephalus: Bench test investigations with Delta valves. J Neurosurg 84:634-641, 1996
- Hebb AO, Cusimano MD: Idiopathic normal pressure hydrocephalus: A systematic review of diagnosis and outcome. Neurosurgery 49:1166-1186, 2001
- McCullough DC: Symptomatic progressive ventriculomegaly in hydrocephalics with patent shunts and antisiphon devices. Neurosurgery 19:617-621, 1986
- McQuarrie IG, Saint-Louis L, Scherer PB: Treatment of normal pressure hydrocephalus with low versus medium pressure cerebrospinal fluid shunts. Neurosurgery 15:484-488, 1984
- Meier U, Zeilinger FS, Schonherr B: Endoscopic ventriculostomy versus shunt operation in normal pressure hydrocephalus: Diagnostics and indication. Acta Neurochir Suppl 76:563-566, 2000
- Mitchell P, Mathew B: Third ventriculostomy in normal pressure hydrocephalus. Br J Neurosurg 13:382-385, 1999
- Oikonomou J, Aschoff A, Hashemi B, et al: New valves—new dangers? 22 valves (38 probes) designed in the ‘nineties in ultralongterm tests (365 days)’. Eur J Pediatr Surg 9 Suppl 1:23-26, 1999
- Portnoy HD, Tripp L, Croissant PD: Hydrodynamics of shunt valves. Childs Brain 2:242-256, 1976
- Rekate HL: Biophysics of cerebrospinal fluid and shunts. Techniques in Neurosurgery 7:186-196, 2002
- Rekate HL, Brodkey JA, Chizeck HJ, et al: Ventricular volume regulation: A mathematical model and computed simulation. Pediatr Neurosci 14:77-84, 1988
- Rekate HL, Wallace D: Lumboperitoneal shunts in children. Pediatr Neurosurg 38:41-46, 2003
- Selman WR, Spetzler RF, Wilson CB, et al: Percutaneous lumboperitoneal shunt: Review of 130 cases. Neurosurgery 6:255-257, 1980
- Vanneste J, Augustijn P, Dirven C, et al: Shunting normal-pressure hydrocephalus: Do the benefits outweigh the risks? A multicenter study and literature review. Neurology 42:54-59, 1992
- Weiner HL, Constantini S, Cohen H, et al: Current treatment of normal-pressure hydrocephalus: Comparison of flow-regulated and differential-pressure shunt valves. Neurosurgery 37:877-884, 1995
- Williams MA, Razumovsky AY, Hanley DF: Evaluation of shunt function in patients who are never better, or better then worse after shunt surgery. Acta Neurochir Suppl (Wien) 71:368-370, 1998
- Zemack G, Romner B: Seven years of clinical experience with the programmable Codman Hakim valve: A retrospective study of 583 patients. J Neurosurg 92:941-948, 2000
