
Sacral Amputation for Sarcoma: Case Report
Roger Hartl, MD
Robert J. Standerfer, MD*
Stephen W. Coons, MD**
Curtis A. Dickman, MD
Divisions of Neurological Surgery and **Neuropathology, Barrow Neurological Institute, and *Department of Surgery, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Abstract
Tumors of the sacrum are rare, and malignant sacral lesions such as sarcomas typically are associated with a poor prognosis. Radical surgical resection is the mainstay of treatment followed by radiation therapy and sometimes chemotherapy. Resection of these lesions can be challenging because the anatomy of this region is complex and nervous and vascular structures are frequently involved. We report a patient with a malignant peripheral nerve sheath tumor in the sacrum who underwent subtotal sacrectomy; stability of the sacroiliac joint was maintained through anterior and posterior approaches performed during the same operative setting. The patient’s motor function was preserved. A multidisciplinary approach involving the neurosurgeon, colorectal surgeon, and plastic surgeon in the operating room and the oncologist and radiation oncologist in the postoperative period is key to the successful and expedient treatment of these lesions.
Key Words: malignant peripheral nerve sheath tumor, sacrectomy, sacrum, sarcoma
Tumors of the sacrum are rare and encompass a variety of lesions, including congenital lesions, inflammatory masses, neurogenic and osseous tumors, metastatic lesions, and others such as aneurysmal bone cysts and sarcomas.[18-20,30] Primary malignant sacral bony lesions, which include Ewing’s sarcomas, osteosarcomas, and chondrosarcomas, are associated with a poor prognosis.[3,5,9,15,26] Aggressive surgical resection is the mainstay of treatment followed by radiation of the surgical bed and sometimes adjuvant chemotherapy. Resection of these lesions is difficult because the anatomy of this region is complex and neurovascular structures are often involved. Because tumors of the sacrum are rare, few institutions have large series of patients who have undergone en bloc resection of these lesions. [19,30] Hence, experience regarding the optimum surgical technique and indications for the resection of sacral tumors is limited.
We present a patient who underwent a sacral laminectomy for treatment of a lesion thought to be a benign nerve sheath tumor but that was diagnosed as a malignant sarcoma after partial resection. The patient then underwent a sacral amputation and en bloc resection of the remaining tumor. This case illustrates a surgical technique for subtotal sacrectomy that maintains stability of the sacroiliac joint. A multidisciplinary approach involving a neurosurgeon, vascular surgeon, colorectal surgeon, and plastic surgeon during surgery and an oncologist and radiation oncologist after surgery is key to aggressive treatment of these lesions.
Illustrative Case
A 49-year-old, previously healthy man was referred for evaluation of a tumor at S2 on the right. Six months earlier he had noticed aching pain in his tailbone radiating down the posterior aspect of his right leg into his foot. Numbness across his right buttock; down his posterior right leg to the foot; and on the right side of his scrotum, penis, and anus followed. He also experienced some sexual dysfunction and urinary urgency.
On physical examination the patient was well nourished, had stable vital signs, and appeared to be in good health. His motor strength was 5/5 in all extremities, and the straight leg sign was negative. There was no tenderness along his lower lumbar spine, hips, or sacroiliac joint. Pinprick sensation was diminished along the dorsum of the right foot and medial aspect of the calf. His right ankle jerk was absent; all other reflexes were 2+.
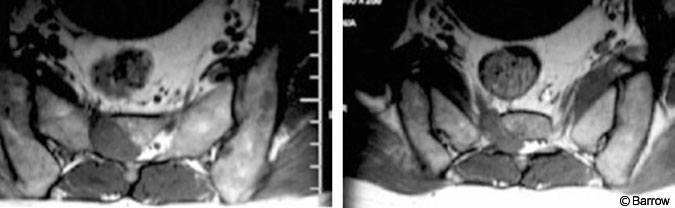
Magnetic resonance imaging of the lumbar spine and sacrum showed a 3-cm enhancing mass involving the right S2 nerve root and extending into the sacrum and S2 neural foramen (Fig. 1). Based on these images, a nerve sheath tumor was diagnosed. The patient underwent a sacral laminectomy for resection of the tumor. A firm, encapsulated, gray-tan, extradural mass appeared to be infiltrating the S2 nerve root, which was thickened. Subtotal resection was achieved, and the S2 nerve root was preserved.
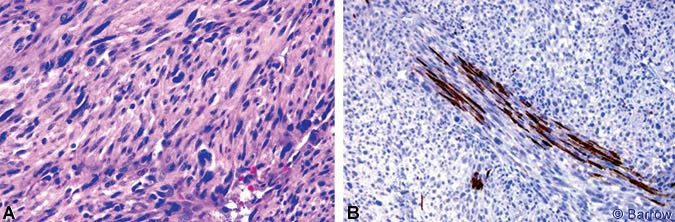
Microscopic examination showed a highly cellular neoplasm composed of spindle cells and large bizarre cells with atypia, pleomorphism, and a high mitotic rate. The neoplastic cells had infiltrated and spread extensively within the S2 nerve root (Fig. 2). The neoplastic cells were negative for S-100, desmin, keratin AE1/AE3, and epithelial membrane antigen. Based on these characteristics, a high-grade sarcoma was diagnosed. The growth pattern was most consistent with a malignant peripheral nerve sheath tumor (MPNST).
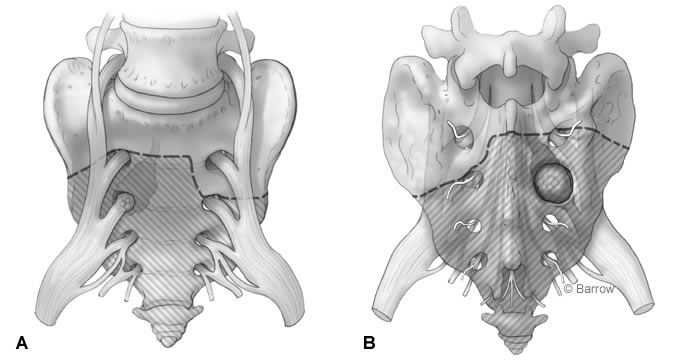
Given the final pathological findings, the decision was made to perform a radical resection of the involved structures with amputation of the sacrum below S1 on the right and below S2 on the left (Fig. 3). This strategy was chosen to preserve stability of the sacroiliac joint and to keep the S1 roots intact in the hope of avoiding postoperative weakness of the lower extremity. The risks and benefits of the procedure were explained to the patient in detail. He was informed of the high likelihood of losing bowel, bladder, and sexual function. The patient decided to proceed with surgery.
Preoperatively, a strategy was outlined in conjunction with a colorectal surgeon, a plastic surgeon, and a vascular surgeon who often participates in neurosurgical approaches to the anterior and posterior spine. The decision was made to use a combined anterior, transabdominal, and posterior approach to amputate the sacrum.
Surgical Technique
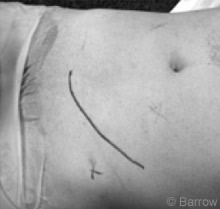
The patient was anesthetized and intubated, and intravenous and arterial lines were placed. Together, the colorectal surgeon and vascular surgeon performed the approach. The patient was placed supine. A curved incision was made in the left lower abdominal quadrant extending late rally from the midline (Fig.4). The anterior rectus and external oblique muscles were cut in line with the incision.
The retroperitoneum was entered, and the dissection proceeded toward the iliac vessels. The internal iliac artery was identified on both sides and followed caudally. Arteries and veins to the gluteal region were carefully dissected from surrounding tissue and divided. The right S2 nerve root, thickened and clearly infiltrated with tumor, was identified. The root was ligated and divided caudally, and the specimen was sent to pathology to confirm that clear margins had been obtained. Osteotomies were performed through almost the complete thickness of the sacrum, starting on the right side immediately below the S1 nerve root and moving toward the left side at a level immediately below the foramen of the S2 nerve root (Fig. 3). Metal clips were placed into the osteotomy defect so the level of the cut could be identified fluoroscopically during the posterior approach. The incision was then closed, and the patient was turned to the prone position.
[one_half]
[/one_half]
[one_half_last]
[/one_half_last]
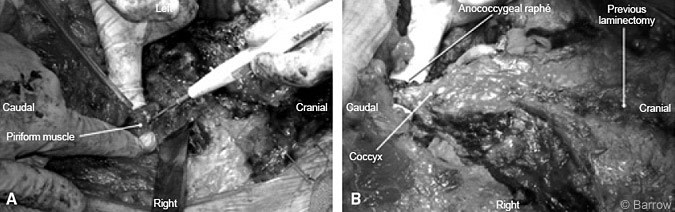
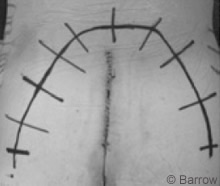
For the posterior approach a transverse arched incision was fashioned to expose the lumbosacral and coccygeal regions (Fig.5). A subcutaneous skin flap was developed and reflected caudally to expose the sacral fascia and gluteus muscle. The sacrum and sacroiliac joint were exposed by dissecting the gluteal and piriform muscles and by transecting the sacrospinous and sacrotuberous ligaments and the anococcygeal raphé (Fig. 6). Care was taken to avoid injuring the sacral plexus and sciatic nerve as they exited the sacroiliac notch.
[one_half]

[/one_half]
[one_half_last]

[/one_half_last]
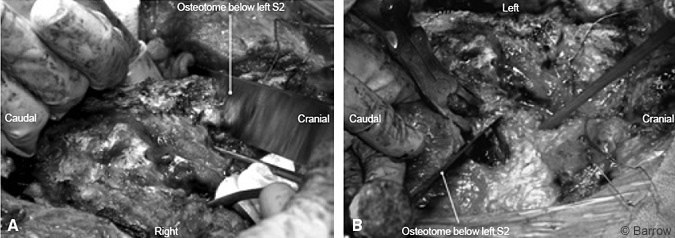
The laminectomy defect from the previous surgery enabled easy identification of the thecal sac and sacral nerve roots. Tumor was still present near the right S1 nerve root, and the decision was made to sacrifice that root as well. The thecal sac was ligated and transected distally to the left S2 nerve root. The specimen was sent for pathological analysis to confirm clean margins. Next, an osteotomy, which connected the earlier anterior cuts, was performed on the left, immediately caudal to the S2 foramen and on the right caudal to the S1 foramen (Fig.7). The sacrum could then be elevated and disconnection of the sacrum completed (Figs. 8 and 9). Anterior to the sacrum the colon became visible.

For closure the plastic surgeon placed Gore-Tex mesh as a barrier between the peritoneal cavity and the muscle layer. The gluteal muscles were reapproximated and two drains were placed.
Outcome
Postoperative radiographs showed the resection of the sacrum and preservation of more than 50% of the sacroiliac joint (Fig. 10). As expected, the patient had genital and buttock numbness. He had lost bladder function and required self-catheterization. Sexual function was lost, but bowel continence was preserved. There was no right S1 radiculopathy, and he had excellent strength in his lower extremities. His postoperative course was complicated by deep vein thrombosis, which was treated with oral anticoagulation therapy. The patient underwent radiation treatment of the operative bed.
Discussion
MPNSTs are rare lesions that usually arise from neurofibromas or form de novo from normal peripheral nerves.[22] They most frequently involve the sciatic nerve. Only a few reports have described lesions like the one presented here.[16,21] These lesions also have been named neurogenic sarcomas, neurofibrosarcomas, and malignant schwannomas. They occur in adults between 20 and 50 years old. More than 50% of these tumors occur in patients with neurofibromatosis (NF-1), which predisposes individuals to the development of MPNSTs. Ionizing radiation also may contribute to the development of MPNSTs.
Histologically, the neural origin of MPNSTs cannot be proven on conventionally stained material, on which they resemble other soft tissue tumors. The diagnosis of MPNST is therefore based on their origin from a peripheral nerve, on their transition from a benign peripheral nerve tumor, or on their development in patients with NF-1.22 Our patient exhibited no signs of NF-1. Given the confirmed association of the tumor with the S2 nerve root and immunohistochemical findings, however, the diagnosis of MPNST was likely. A malignant fibrous histiocytoma was also considered in the differential diagnosis.
Treatment
Surgical resection is the mainstay of treatment for MPNSTs, and wide en bloc resection is desirable. By definition “radical” resection of a tumor involving the spine is impossible because the intrathecal space communicates with cranial nervous structures.[2] Therefore, en bloc resection with wide margins, the next best procedure, is the treatment of choice for sarcomas involving the spine (Table 1). A positive surgical margin is the main factor determining local recurrence and a major factor determining survival.[13,27]
Postoperative radiation treatment of soft tissue tumors, including MPNSTs, significantly reduces the rate of local recurrences. [4,23,27,28] To date, no chemotherapeutical regimen has proven effective in the treatment of these tumors. The prognosis of patients with high-grade sarcomas and MPNSTs is poor. In longterm studies the 5- and 10-year survival rates have ranged between 34 and 52% and between 23 and 43%, respectively.[7,11]
The operative plan in our patient was guided by basic oncological principles. The goals were to achieve en bloc resection of the tumor through a sacral amputation with a wide margin of normal tissue and to avoid violating the tumor intraoperatively.[8,17] Surgical removal of primary or malignant tumors of the sacrum or both, with and without lumbopelvic fixation, has been successful, but such case series have been small.[6,12,14,18,20,29,30]
Because of the complexity of the anatomy, anterior transabdominal and posterior sacral approaches have been combined to resect these tumors.[6,12,18] These procedures can be performed simultaneously, consecutively, or in a staged fashion. Tumor size, location, histological diagnosis, and involvement of surrounding structures must be considered during preoperative planning. A team approach that includes a neurosurgeon and colorectal, vascular, and plastic surgeons is crucial for success. The combined abdominosacral approach is probably the most versatile of the combined approaches. A consecutive procedure that allows the patient’s position to be changed under the same anesthesia is preferable. A simultaneous anterior and posterior approach with the patient in a lateral position has been described.[24] In our experience, however, visualization of the anatomy is limited and the pathology is difficult to access.
Stener initially popularized the surgical technique for sacrectomy.[25] A high sacrectomy is performed either between the first or second sacral vertebrae or rostral to the foramen of S1. The level of amputation should be at least one sacral segment above the most rostrally involved segment. Biomechanical studies have shown that stability is preserved even after sacral resection through the S1 foramen.[10] Lumbopelvic fixation can be avoided by leaving the S1 vertebral body intact. We chose this strategy to avoid complications associated with hardware implantation while achieving the most radical excision of the lesion possible.
Complications
Total or subtotal sacrectomies are associated with a significant rate of morbidity. These procedures should only be considered if a cure is the goal. Surprisingly, one S1 nerve root can often be sacrificed without incurring a significant neurological deficit. Sparing the S2 nerve root unilaterally or bilaterally often preserves some bowel or bladder function and occasionally some sexual function.[1] Sparing one or two S3 nerve roots guarantees almost normal bowel and bladder function. To retain normal sexual function, however, presacral dissection and destruction of the sympathetic and parasympathetic structures should be minimized.
Other significant risks associated with the procedure include injury to the major vessels, ureters, and other contents of the pelvis and abdomen; deep and superficial wound infections; wound dehiscence; and deep vein thrombosis. Wound complications, probably the most common postoperative problems, often require surgical intervention.[29] However, case series demonstrate that total or subtotal sacrectomy for the treatment of primary or malignant sacral tumors or both can be associated with acceptable rates of morbidity and mortality when performed in experienced centers.[6,12,14,18,20,24,29,30]
Conclusions
Sacrectomy is a valuable procedure for achieving local tumor control and improving survival from malignant sacral tumors. It should only be considered if a serious attempt is made to cure the underlying disease. The procedure should be planned carefully, and the patient should be thoroughly informed of the purpose of surgery and have realistic expectations of the outcome. Preserving as many sacral nerve roots as possible can dramatically reduce the morbidity rate associated with the procedure. Sacrectomy requires a multidisciplinary approach and should only be performed in centers with experience and the infrastructure to support management of this disease.
References
- Anson KM, Byrne PO, Robertson ID, et al: Radical excision of sacrococcygeal tumours. Br J Surg 81:460-461, 1994
- Boriani S, Weinstein JN, Biagini R: Primary bone tumors of the spine. Terminology and surgical staging. Spine 22:1036-1044, 1997
- Bradway JK, Pritchard DJ: Ewing’s tumor of the spine, in Sundaresan N, Schmidek HH, Schiller AL, et al (eds): Tumors of the Spine. Diagnosis and Clinical Management. Philadelphia: WB Saunders, 1990, pp 235-239
- Brennan MF, Hilaris B, Shiu MH, et al: Local recurrence in adult soft-tissue sarcoma. A randomized trial of brachytherapy. Arch Surg 122:1289-1293, 1987
- Cammisa FP, Jr., Glasser DB, Lane JM: Chondrosarcoma of the spine: Memorial Sloan-Kettering Cancer Center experience, in Sundaresan N, Schmidek HH, Schiller AL, et al (eds): Tumors of the Spine. Diagnosis and Clinical Management. Philadelphia: WB Saunders, 1990, pp 149-154
- Doita M, Harada T, Iguchi T, et al: Total sacrectomy and reconstruction for sacral tumors. Spine 28:E296-E301, 2003
- Ducatman BS, Scheithauer BW, Piepgras DG, et al: Malignant peripheral nerve sheath tumors. A clinicopathologic study of 120 cases. Cancer 57:2006-2021, 1986
- Enneking WF, Spanier SS, Goodman MA: A system for the surgical staging of musculoskeletal sarcoma. Clin Orthop 153:106-120, 1980
- Goodacre BW, Connell DG, Beauchamp CP, et al: High-grade chondrosarcoma of the sacrum—the crucial role of MR in surgical planning. Australas Radiol 36:168-171, 1992
- Gunterberg B, Romanus B, Stener B: Pelvic strength after major amputation of the sacrum. An experimental study. Acta Orthop Scand 47:635-642, 1976
- Hruban RH, Shiu MH, Senie RT, et al: Malignant peripheral nerve sheath tumors of the buttock and lower extremity. A study of 43 cases. Cancer 66:1253-1265, 1990
- Huth JF, Dawson EG, Eilber FR: Abdominosacral resection for malignant tumors of the sacrum. Am J Surg 148:157-161, 1984
- Kourea HP, Bilsky MH, Leung DH, et al: Subdiaphragmatic and intrathoracic paraspinal malignant peripheral nerve sheath tumors: A clinicopathologic study of 25 patients and 26 tumors. Cancer 82:2191-2203, 1998
- Kuklo TR, Bridwell KH, Lewis SJ, et al: Minimum 2-year analysis of sacropelvic fixation and L5-S1 fusion using S1 and iliac screws. Spine 26:1976-1983, 2001
- Malawski SK: The results of surgical treatment of primary spinal tumors. Clin Orthop 272:50-57, 1991
- Nagano Y, Nanko M, Nagahori Y: Presacral malignant peripheral nerve sheath tumor in a patient with von Recklinghausen disease: Case report. [Japanese]. Nippon Geka Gakkai Zasshi 101:429-432, 2000
- Neff JR: Anatomy and musculoskeletal oncology. Anatomic considerations for biopsy in orthopaedic oncologic management, in Reckling FW, Reckling JB, Mohn MP (eds): Orthopaedic Anatomy and Surgical Approaches. St. Louis: Mosby-Year Book, 1990, pp 493-518
- Ozdemir MH, Gurkan I, Yildiz Y, et al: Surgical treatment of malignant tumours of the sacrum. Eur J Surg Oncol 25:44-49, 1999
- Post KD, McCormick PC: Surgical considerations in pelvic tumors with intraspinal extension, in Sundaresan N, Schmidek HH, Schiller AL, et al (eds): Tumors of the Spine. Diagnosis and Clinical Management. Philadelphia: WB Saunders, 1990, pp 391-410
- Sar C, Eralp L: Surgical treatment of primary tumors of the sacrum. Arch Orthop Trauma Surg 122:148-155, 2002
- Scharl A, Feaux dL, Honig T, et al: Presacral malignant schwannoma. [German]. Geburtshilfe Frauenheilkd 47:747-749, 1987
- Scheithauer BW, Woodruff JM, Erlandson RA: Primary malignant tumors of peripheral nerve, in Scheithauer BW, Woodruff JM, Erlandson RA (eds): Atlas of Tumor Pathology. Tumors of the Peripheral Nervous System. Washington D.C.: Armed Forces Institute of Pathology, 1999, pp 303-378
- Shiu MH, Turnbull AD, Nori D, et al: Control of locally advanced extremity soft tissue sarcomas by function-saving resection and brachytherapy. Cancer 53:1385-1392, 1984
- Simpson AH, Porter A, Davis A, et al: Cephalad sacral resection with a combined extended ilioinguinal and posterior approach. J Bone Joint Surg Am 77:405-411, 1995
- Stener B: Technique of high sacral amputation, in Sundaresan N, Schmidek HH, Schiller AL, et al (eds): Tumors of the Spine. Diagnosis and Clinical Management. Philadelphia: WB Saunders, 1990, pp 411-416
- Sundaresan N: Osteosarcoma of the spine, in Sundaresan N, Schmidek HH, Schiller AL, et al (eds): Tumors of the Spine. Diagnosis and Clinical Management. Philadelphia: WB Saunders, 1990, pp 128-145
- Wong WW, Hirose T, Scheithauer BW, et al: Malignant peripheral nerve sheath tumor: Analysis of treatment outcome. Int J Radiat Oncol Biol Phys 42:351-360, 1998
- Woodruff JM: The pathology and treatment of peripheral nerve tumors and tumor-like conditions. CA Cancer J Clin 43:290-308, 1993
- Wuisman P, Lieshout O, Sugihara S, et al: Total sacrectomy and reconstruction: Oncologic and functional outcome. Clin Orthop 381:192-203, 2000
- Xu WP, Song XW, Yue SY, et al: Primary sacral tumors and their surgical treatment. A report of 87 cases. Chin Med J (Engl ) 103:879-884, 1990

