
Cerebral Sinovenous Thrombosis in Children: Review and Case Report
Mohammad Khan, BS*
Shahram Partovi, MD
C. Roger Bird, MD
Division of Neuroradiology, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona *Medical Student, College of Medicine, University of Arizona, Tucson, Arizona
Abstract
As increasingly sophisticated neuroimaging technology becomes available, the frequency with which cerebral sinovenous thrombosis is detected in children is likewise increasing. However, the subtle and variable clinical presentations associated with the condition make its diagnosis difficult. We present a 7-week-old girl with seizures who underwent MR imaging, which showed thrombosis of the straight sinus and internal cerebral veins. She was treated with antiepileptic and anticoagulant therapy and recovered uneventfully within 2 weeks. Follow-up MR venography showed recanalization of the vessels, and the patient was discharged home. The key to treating this potentially devastating disease is a solid understanding of the variable presentations of cerebral sinovenous thrombosis in children and of the differences in imaging findings between neonates and adults. When combined with a multidisciplinary approach involving the referring physician and neuroradiologist, this knowledge will help to avoid the neurological complications associated with delayed diagnosis and treatment.
Key Words: sinovenous thrombosis, children, seizures, prothrombotic states
Abbreviations used: CT, computed tomography; CSVT, cerebral sinovenous thrombosis; MR, magnetic resonance; PICU, pediatric intensive care unit
Cerebral sinovenous thrombosis is a rare but potentially serious disorder in children. It is now increasingly diagnosed because clinical awareness of the entity and neuroimaging techniques have improved.[8] Furthermore, the survival rate of children with previously lethal diseases conferring a predisposition to sinovenous thrombosis has increased.[8] It is estimated that the annual incidence of CSVT is 0.29 cases per 100,000 children per year. Neonates are the most commonly affected age group.[4] CSVT is now considered more common than previously believed, and with subtle and variable clinical presentations, it still remains a diagnostic challenge.
Case Report
A 7-week-old girl was born prematurely after the amniotic fluid ruptured at 36 weeks of gestation. She had a 2- to 3-day history of respiratory distress. She was placed on a ventilator for respiratory support, admitted to the PICU, and treated with antibiotics and intravenous fluids. She recovered in a few days and was transferred to the pediatrics ward. Ten days after admission, she was noted to have rhythmical jerking of the upper extremities that lasted about 1 minute. She had more episodes and was returned to the PICU where she had another tonic-clonic seizure. She was loaded with the antiepileptic drug, fosphenytoin, which helped eliminate the seizures.
On readmission to the PICU, the patient was slightly febrile (100.1º F) and had normal tone and reflexes with strong pulses and perfusion of her extremities. Her extraocular movements were intact, and her pupils were equal, round, and reactive to light and accommodation. The rest of her physical examination was normal.
On Day 10 her metabolic profile was as follows: sodium, 138 mEq/L; potassium, 3.8 mEq/L; chloride, 105 mEq/L; bicarbonate, 20 mEq/L; blood-urea-nitrogen level, 5 mg/dL; creatinine, 0.2 mg/dL; and glucose, 80 mg/dL. Her white blood cell count was 4.13 x 103/mL, hemoglobin was 13.3 gm/dL, and hematocrit was 38.6%. Her international normalized ratio was 1.0, prothrombin time was 10.6 sec, and partial thromboplastin time was elevated to 37 sec. Further coagulation studies revealed decreased levels of protein S (52 mg/L), protein C (30 mg/L), and antithrombin III (43 mg/dL). Blood-gas analysis was normal.
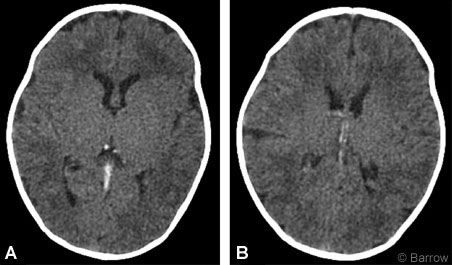
Electroencephalography revealed abnormal, poorly organized activity with some sharp transients over the temporal lobe regions, but there was no evidence for ongoing seizure activity. Head CT with contrast revealed increased density of the straight sinus and internal cerebral veins (Fig. 1). On MR imaging and MR venography, there was no flow within the straight sinus and internal cerebral veins, consistent with thrombosis (Fig. 2).
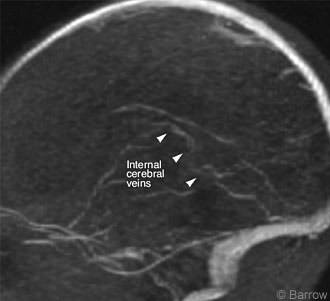
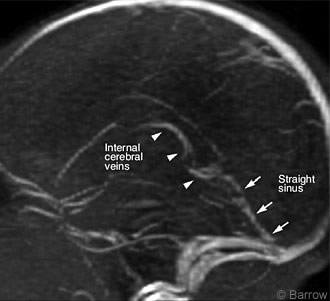
The patient was monitored in the PICU for several days and returned to the pediatrics ward. She was treated with lovenox and dilantin for seizure activity and CSVT. MR imaging repeated 1 week later showed blood flow through the straight sinus and internal cerebral veins (Fig. 3). During this time, the girl’s condition had improved, and she was neurologically intact upon examination. She suffered no complications and was discharged home on lovenox and phenobarbital with instructions to follow-up with her primary-care physician.
Discussion
Clinically, CSVT manifests differently in neonates and older children, suggesting that the presenting features can be described as age-dependent. Seizures, fever, lethargy, or irritability and respiratory distress are common signs in neonates.[4] In contrast, older children often become symptomatic with the classic signs of intracranial hemorrhage, such as vomiting, headache, papilledema, and abducens nerve palsy.[4,8] Other presentations can vary from minimal and nonspecific symptoms such as decreased oral intake and irritability to more ominous signs such as coma and hemiplegia.[13]
Diagnosis
The diagnosis of CSVT is confirmed by various neuroimaging studies. The widespread availability of CT, MR imaging, and MR venography has allowed earlier and more efficient diagnosis. In previous studies, CT scans were obtained in 96% of children, but the diagnosis was missed in 10 to 20% and the extent of thrombosis was underestimated in a few cases.[4,8] Especially in neonates, CT scans have been associated with false-positive results because of increased hematocrit, decreased density of unmyelinated white matter, and slower venous flow.[8] These findings can mimic the dense triangle sign on CT, which represents opacification of the torcula by freshly thrombosed blood.[4] Other signs on CT include he cord sign, which is a linear area of increased density related to thrombus in veins and sinuses, and the empty delta sign, which is an area of hypodensity in the torcula correlated with thrombus in the sinus surrounded by contrast. The latter is the most common sign, present in 30% of cases.[16] Other findings include dural and tentorial enhancement and dilatation of transcerebral intermedullary veins. Brain involvement may show hemorrhagic infarcts, edema, compression, or enlargement of ventricles.
The optimal technique for establishing the diagnosis is MR imaging with MR venography.[8] These modalities show details of the infarct and visualize the absence of flow and presence of thrombus.[8] Acute thrombosis is isointense on T1-weighted and markedly hypointense on T2-weighted MR images, while both studies show increased intensity in the intermediate stage.[13] MR angiography has been used in doubtful or difficult cases. It provides more detail on the partial or complete lack of filling of vessels, the size of enlarged collateral veins, a decrease in venous emptying, and the reversal of normal venous flow.[4]
Pathogenesis
Thrombosis of the sinovenous system usually is caused by vessel stasis, prothrombotic states, and damage to the vessel wall (Virchow’s triad). Slower blood flow leads to propagation of thrombosis while mural damage helps to perpetuate clot formation by serving as a nidus. Ischemia results when impaired venous drainage causes congestion and swelling of underlying white matter.[4] There is also a lack of thrombomodulin, which normally provides an antithrombotic effect.[11] It is believed that neonates may be at a higher risk for CSVT because of mechanical disruption. Because sinuses are located along suture lines, calvarial molding during birth can predispose infants to the development of thrombus.[14]
The superior sagittal and transverse sinuses are the most common sites involved.[4] In a study of 197 children, 86% had thrombus in the superficial sinuses, while 38% had thrombus in the deep sinuses.[8] Multiples sites were involved in 49% of the children. The transverse sinus was more likely to be involved in non-neonates (60%) than in neonates (39%), but there were no significant differences between deep and superficial involvement.[8]
CSVT is associated with a variety of local or systemic conditions (or both) that can be age-dependent. In one study, perinatal complications, such as hypoxic encephalopathy, predominated in neonates.[8] Altered cerebral hemodynamics such as shock and dehydration have also occurred in this age group.[3,15] Head and neck infections that led to septic thrombosis, such as otitis media, mastoiditis, and sinusitis, predominated in preschool children. Chronic conditions, such as connective tissue disorders (e.g., systemic lupus erythematosus, rheumatoid arthritis, and nephritic syndrome), were more frequent in older children.[5] Certain drugs, including oral contraceptives, which are increasingly used by adolescent girls, also predispose to CSVT.[2] Risk factors in adults such as pregnancy and cancer were nonapplicable or rare in this study.
The most common prothrombotic state in neonates and children is the presence of factor V Leiden.[12,17] Other common genetic abnormalities include protein C, protein S, and antithrombin III factor deficiencies. In children with CSVT, the frequency of prothrombotic disorders is 12 to 50%,[1,9] which is higher than the frequency in adults (i.e., 15 to 21%).[7] The most common acquired disorder is the presence of anticardiolipin antibody.[8] In about 3% of children, a cause for CSVT cannot be found despite extensive search, as compared to 25% in adults.[10] Caution must be exercised when diagnosing idiopathic CSVT because continued surveillance may uncover causes that can be treated.
Treatment
The mainstay of treatment for CSVT is conservative medical and neurological supportive care, including hospitalization with adequate hydration and close monitoring. Aggressive antibiotic therapy and antiepileptics should be used when appropriate. Whether systemic fibrinolysis and anticoagulation therapy should be administered to pediatric patients is still controversial[4] because data regarding its safety and efficacy in this population are limited.
Some authors have treated patients with heparin and urokinase with good results;[6] others have recommended that anticoagulation not be used. Isolated reports have shown improvement in patients with extensive thrombosis and a low incidence of complications. Nevertheless, in the newborn period, CSVT is frequently a self-limited disease requiring no treatment. In children who are symptomatic, treatment should be based on clinical judgment because a real consensus about the best treatment for severe cases is lacking.
Prognosis
The long-term neurological outcome in children largely remains unclear.[10] In one estimate 77% of neonates and 52% of nonneonates were completely normal after a mean of 2.1 years.[13] According to a recent study,[6] children who survive CSVT have a fair prognosis. In this study, of the 12 children younger than 16 years old who survived CSVT, only 1 had mild neurological deficits at a mean follow-up of 2.8 years. Cognitive functioning was normal in all children except two, who had mild cognitive problems. In terms of quality of life, three children suffered from negative well being (i.e., feelings of decreased self-esteem, depression, and anxiety). The findings from this study also suggest that neonates have a worse prognosis compared to their older counterparts. Long-term follow-up is vital because the onset of neurological injury is delayed in children, especially in neonates.
Conclusion
The variable clinical presentation of CSVT in children and its unique radiological findings in neonates and older children must be understood to ensure prompt diagnosis of patients afflicted with this disease and to avoid the neurological complications associated with its delayed diagnosis and treatment.
References
- Bonduel M, Sciuccati G, Hepner M, et al: Prethrombotic disorders in children with arterial ischemic stroke and sinovenous thrombosis. Arch Neurol 56:967-971, 1999
- Buchanan DS, Brazinsky JH: Dural sinus and cerebral venous thrombosis. Incidence in young women receiving oral contraceptives. Arch Neurol 22:440-444, 1970
- Carvalho KS, Bodensteiner JB, Connolly PJ, et al: Cerebral venous thrombosis in children. J Child Neurol 16:574-580, 2001
- Carvalho KS, Garg BP: Cerebral venous thrombosis and venous malformations in children. Neurol Clin 20:1061-1077, 2002
- Crassard I, Bousser MG: Cerebral venous thrombosis. J Neuroophthalmol 24:156-163, 2004
- De Schryver EL, Blom I, Braun KP, et al: Longterm prognosis of cerebral venous sinus thrombosis in childhood. Dev Med Child Neurol 46:514-519, 2004
- Deschiens MA, Conard J, Horellou MH, et al: Coagulation studies, factor V Leiden, and anticardiolipin antibodies in 40 cases of cerebral venous thrombosis. Stroke 27:1724-1730, 1996
- deVeber G, Andrew M, Adams C, et al: Cerebral sinovenous thrombosis in children. N Engl J Med 345:417-423, 2001
- deVeber G, Monagle P, Chan A, et al: Prothrombotic disorders in infants and children with cerebral thromboembolism. Arch Neurol 55:1539-1543, 1998
- deVeber GA, MacGregor D, Curtis R, et al: Neurologic outcome in survivors of childhood arterial ischemic stroke and sinovenous thrombosis. J Child Neurol 15:316-324, 2000
- Lin JH, McLean K, Morser J, et al: Modulation of glycosaminoglycan addition in naturally expressed and recombinant human thrombomodulin. J Biol Chem 269:25021-25030, 1994
- Martinelli I, Sacchi E, Landi G, et al: High risk of cerebral-vein thrombosis in carriers of a prothrombin-gene mutation and in users of oral contraceptives. N Engl J Med 338:1793-1797, 1998
- Medlock MD, Olivero WC, Hanigan WC, et al: Children with cerebral venous thrombosis diagnosed with magnetic resonance imaging and magnetic resonance angiography. Neurosurgery 31:870-876, 1992
- Newton TH, Gooding CA: Compression of superior sagittal sinus by neonatal calvarial molding. Radiology 115:635-640, 1975
- Tyler HR, Clark DB: Cerebrovascular accidents in patients with congenital heart disease. AMA Arch Neurol Psychiatry 77:483-489, 1957
- Virapongse C, Cazenave C, Quisling R, et al: The empty delta sign: Frequency and significance in 76 cases of dural sinus thrombosis. Radiology 162:779-785, 1987
- Zuber M, Toulon P, Marnet L, et al: Factor V Leiden mutation in cerebral venous thrombosis. Stroke 27:1721-1723, 1996
