Surgical Approaches for Resection of Acoustic Neuromas
L. Fernando Gonzalez, MD
Gregory P. Lekovic, MD, PhD, JD
Randall W. Porter, MD
Mark J. Syms, MD**
C. Phillip Daspit, MD**
Robert F. Spetzler, MD
Division of Neurological Surgery and **Section of Neurotology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona
Download a .pdf of this article.
Abstract
The internal auditory canal can be accessed through different surgical approaches based on different trajectories. This article describes the progressive widening of the posterior trajectory that begins with progressive drilling of the petrous bone (the retrosigmoid approach). A more lateral trajectory that attacks the tumor from a lateral (the translabyrinthine approach) and superior-inferior approach (the middle fossa approach) is then described. Imaging and relevant clinical information such as hearing status, facial nerve compromise, and tumor size are key considerations when selecting an approach to acoustic neuromas.
Key Words: acoustic neuromas, middle fossa approach, partial labyrinthectomy, retrosigmoid, transcochlear, translabyrinthine, transpetrosal
Acoustic neuromas are the most common tumors of the cerebellopontine angle. Most are benign lesions that grow from the transition point between the central and peripheral myelin. They cause a wide variety of symptoms such as hearing loss, tinnitus, and balance abnormalities. Large lesions compress the contents of the posterior fossa and may cause cranial nerve dysfunction, long tract compromise, and even hydrocephalus if they grow large enough to obstruct cerebrospinal fluid (CSF) pathways. The internal auditory canal (IAC) can be accessed from different angles, and decisions about the right approach are based on clinical and radiological information.
A “team approach” is standard for patients with cerebellopontine angle tumors, especially acoustic neuromas. The team is composed of otolaryngological surgeons specializing in temporal bone anatomy and neurotology and of neurosurgeons dedicated to microneurosurgery. A team approach offers patients the expertise of both disciplines. The neurotologist performs the surgical exposure, which frequently involves progressive drilling of the petrous bone, and the neurosurgeon removes the tumor. The microanatomy of these approaches, which is beyond the scope of this review, is illustrated elsewhere.[21,22] This article discusses the advantages and indications of each approach, relevant surgical anatomy, and surgical nuances.
Surgical Overview
The posterior petrosectomies involve progressive, stepwise drilling of the petrous bone: The presigmoid, translabyrinthine, and transcochlear approaches are discussed (Table 1). Transpetrosal approaches progressively flatten the temporal bone to maximize surgical exposure and to minimize retraction on the cerebellum. The surgical corridor between the cerebellum and the petrous bone is also progressively widened during transpetrosal approaches. Anterior transpetrosal approaches aim for the petrous apex region while posterior approaches target the region posterior and lateral to the IAC. The middle fossa approach and Kawase’s approach are examples of anterior petrosectomies.
Retrosigmoid Approach
The retrosigmoid approach, with which most neurosurgeons are familiar, is versatile for the treatment of different pathologies. It is the most common approach for exposing the IAC with a trajectory parallel to the petrous surface. It allows resection of tumors of different sizes with the possibility of preserving facial and cochlear function. Preservation of hearing depends on the size of the lesion. If the tumor is less than 2 cm, the chance of preserving hearing is as high as 53%. This figure increases to 83% if the lesion is 1 cm or less.[20] The narrow corridor created provides adequate exposure of the contents of the posterior fossa and IAC. When a tumor is limited to the IAC or when it extends laterally beyond the “accessible” region after the posterior lip of the IAC has been drilled, other approaches should be attempted.
Surgical Technique
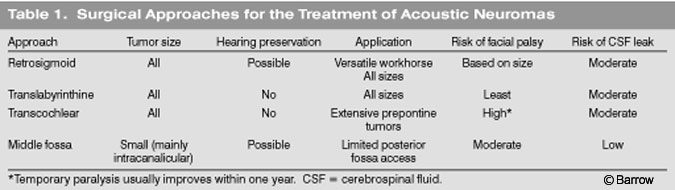
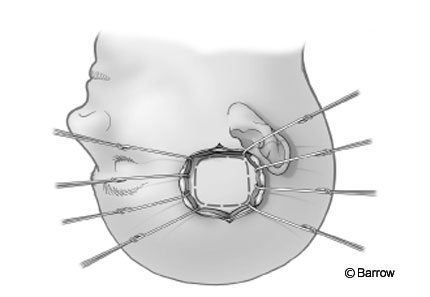
The patient is placed (Fig. 1) supine or in the park bench or sitting position. The sitting position has almost been abandoned because it is associated with a significant risk of air embolism. The sitting position, however, does offer a clean field because blood and CSF drain from the surgical field. The head is slightly flexed and rotated toward the ipsilateral side. This position can be uncomfortable for the surgeon, leading to early interruption of the procedure.
In the supine position, the patient’s head is rotated toward the contralateral side and is supported on a Mayfield three-point fixation device. A sand bag or a foam roll can be placed under the ipsilateral shoulder to minimize rotation of the neck. This same position is used for all posterior transpetrosal approaches. If the patient’s body habitus precludes contralateral head rotation due to neck compression or an elevated ipsilateral shoulder, a modified park bench position can be used. After anesthesia is induced, we routinely place a lumbar drain to help minimize retraction on the cerebellum and to avert a CSF leak after surgery. Frameless stereotactic image guidance and monitoring of the facial nerve and somatosensory evoked potentials are standard adjuncts.
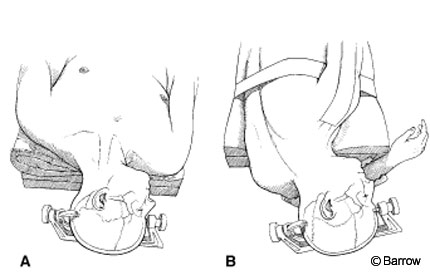
Venous bleeding, which is common during drilling, should be covered with hemostatic agents such as Nu-Knit (Johnson & Johnson, Arlington, TX) or Gelfoam (Pharmacia & Upjohn Company Kalamazoo, MI). To avoid sinus thrombosis, no hemostatic agent should be introduced within the lumen. Exposure of mastoid cells is unavoidable; they should be waxed to prevent CSF leakage.
If image guidance is unavailable, the sinuses are located using traditional landmarks. The transverse sinus is located beneath the imaginary line between the external occipital protuberance and the origin of the zygomatic arch. The sigmoid sinus is identified by drawing a line two fingerbreadths behind the ear aimed toward the mastoid tip. Alternatively, once the mastoid has been exposed, an imaginary line can be drawn from the parietomastoid suture to the mastoid tip.
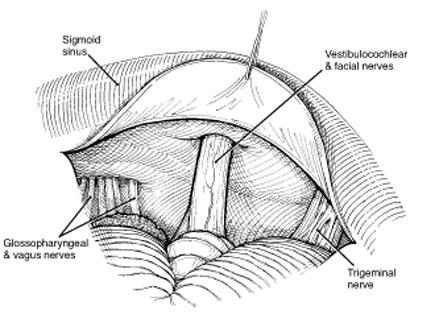
Early in the dissection when it is easier to distinguish tumor from surrounding tissues, arachnoid adhesions are released and the cerebellum and lower cranial nerves are separated from the tumor. The internal auditory canal should be drilled before the arachnoid over the facial and vestibulocochlear nerve complex is opened. This maneuver protects the nerves from bone dust generated during drilling. The cerebellum can be protected with an aluminum suture pack. The canal should be drilled approximately 8 mm. More aggressive drilling will violate the common crus and can result in hearing loss.
Once the cranial nerves have been identified, the tumor is morcellized from its core. Ultrasonic aspiration is especially helpful when resecting the core of the tumor. Small fragments are removed. As the anterior aspect is approached, the facial nerve is monitored. If there is evidence of facial nerve irritation, the area of dissection is changed. Once the facial nerve is identified, it is dissected from the tumor with a round knife. The facial nerve is usually displaced anteriorly against the anterior rim of the IAC, and this spot is most susceptible to injury during dissection. However, the facial nerve also can be superior or posterior to the tumor.
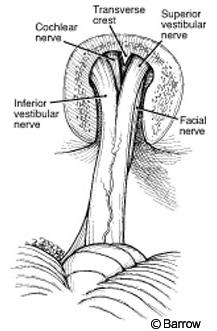
Once the extracanalicular portion of the tumor is removed, the posterior rim of the IAC should be opened (Fig. 4). The dura is cut in an arciform fashion with the base of the arc facing the cranial nerves. The posterior lip is drilled with a diamond bit drill with copious irrigation to prevent thermal injury. At this stage continuous monitoring of the facial nerve is essential. As the anterior portion of the tumor is approached, the dissection is performed with a round knife. The use of bipolar coagulation is avoided. The facial nerve usually takes a superior course from the brain stem and is located anterior to the vestibulocochlear nerve and anterior and inferior to the flocculus.
All exposed air cells are covered with bone wax to prevent CSF leakage. Once the tumor is removed and hemostasis is obtained, the dura is closed in a watertight fashion. The suture line is reinforced with fibrin glue. We routinely use a lumbar drain to help prevent CSF leakage. During closure the mastoidectomy should be filled with fat strips harvested from the abdomen. The bony defect is filled with acrylic or polymethyl methacrylate (PMMA) to reduce the risk of postoperative headache.[9]
Transpetrosal Approaches (Posterior Petrosectomy)
Drilling the temporal bone in a stepwise fashion is known as a posterior petrosectomy. The anatomy of the temporal bone is constant. Therefore the contents of the posterior fossa are exposed predictably as drilling proceeds. The risk of complications (e.g., facial paralysis, deafness, CSF leakage) increases as the amount of petrous bone removed increases.
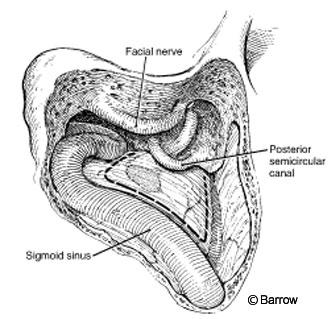
Mastoidectomy is the basis for the transpetrosal approaches, which involve progressively more removal of bone as described in the following sections. This sequential drilling implies that one step must be completed before proceeding with the next. Closure follows the same principles: watertight dural closure, the use of fibrin glue and fat graft implants harvested from the abdomen, coverage of bone defects with synthetic materials, and multilayer soft tissue reapproximation. The routine use of lumbar drains is recommended.
Presigmoid Approach
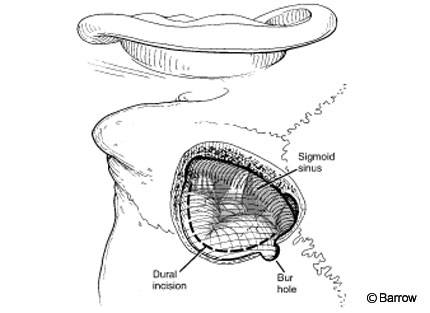
The surgical position and incision are the same as described for the retrosigmoid approach. Trautmann’s triangle (Fig. 5) is a triangular-shaped area of dura with an inferior vertex (jugular bulb), limited by the sigmoid sinus (posteriorly) and the otic capsule (anteriorly). The middle fossa dura and superior petrosal sinus form its superior base. This area is small and provides a narrow corridor that makes it difficult to manipulate instruments. The exposure can be enlarged by combining it with a traditional retrosigmoid exposure (see above). The sigmoid sinus is located in the middle of the exposure and provides limited access on each side of the exposure. The sigmoid sinus can be resected in selected cases.
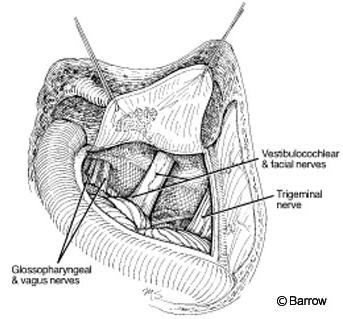
Translabyrinthine Approach
The translabyrinthine approach is ideal for tumors confined to the IAC (although there is no size limitation for a tumor removed via the translabyrinthine approach) and when hearing is severely compromised or is not an issue because the tumor is large (Gardner-Robertson grade III or IV).[4] This approach tends to be used for tumors larger than 2 cm when hearing preservation is unlikely. To gain access to the lateral recess of the IAC, hearing must be sacrificed.
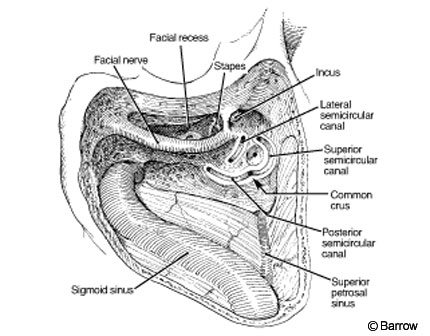
A C-shaped skin incision starts 1 cm superior to the auricle at the anterior-most aspect of the ear behind the sigmoid and extends to the mastoid tip. Drilling follows that used for the retrolabyrinthine presigmoid approach. Once the mastoidectomy is completed (Fig. 7) to the point that the antrum is exposed, the incus is identified. Its tip points in the direction of the facial nerve. The drilling is performed anterior to the facial nerve (facial recess), and the ossicles are fully exposed. The entire vertical portion of the facial nerve is drilled. The anterior bone of the facial recess is drilled to expose the incus and malleus. The incus is removed and the tendon for the tensor tympani is cut. The eustachian tube is identified, and it and the middle ear are packed with temporalis muscle to minimize the risk of CSF leakage.
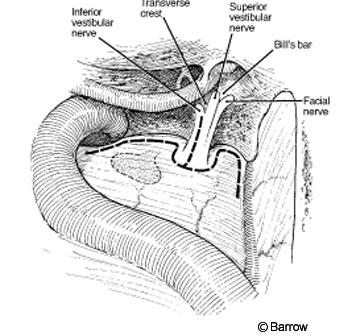
The vestibule (the junction between the lateral and posterior semicircular canals) that constitutes the lateral wall of the IAC is opened. The IAC is exposed superiorly, posteriorly, and inferiorly. The translabyrinthine approach exposes 270 degrees of the IAC. Inside the IAC (Fig. 8), the vertical crest (“Bill’s bar”), which shows the position of the facial nerve at the fundus of the canal, must be identified. The facial nerve is anterior and the superior vestibular nerve is posterior to the vertical crest. A great advantage of the translabyrinthine approach is that the labyrinthine segment of the facial nerve can be identified. Exposing the entry of the facial nerve into the IAC enables the transition between normal facial nerve involved with tumor to be identified. To preserve the integrity of the facial and superior vestibular nerves, their early identification is important. Skeletonizing the facial nerve jeopardizes its function and makes it more susceptible to iatrogenic injury.
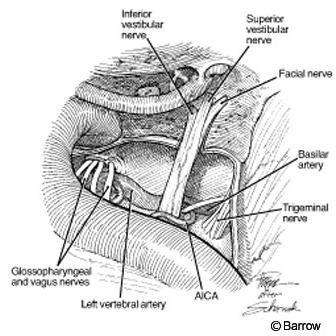
Once the dura is open, the entire subarachnoid, intracanicular, and vertical portions of the facial nerve are evident (Fig. 9).
[one_half padding=”0 0 20px 0″]
[/one_half]
[one_half_last]
[/one_half_last]
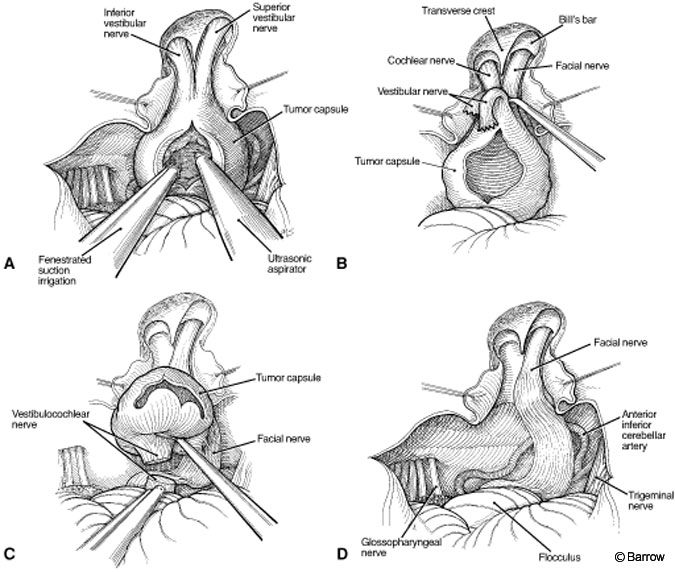
The facial nerve and the superior and inferior limits of the tumor are identified, and the tumor is debulked from it core (Fig. 10A). Once a significant portion of the tumor has been removed, the superior and inferior vestibular nerves are cut and the tumor is peeled away from the facial nerve (Fig. 10B). The tumor is separated from the cerebellum using the intervening arachnoid plane (Fig. 10C). Finally, the facial nerve is decompressed completely (Fig. 10D). During this stage, stimulation of the facial nerve is critical to preserve its function.
Once the tumor is removed, the closure proceeds as described. The bony defect is filled with fat strips harvested from the abdomen, and the bone defect is covered with PMMA or acrylic.
Partial labyrinthectomy (superior and posterior) may preserve hearing.[23] In a promising study, hearing was preserved in 100% of patients who underwent a “transcrusal” approach, in which only the superior and posterior semicircular canals, starting at the common crus, were removed. During resection of petroclival tumors, this partial labyrinthectomy adds millimeters to the exposure without sacrificing hearing. This approach plays a small role in the resection of acoustic neuromas.
Transcochlear Approach
This approach constitutes the most direct approach to the midclival region, prepontine cistern, and brainstem and follows the traditional principles of skull base surgery: maximizing bony resection and minimizing retraction on the adjacent brain. The approach provides the maximum amount of exposure from drilling the petrous bone by extending the amount of bone removed in the presigmoid and translabyrinthine approaches. It offers a wide corridor to the midaspect of the clivus and anterior brainstem. This approach is not very useful for the resection of acoustic neuromas and is associated with intrinsic associated morbidity, such as facial paralysis and potential CSF leakage.
There are two variations of the transcochlear approach. The original, as described by House and Hitselberger,[12] implies facial nerve transposition. It involves complete skeletonization of the facial nerve to mobilize the nerve posteriorly and to obtain a flat exposure. The greater superficial petrosal nerve (GSPN) is cut where it emerges from the geniculate ganglion so that the facial nerve can be released and mobilized. Once the facial nerve is mobilized posteriorly, the cochlea is entered. In the transotic approach described by Fisch,[7] the facial nerve is not mobilized, but the external auditory canal is occluded and resected. Drilling continues through the petrous apex up the inferior petrosal sinus. The petrous portion of the internal carotid artery (ICA) constitutes the lateral aspect of the exposure.[6]
Middle Fossa Approach
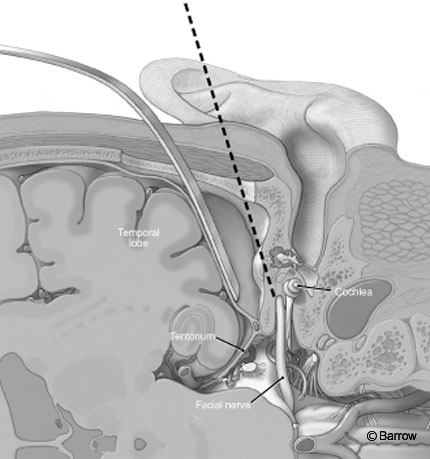
The middle fossa approach is versatile and associated with many potential uses. Anterior extension of this approach, known as the transKawase or extended middle fossa route, can be helpful in the removal of petroclival meningiomas or lesions in the petrous apex (e.g., cholesterol granulomas).
In 1904 Parry[19] first described the middle fossa approach for section of the vestibular nerve. In 1961 House,[10] a neurotologist working with Dr. Theodore Kurze, a neurosurgeon, described the approach for decompression of the IAC for the treatment of otosclerosis. The technique was abandoned, but its utility for removing acoustic neuromas was evident.[13] Dr. House also described the middle fossa approach for the removal of small acoustic neuromas.
Compared to other approaches to the IAC, the middle fossa approach adequately exposes the IAC and its contents, including the facial and superior vestibular nerves, from a superior trajectory (Fig. 11). It permits exposure of the subarachnoid, intracanalicular, labyrinthine, and horizontal or tympanic segment of the facial nerve. It is useful for small lesions (up to 2 cm in diameter) mainly within the IAC, especially when preservation of hearing is desired.
Surgical Technique
The long axis of the head is positioned parallel to the floor, with the ear facing the ceiling and the neck slightly extended. A vertical incision is made anterior to the tragus and perpendicular to the zygomatic arch (Fig. 12). The blood supply of the temporalis muscle is preserved. Fascia and temporal muscle are exposed and opened with the Bovie electrocauterization device and then retracted with a self-retractor or fishhooks. The bone below the squamous suture, which is a good landmark for identifying the superior limit of the craniotomy, is exposed.
The craniotomy is square. A third of it is positioned posterior to and twothirds of it is positioned anterior to the external auditory canal. This configuration adequately exposes the floor of the middle fossa, which must be flush to visualize and minimize retraction on the temporal lobe.

The dura is elevated in a posteriorto-anterior direction to prevent injury to the GSPN (Fig. 13). The geniculate ganglion is often dehiscent, and a posterior-to-anterior dissection minimizes facial nerve injury during elevation of the dura. The GSPN and lesser petrosal nerve are just beneath the dura and become visible once it is exposed.[2,24] Both nerves are delicate, and the operating microscope should be used to identify them. When the dura is retracted medially, the middle meningeal artery, with its anterior and posterior branches, can be seen emerging from the foramen spinosum. The middle meningeal artery is sectioned immediately after it exits the foramen spinosum, which is then exposed along with the mandibular nerve.
In the posterior part of the exposure near the petrous ridge, the arcuate eminence becomes visible. The angle between the arcuate eminence and the GSPN is about 120 degrees. After the GSPN, arcuate eminence, and petrous ridge are identified, the IAC can be localized by any of the following three techniques.

Garcia-Ibanez Technique
This technique (Fig. 14A) relies on the important relationship between the GSPN and the arcuate eminence, which are separated by 120 degrees. Bisecting this angle provides the site at which to start drilling the temporal bone to expose the IAC.[8]
Fisch Technique
A line is drawn over the long axis of the arcuate eminence (Fig. 14B). Another line is drawn 60 degrees to the first line away from the arcuate eminence. The second line provides the location of the IAC, which typically is 3 to 4 mm below the petrous ridge.[5] However, it may be as much as 7 mm below the floor of the middle fossa.[15]
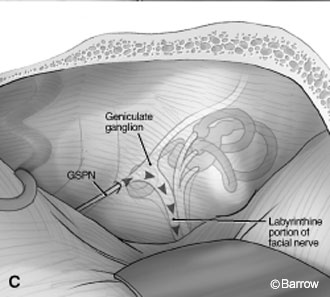
House Technique
House proposed that once the GSPN was identified, the floor of the middle fossa could be drilled (2 to 3 mm) to identify the geniculate ganglion and to follow the labyrinthine portion of the facial nerve medially until the IAC was reached (Fig. 14C).[8,10] The junction between the geniculate ganglion and the facial nerve is not on the same plane as the IAC; rather, it is slightly posterior. Therefore, most of the geniculate ganglion must be uncovered to expose the facial nerve medially. The exposure extends to the posterior limit, which is the superior semicircular canal. The anterior limit along the petrous ridge is the facial nerve. More laterally, the cochlea is the limiting factor for exposure.
After the roof of the IAC has been drilled (3 to 4 mm below the floor of the middle fossa), the vertical bar (Bill’s bar) of bone is easily identified. This bar separates the facial nerve from the superior vestibular nerve. Inside the IAC is the meatal portion of the anterior inferior cerebellar artery (AICA), which forms an important loop, enters the IAC, and then passes through the nerves. The anatomy of the AICA in this region is quite variable as is the percentage of its loop located inside the IAC.[18]
Early identification and preservation of these arterial branches inside the IAC are fundamental to the preservation of hearing during the resection of acoustic neuromas.
Extended Middle Fossa Approach to the IAC and Clivus
House[11] first described an area of the temporal bone that can be removed with impunity. Kanzaki et al.[14] later modified the approach. For the treatment of lowneck basilar tip aneurysms (between the sellar floor and IAC on a lateral projection), they described an approach that later proved useful for resecting meningiomas of the petroclival region.[3,16,17] In 1986 Day et al.[5] developed a geometric construct by compartmentalizing structures in and adjacent to the cavernous sinus. They named the area described by Kawase as the posteromedial triangle and its limits were as follows: the porus trigeminus, cochlea, and posterior border of the mandibular branch (Fig. 13).[8] The triangle is devoid of any vascular or nerve branches. Posteriorly, it is limited by the margin of the mandibular nerve, the petrous ridge, the GSPN laterally, and the nerves inside the IAC. The principle of this approach is the same as for the middle fossa approach. Hence, early identification of the IAC is important.
Surgical Technique
The head is positioned as described for the traditional middle fossa approach. The incision is in the shape of a question mark big enough to include the squamous portion of the temporal bone and part of the sphenotemporal bone. Below the skin incision, the fascia is incised in a semicircular fashion. The craniotomy must include the sphenotemporal junction. Its position varies, depending on if it needs to be extended anteriorly enough to include the posterior part of cavernous sinus.
The petrous apex is exposed extradurally until the petrous ridge is identified. The dura is again elevated in a posterior-to-anterior direction, and the foramen spinosum is identified. The middle meningeal artery is coagulated so that the dura can be elevated and the mandibular nerve can be visualized. The petrosal nerves are located medially, just under the dura.
To expose the IAC, the superior petrosal sinus and petrous ridge must be identified. An incision is made above and below the ridge so that the sinus can be ligated just posterior to the porus trigeminus. The sinus must be ligated before the entry point of the superior petrosal vein so that it will drain normally into the transverse sinus. Utmost care must be exercised to protect the trochlear nerve, which courses just below the tentorium and easily can be damaged. After the mandibular nerve, GSPN, and vestibulocochlear complex have been identified in the IAC and the superior petrosal sinus has been ligated, this piece of bone, which is devoid of important structures, can be drilled until the dura of the posterior fossa becomes visible.
In this exposure the basilar artery, the emergence of the AICA, and the abducens nerve are visible medially. The nerves inside the IAC are visible posteriorly. The brain stem is exposed from the medullopontine sulcus, and the anterolateral portion of the pons between the trigeminal and facial nerves is also visible.
Conclusions
The IAC can be approached from different directions, and the choice of approach is based on clinical and imaging factors. A team approach offers patients the expertise from two surgical specialties: neurosurgery and neurotology. Expertise in microneurosurgery and in-depth knowledge of the temporal bone anatomy are key for treating patients with acoustic neuromas.
The suboccipital-retrosigmoid approach is the workhorse procedure. It is useful for tumors of all sizes. Preservation of hearing is based on preoperative hearing and tumor size. For pure intracanalicular tumors, the translabyrinthine or middle fossa are reasonable options. Progressive drilling of the petrous bone is the basis for the transpetrosal approaches, which were developed to maximize bone resection to avoid retraction over the cerebellum. The progressive drilling, however, is associated with intrinsic complications (e.g., hearing loss, facial paralysis, CSF leakage). If preservation of hearing is a goal, the middle fossa or retrosigmoid approach would be preferable. If hearing is severely compromised, the translabyrinthine approach is indicated. Transcochlear and transKawase approaches are indicated for large tumors primarily anterior to the pons, clival lesions, and, rarely, acoustic schwannomas. Partial labyrinthectomy is promising, but larger series are needed to determine its efficacy for hearing preservation. The middle fossa approach is an option for removing small acoustic neuromas (inside the IAC).
References
- Ammirati M, Spallone A, Feghali J, et al: The endolymphatic sac: Microsurgical topographic anatomy. Neurosurgery 36:416-419, 1995
- Brazis PW, Masdeu JC, Biller J: The localization of lesions affecting cranial nerve VII (the facial nerve), in Brazis PW, Masdeu JC, Biller J (eds): Localization in Clinical Neurology. Boston: Little, Brown and Company, 1996, pp 271-291
- David CA, Spetzler RF: Petroclival meningiomas. BNI Quarterly 15:4-14, 1999
- Day JD, Chen DA, Arriaga M: Translabyrinthine approach for acoustic neuroma. Neurosurgery 54:391-395, 2004
- Day JD, Fukushima T, Giannotta SL: Microanatomical study of the extradural middle fossa approach to the petroclival and posterior cavernous sinus region: Description of the rhomboid construct. Neurosurgery 34:1009-1016, 1994
- de Oliveira E, Tedeschi H, Rhoton AL, Jr., et al: Microsurgical anatomy of the internal carotid artery: Intrapetrous, intracavernous, and clinoidal segments, in Carter LP, Spetzler RF (eds): Neurovascular Surgery. New York: McGraw-Hill, 1995, pp 3-10
- Fisch U: Infratemporal fossa approach to tumours of the temporal bone and base of the skull. J Laryngol Otol 92:949-967, 1978
- Fukushima T, Day JD: Manual of Skull Base Dissection. Pittsburgh: AF NeuroVideo, 1996
- Harner SG, Beatty CW, Ebersold MJ: Impact of cranioplasty on headache after acoustic neuroma removal. Neurosurgery 36:1097-1099, 1995
- House WF: Surgical exposure of the internal auditory canal and its contents through the middle, cranial fossa. Laryngoscope 71:1363-1385, 1961
- House WF: Middle cranial fossa approach to the petrous pyramid: A report of 50 cases. Arch Otolaryngol 78:460-469, 1963
- House WF, Hitselberger WE: The transcochlear approach to the skull base. Arch Otolaryngol 102:334-342, 1976
- House WF, Shelton C: Middle fossa approach for acoustic tumor removal. Otolaryngol Clin North Am 25:347-359, 1992
- Kanzaki J, Kawase T, Sano K, et al: A modified extended middle cranial fossa approach for acoustic tumors. Arch Otorhinolaryngol 217:119-121, 1977
- Kawase T, Shiobara R: Extended middle cranial fossa approaches to the clivus and acoustic meatus, in Torrens M, Al-Mefty O, Kobayashi S (eds): Operative Skull Base Surgery. New York: Churchill Livingstone, 1997, pp 263-278
- Kawase T, Shiobara R, Toya S: Anterior transpetrosal-transtentorial approach for sphenopetroclival meningiomas: Surgical method and results in 10 patients. Neurosurgery 28:869-875, 1991
- Kawase T, Toya S, Shiobara R, et al: Transpetrosal approach for aneurysms of the lower basilar artery. J Neurosurg 63:857-861, 1985
- Martin RG, Grant JL, Peace D, et al: Microsurgical relationships of the anterior inferior cerebellar artery and the facial-vestibulocochlear nerve complex. Neurosurgery 6:483-507, 1980
- Parry RH: A case of tinnitus and vertigo treated by division of the auditory nerve. J Laryngol 19:402-406, 1904
- Post KD, Eisenberg MB, Catalano PJ: Hearing preservation in vestibular schwannoma surgery: What factors influence outcome? J Neurosurg 83:191-196, 1995
- Rhoton AL Jr: The cerebellopontine angle and posterior fossa cranial nerves by the retrosigmoid approach, in Rhoton AL Jr (ed): Rhoton: Cranial Anatomy and Surgical Approaches. Baltimore, MD: Lippincott Williams & Wilkins, 2003, pp 525-561
- Rhoton AL Jr: The temporal bone and transtemporal approaches, in Rhoton AL Jr (ed): Rhoton: Cranial Anatomy and Surgical Approaches. Baltimore, MD: Lippincott Williams & Wilkins, 2003, pp 643-697
- Sekhar LN, Schessel DA, Bucur SD, et al: Partial labyrinthectomy petrous apicectomy approach to neoplastic and vascular lesions of the petroclival area. Neurosurgery 44:537-550, 1999
- Wilson-Pauwels L, Akesson EJ, Steward PA: Glossopharyngeal nerve, in Wilson-Pauwels L, Akesson EJ, Steward PA (eds): Cranial Nerves. Anatomy and Clinical Comments. Toronto, Ontario: B.C. Decker, 1988, pp 114-123