Sattler Laboratory
Laboratory Focus
Our laboratory studies the cellular and molecular mechanisms of neurodegeneration in central nervous system (CNS) disorders such as:
We are particularly interested in elucidating the role of synaptic dysfunction in these diseases. We utilize different models to study synaptic biology including patient-derived human induced pluripotent stem cells (hiPSCs), patient autopsy brain tissue, and animal models of disease. We employ state-of-the-art molecular, biochemical, and microscopy technologies.
Goals
The research interest of our laboratory lies in the elucidation of disease pathways in neurodegenerative disorders including, but not exclusively, ALS and FTD. Specifically, we focus on the role of the synaptic structures and synaptic proteins during disease progression, with an emphasis on the regulation and dysregulation of the synaptic glutamate homeostasis.
With the development of innovative techniques for acquiring adult patient-derived human induced pluripotent stem cells (hiPSCs), we have adopted and validated this extremely valuable human cell culture platform to better understand the human molecular and cellular aspects of disease pathways. Human culture model systems will allow us to overcome known interspecies differences, which are especially critical at the level of transcriptional regulation and RNA processing and for the translation of our basic science discoveries into the clinic.
Using a combination of advanced molecular, cellular, and imaging technologies, together with hiPSCs, patient autopsy brain tissue, and novel animal models, we aim to discover novel pathways that harbor potential specific drug targets for future therapeutic development.
Ongoing research in our laboratory is focused on the discoveries of the molecular mechanisms of the newly identified gene mutation in C9orf72. This mutation is characterized by an expanded GGGGCC hexanucleotide repeat in the noncoding region of the C9orf72 gene on chromosome 9p21 and represents the most common genetic abnormality in FTD (10-30%) and ALS (20-50%). Specifically, we are interested in the contribution of synaptic dysfunction to C9orf72-mediated cognitive impairments observed in C9 patients.
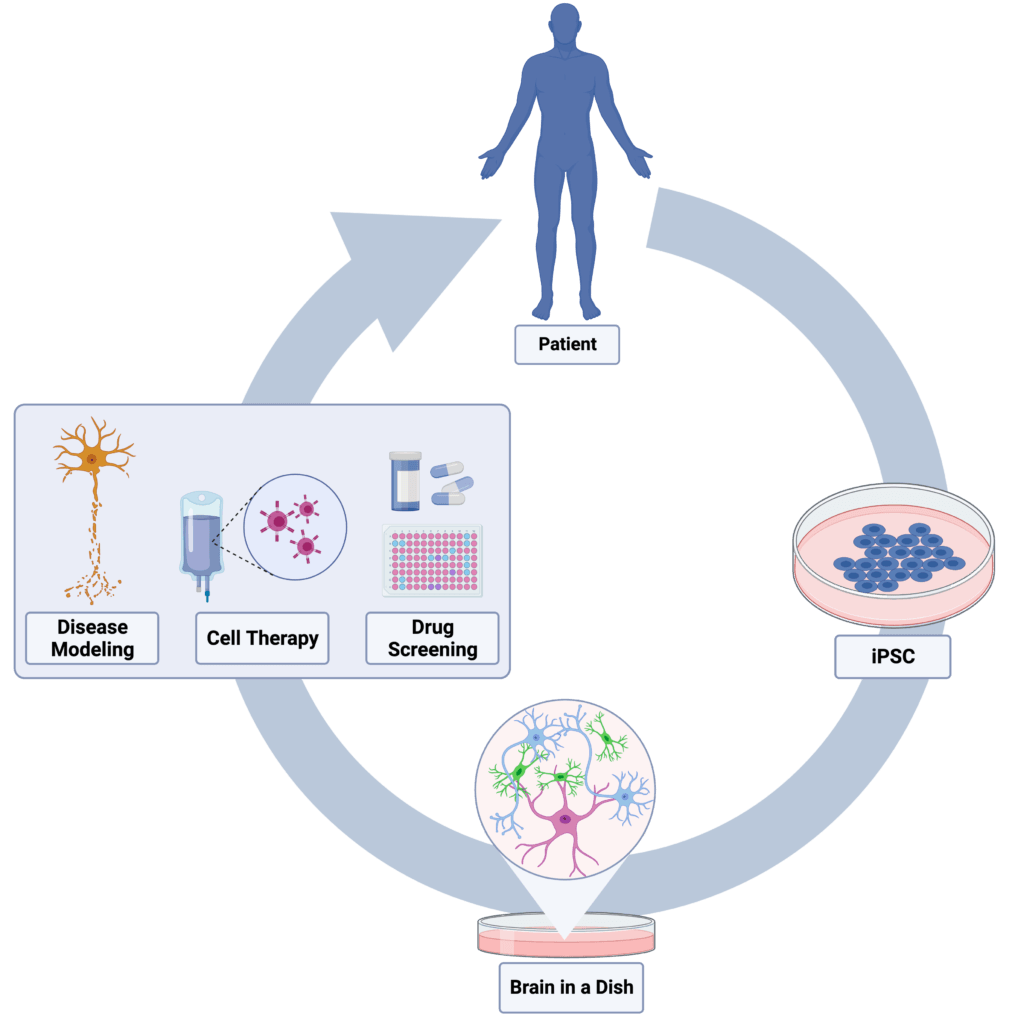
The Sattler Laboratory’s research focus lies in the elucidation of mechanisms of neurodegeneration in different forms of dementia. We use a human model system, including patient autopsy tissues and patient-derived induced pluripotent stem cells (iPSC) differentiated into neurons and glial cells to generate a “human brain in a dish.” The dementias we study include Frontotemporal Dementia (FTD) with and without Amyotrophic Lateral Sclerosis (ALS); Alzheimer’s disease (AD), and Lewy Body Dementias (LBD).
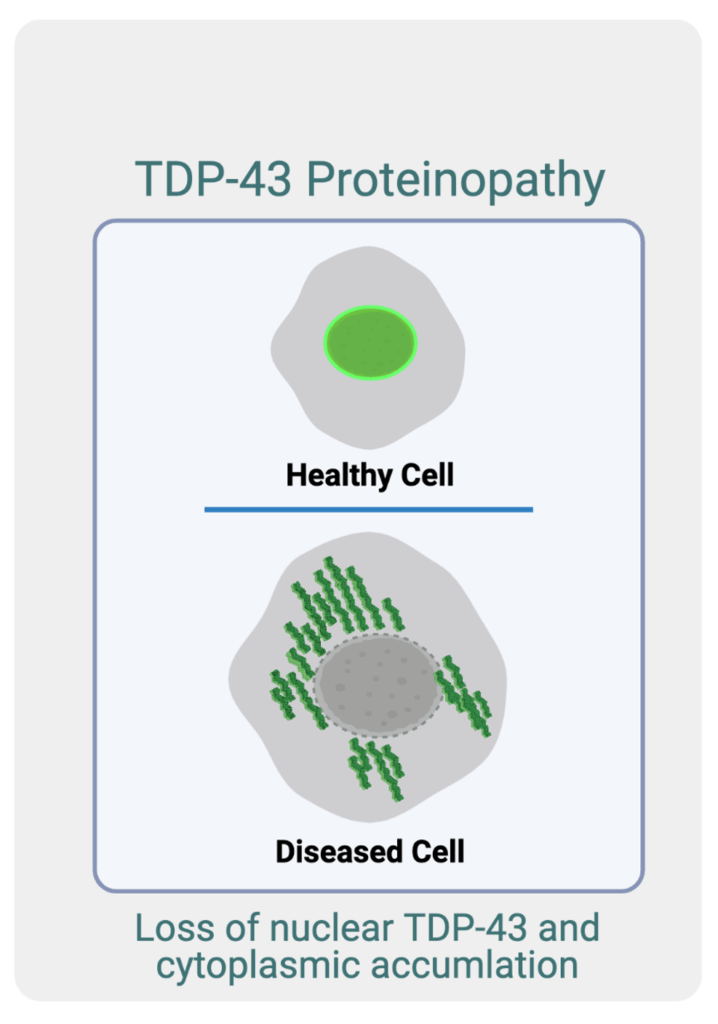
Mechanisms of A-I RNA Editing-mediated Nuclear Export of TDP-43
Funding Source: R21 NS130492-01
DP-43 is a protein that is primarily found in the nucleus of most cells in the human body. There, it is involved in many important activities necessary for cellular function and survival. In 2006, it was reported that in Frontotemporal dementia (FTD) and in Amyotrophic Lateral Sclerosis (ALS), TDP-43 is primarily found outside of the cell’s nucleus in the cytoplasm where it accumulates into toxic aggregates, also described as TDP-43 proteinopathy (see image to the right). Follow up studies suggested that this proteinopathy significantly contributes to neuronal cell death in FTD and ALS, which is the cause of disease symptoms in these disorders. Even though these aggregations were discovered over 15 years ago, we still do not know how TDP-43 gets transported from the nucleus to the cytoplasm, and once there, how it contributes to neuronal cell death.
This R21 award is aimed at investigating a novel mechanism explaining how TDP-43 gets trafficked out of the cell nucleus into the cytoplasm. This mechanism involves molecular changes in the nucleus known as A-I RNA editing, triggered by yet another protein called ADAR2. The Sattler laboratory has preliminary data to support the idea that increased A-I RNA editing transforms existing RNA molecules into chaperones, which then in turn bind to TDP-43 and guide it out of the nucleus. The proposed studies will help uncover the molecular identity of these chaperones. The Sattler lab proposes that this knowledge will enable the targeting of these chaperones therapeutically to prevent their binding to TDP-43, and consequently the transport of TDP-43 into the cytoplasm, which in turn will protect neurons from dying and improve disease symptoms.
Collaborators of this project include Dr. Kendall Van Keuren-Jensen (TGen) and Dr. Gene Yeo (University of California San Diego and Salk Institute).
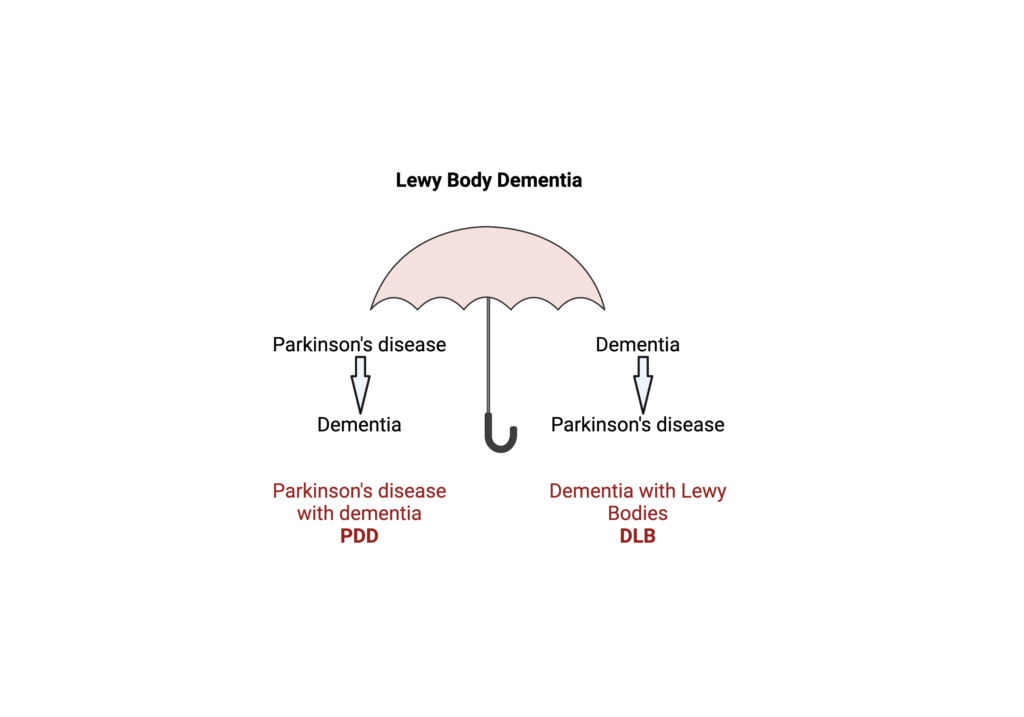
Transcriptomic Assessment of Pathology in PD with Dementia and Dementia with Lewy Bodies Using iPSC Neurons and Brain Tissue of the Same Individuals
Funding Source: R21 NS1285550-01
Lewy body dementia (LBD) is a spectrum disease that includes dementia with Lewy bodies (DLB) and Parkinson’s disease dementia (PDD). The two dementias share neuropathological characteristics of so-called Lewy bodies, in addition to pathologies related to Alzheimer’s disease – amyloid-beta plaques and/or neurofibrillary tangles of hyperposphorylated tau. One of the most distinct differences between PDD and DLB is the temporal occurrence of motor impairments relative to cognitive impairments. PDD patients develop PD before showing signs of dementia, while patients with DLB have dementia symptoms first, and then over time develop Parkinson’s disease-related movement dysfunctions. This often challenges an accurate diagnosis and consequently appropriate patient enrollment in clinical trials, patient care, and existing symptomatic treatment.
To better understand the distinct temporal progression of these two dementias, this R21 grant proposal will generate patient-derived human brain cells utilizing the groundbreaking technology of induced pluripotent stem cell (iPSC) culture models. This disease model system offers personalized patient analyses and allows for the examination of disease progression in PDD compared to DLB in a temporal manner. To ensure that this model system accurately reflects the individual patient’s disease pathogenesis, the study will generate iPSCs differentiated into neurons from individuals from which there also exists postmortem autopsy tissue. This provides the research team with the unique opportunity to directly compare molecular transcriptomic information and disease pathology from brain tissue and differentiated iPSC-neurons from the same individual.
These studies will for the first time investigate the transcriptome profile of PDD and DLB, examine cellular disease phenotypes in a temporal manner, and address the disease mechanisms of LBD in this spectrum disorder. Additionally, this work will provide novel opportunities for drug target identification with the hope of identifying novel therapeutics and biomarkers specific for each of the two disorders. This in return will facilitate the much-needed improvement of disease diagnosis and management of PDD and DLB patients.
Collaborators on this project include Dr. Kendall Van Keuren-Jensen (TGen) and Drs. Serrano and Beach (Banner Sun Health Research Institute).
Contribution of Microglia and Astrocytes to FTD/ALS-Mediated Cortical Neurodegeneration
Funding Sources: R01 NS120331-01; R21 NS125861; R21 NS125861-S1; Barrow Neurological Foundation
To better understand how patients develop FTD/ALS, and most importantly, how we can provide specific treatments for these patients, we are investigating non-neuronal cell-to-cell communication and regulation. This is an aspect of FTD disease mechanisms that has largely been ignored in recent years as most attention has been given to neuronal dysfunction. To study these non-neuronal cells (microglia and astrocytes) and its crosstalk, we will create a human disease model, or a “brain in a dish.” This model consists of patient-derived adult stem cells that we can convert into brain-like microglia and astrocytes.
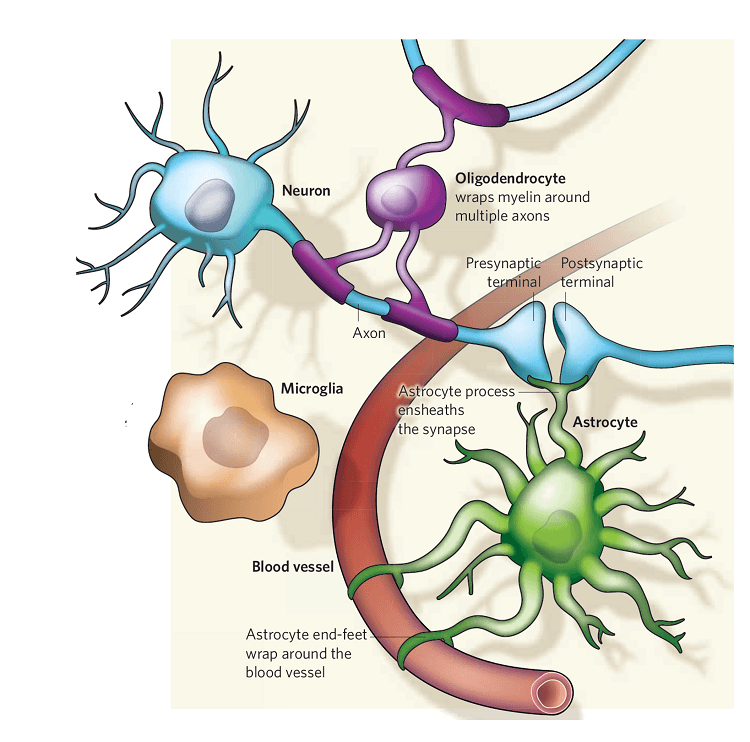
We will study these brain cells using molecular and cellular techniques with the goal of uncovering how microglia regulate astrocytes and vice versa and, most importantly, how this glial coregulation affects neuronal dysfunction in FTD/ALS patients. Once we have accomplished this, we plan to test novel therapeutics using these patient brain cells to determine which drug will prevent the cells triggering neurodegenerative disease mechanisms—a process we know happens in the brain of these patients and eventually contributes to disease progression and deterioration of health.
Collaborators of this project include Dr. Kendall Van Keuren-Jensen (TGen), Dr. Tania Gendron (Mayo Clinic Jacksonville), and Dr. Justin Ichida (University of Southern California).
Single-Cell RNA Sequencing Analyses of C9orf72 Human Brain Tissues
Funding Sources: DOD PR180487; ALSA Milton Safenowitz Postdoctoral Fellowship (To Dr. Lauren Gittings)
To gain more knowledge about how mutations in C9orf72 lead to FTD, we are generating a genetic signature profile of postmortem patient tissues specifically affected in FTD patients—the frontal cortex. We use a novel technology that will allow us to obtain a genetic profile of each individual cell type present in this brain region. While FTD leads to the degeneration of neurons, it is suggested that other cell types in the brain contribute equally to disease manifestation and progression. Using single cell RNA sequencing, we will be able to isolate these different cell types from the frontal cortex and look at the genetic makeup of each individual cell. By comparing the genetic profiles obtained from C9 FTD patients to those obtained from healthy control subjects, we can identify genes that are regulated differently, which are therefore likely to contribute to the disease pathogenesis.
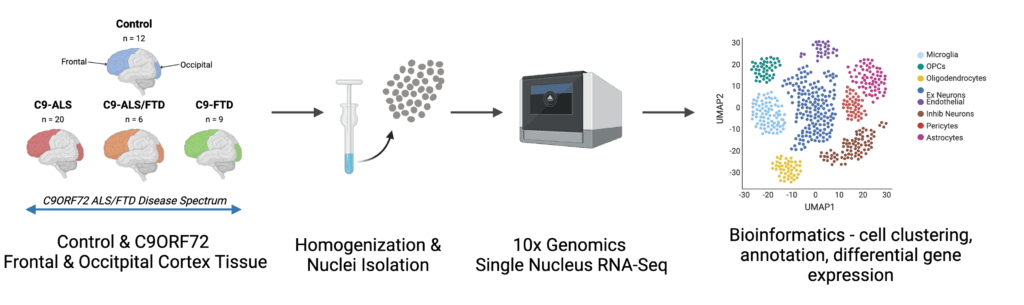
In addition to the analysis of postmortem tissue, we will generate and culture live human neurons from C9 FTD patients using another novel technology, induced pluripotent stem cells or iPSCs. To obtain iPSCs from C9 FTD patients, blood or skin fibroblasts from a patient will be converted into an iPSC, which can then be transformed into a neuron. These so-called cortical neurons grow in vitro and resemble the neurons present in the frontal cortex of humans. iPSC-cortical neurons from C9 FTD patients will then be analyzed for their genetic signature profile, similar to what is proposed for the postmortem frontal cortex brain tissue, and compared to those findings. We also will study neuronal function in these living neurons, for example neuronal electrical activities. Electrical firing of neurons is required for normal neuronal function, and deficiencies in these electrical properties are indicative of neuronal degeneration. By comparing C9 FTD cortical neurons to cortical neurons obtained from healthy control subjects, we will identify specific cellular phenotypes that are only present in the diseased cells, and which are therefore likely to contribute to disease progression.
Finally, after analyzing the genetic signature profiles from postmortem tissue and individual patient-derived cells, we will select candidate genes that we hypothesize play a significant role in C9 FTD disease pathogenesis. To test whether these genes will affect the behavioral cognitive phenotypes of C9 FTD patients, we will use a Drosophila (fruit fly) model of C9 FTD and use genetics to identify which of these candidate genes contribute to disease in an in vivo dementia model. Specifically, we will genetically manipulate flies to test whether specific gene mutations will affect the cognitive behavior of flies that have mutations in the C9orf72 gene, similar to C9 FTD patients. Those genes that either ameliorate or exacerbate the behavior of C9 flies will then be re-examined in the patient-derived iPSC cortical neurons and the patient postmortem tissue to validate findings from flies with humans as we have successfully done in the past for ALS
Collaborators of this project include Dr. Kendall Van Keuren-Jensen (TGen) and Dr. Daniela Zarnescu (Penn State University School of Medicine).
Targeting Synapse Loss in FTD/ALS using Spine-Regenerating Compounds
Funding Source: DOD AL200139
The goal of this proposal is to test the efficacy of a novel therapeutic small molecule compound in three different models of FTD/ALS. One of the disease mechanisms in FTD/ALS that is thought to occur early in disease development, independent of disease etiology, is the reduced ability of neurons to efficiently communicate with each other or, in the case of a motor neuron, to signal to connecting muscle tissues. These neuronal communications are happening at a very special cellular structure present on neuronal processes, called spines. These spines allow one neuron to link onto a neighboring neuron by forming a synapse. Many neurodegenerative diseases, including Alzheimer’s disease, but also FTD/ALS, are characterized by the loss of these spines and synapses. This is thought to eventually cause the degeneration of the neuron. The collaborating biotech company, Spinogenix Inc., has developed, synthesized, and optimized novel small molecule compounds that have been shown in other models of neurodegeneration to regenerate those lost spines and synapses.
We will test one of Spinogenix’s lead compounds for its spine regenerating activities in three different human ALS/FTD patient-derived induced pluripotent stem cell (iPSC) models (iPSC cortical neurons, iPSC motor neurons, and iPSC cortical organoids), as well as in a transgenic murine model of ALS/FTD. The murine studies will be accompanied by a biomarker evaluation of a synapse-specific PET imaging tracer which, if successful, can easily be translated into patient trials.
In parallel efforts, the proposed compound is undergoing preclinical toxicology evaluations. If the proposed studies show significant improvement and efficacy, this compound can be advanced into clinical trials for FTD/ALS patients rapidly. This is, to our knowledge, the only current therapeutic under evaluation to regenerate synapses lost in FTD/ALS. Therefore, our lead compound could be used as a monotherapy and, in principle, as a co-therapy with any among the large and diverse set of emerging therapeutics aimed at inhibiting neurodegeneration in FTD/ALS patients.
Collaborators of this project include Spinogenix Inc. and Dr. Justin Ichida (University of Southern California).
Role of TDP-43 Protein Aggregations in Alzheimer’s Disease
Funding Source: Barrow Neurological Foundation
TDP-43 is a protein that is primarily found in the nucleus of most cells in the human body. There, it is involved in many important activities necessary for cellular function and survival.
In 2006, it was reported that, in FTD, TDP-43 is primarily found outside of the cell nucleus in the cytoplasm, where it accumulates into toxic aggregates. This process is also described as TDP-43 proteinopathy (see image below). Follow-up studies suggested that this proteinopathy significantly contributes to neuronal cell death in FTD. Though these aggregations were discovered more than 15 years ago, we still do not know how TDP-43 is transported from the nucleus to the cytoplasm, and once there, how it contributes to neuronal cell death.
Over the last several years, TDP-43 aggregations have also been found in neurons in the brains of patients with Alzheimer’s disease (AD) and related dementias. Numerous studies have investigated the pathobiology of TDP-43 in FTD, but little is known about the molecular mechanisms and consequences of TDP-43 inclusions in AD, and whether these mechanisms differ from those identified in FTD. A better understanding of these mechanisms is critically important for the development of biomarkers and therapeutics for AD. The hypothesis is that removal of these cytoplasmic TDP-43 aggregates, or preventing their formation in the first place, will provide protection against neuronal cell death, which in turn will provide improvements of clinical symptoms and disease progression in AD patients.
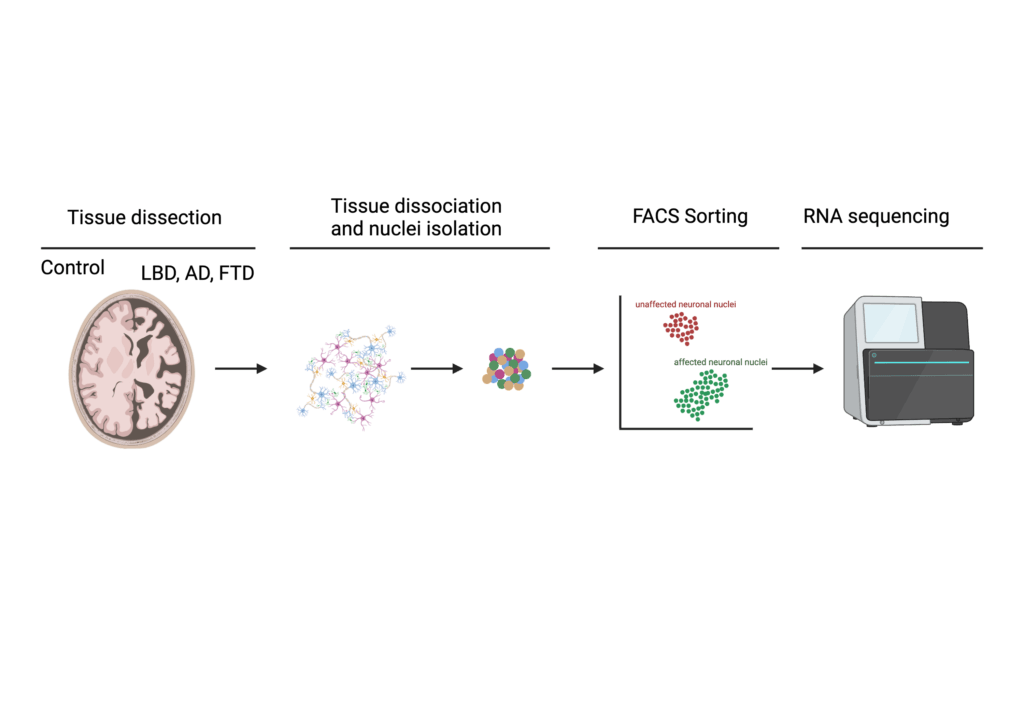
To understand how these protein aggregations damage neurons in the AD brain, we will utilize two separate approaches, both of which are based on human disease model systems.
First, we will analyze postmortem autopsy brain tissue generously donated from AD patients. A recently developed technology allows us to use these brain tissues to separate neurons affected from TDP-43 proteinopathy from unaffected neurons (see figure above). We will then analyze the genetic profile of these neurons and determine how changes in gene expression and gene modifications might contribute to neuronal dysfunction and degeneration.
Next, we will create an “AD brain in a dish.” We will accomplish this by collecting skin cells from patients with AD and, through sophisticated and novel technologies, transform these skin cells into brain cells. We will then carefully study these brain cells using molecular and cellular techniques with the goal of uncovering what distinguishes brain cells with TDP-43 proteinopathies from those without.
Once we have accomplished this, we plan to test novel therapeutics using these patient brain cells to determine which drug will prevent the cells from forming TDP-43 aggregations—a process we know happens in the brains of these patients and eventually contributes to disease progression and health deterioration.
Collaborators of this project include Dr. Kendall Van Keuren-Jensen (TGen).
Collaborative Projects
In addition to these major research projects, we have several small pilot projects in collaboration with local investigators from the valley:
- Investigation of the role of RNA dysregulation in TDP-43 FTD/ALS with Dr. Daniela Zarnescu (Penn State University School of Medicine). Funding source: R01 NS091299-06 (Zarnescu PI)
- Studies on genetic forms of cerebral palsy using patient-derived iPSC neurons in collaboration with Dr. Michael Kruer (Phoenix Children Hospital, University of Arizona). Funding Source: R01 pending (Kruer PI)

Professor, Translational Neuroscience

Contact Information
Rita Sattler, PhD
Professor, Translational Neuroscience
Barrow Neurological Institute
2910 North 3rd Avenue
Phoenix, AZ 85013
Rita.Sattler@BarrowNeuro.org
(602) 406-8685
Sattler Lab Funding
Our research is funded by the following organizations:
Sattler Laboratory News

May 2020
- Due to the COVID-19 pandemic, we have had reduced activities in the lab this month and most of the lab have been working remotely at home, analysing data, writing manuscripts, and submitting grant applications. Our graduate students are leading the efforts to get us back in the lab with Stephen, Lynette, and Ryan all returning to do experiments but trialing out and adhering to new social distancing measures.
- Friday, May 8 was “Drink one for Dane” day at Dutch Bros Coffee. This is an annual fundraising event for the Muscular Dystrophy Association (MDA) in honor of the co-founder, Dane Boersma, who died of ALS in 2009. Dutch Bros donates proceeds from each drink sold on this day. In 2019, the event raised more than $1.3 million for the MDA! To support this cause, several members of the lab took a trip to Dutch Bros to grab a to-go coffee.
March 2020
- At the beginning of March, several members of the lab attended the Annual Packard Center ALS Meeting in Baltimore. This is a three-day meeting focused on new and exciting research in the ALS field, with many speakers from all over the world. Members of the Sattler lab attend this meeting every. This year, it was Rita, Lauren, Stephen, Lynette, and Ryan who got to go. It was great to hear all about progress being made in the last year in ALS labs across the world. It was also a great opportunity for us to meet with collaborators and discuss our projects.
February 2020
- Congratulations to Stephen and Ben on the publication of their review!! Their article titled, “The Hitchhiker’s Guide to Nucleocytoplasmic Trafficking in Neurodegeneration,” was published on February 21 in the journal Neurochemical Research. It provides a comprehensive overview of the cellular mechanisms that regulate nucleocytoplasmic trafficking and how these mechanisms can fail in neurodegenerative diseases.
- S. Senator for Arizona, Martha McSally, visited Barrow Neurological Institute on February 21. As part of her visit, she was given a tour of our lab facilities and met with Dr Sattler to learn about the ALS research we are doing and how this could impact ALS patients.
- Dr Sattler recently attended the International Stroke Conference where investigators presented the results of a clinical trial for a new treatment of ischaemic stroke, nerinetide. Seeing the results of this trial was particularly special for Dr. Sattler because work she published during her PhD graduate studies at the University of Toronto in the 1990s helped pave the way for clinical trials of this neuroprotective drug. Although Rita’s research now primarily focuses on ALS, she likes to keep up to date with the progress of trials relating to her previous research. The Sattler lab are very proud of Rita and the contribution she has made to the stroke research community.
- Ben officially accepted an offer to join the Neuroscience PhD graduate program at the Mayo Clinic! Congratulations, Ben! You will be missed by all of us, and we look forward to seeing you succeed in your future graduate studies!
January 2020
- New year, new student! We would like to extend a warm welcome to Ryan Pevey who officially joined the lab in January. Ryan is a graduate student from the Interdisciplinary Graduate Program in Neuroscience at Arizona State University. Ryan’s research will be focused on investigating the role of the RNA-binding protein TDP-43 in RNA splicing and how these functions are dysregulated in ALS, FTD, and Alzheimer’s disease. Welcome to the lab, Ryan!
December 2019
- Dr. Sattler and Lynette headed to the U.K. at the beginning of December to participate in the Wellcome Trust Molecular Degeneration course. Dr. Sattler co-organizes this annual week-long residential course at the Wellcome Genome Campus near Cambridge (UK). The program provides an overview of the molecular basis of several neurodegenerative disorders—including Alzheimer’s disease, Parkinson’s disease, and ALS—and highlights experimental techniques used in neurogenetics, bioinformatics, and stem cell research that aid the development of therapeutic interventions to combat neurodegenerative disorders. Lauren and Stephen previously attended this course in 2018, and this year it was Lynette’s turn!
November 2019
- On Saturday, November 2, many members of the lab, plus their families and pets, teamed up with members of the Fulton ALS Center to take part in the 19th ‘Walk to Defeat ALS’ in Scottsdale. This was a three-mile sponsored walk in support of the ALS Association.
- Sattler Lab Grand Canyon Hike! After months of training by climbing the stairs, on November 12th the Sattler lab hiked to the bottom of the Grand Canyon and back up again in a day! This 19-mile challenge took us approximately 12 hours, but we all successfully made it out of the canyon together (with some people in better shape than others!) and have brilliant memories and photographs to prove it. Thank you to Rita for encouraging us to do this challenge and taking care of us while we did it! We can’t wait for next year’s challenge…
- On Friday, November 22, Ileana and Lauren attended the ‘Passport to a Cure’ gala in support of the Muscular Dystrophy Association (MDA). The MDA is just one of several organizations that supports research into ALS and other related neuromuscular diseases.
October 2019
- During October, several members of the lab traveled to Chicago for the annual Society for Neuroscience (SfN) conference. This is one of the largest scientific conferences in the world and is a great opportunity to learn about all sorts of fascinating neuroscience research going on in the world! At this year’s conference, Rita chaired a Nanosymposium on the Mechanisms of Motor Neuron Disease. Ileana, Jenny, and Stephen presented posters about their research. We really enjoyed discussing some of our projects and received great feedback from the scientific community.
- As a collaborating investigative team, the Sattler lab received a National Institutes of Health (NIH) supplement award, submitted by Dr. Daniela Zarnescu (UofA) to study the role of TDP-43 in cognitive impairment in FTD.
September 2019
- During the last weekend in September, nearly all members of the Sattler lab traveled down to University of Arizona in Tucson for the 4th annual Arizona ALS Symposium. This event brought together ALS researchers and clinicians from across Arizona, and provided an opportunity to share ideas, collaborate, and discuss cutting-edge ALS research. As part of the symposium, Lynette, Stephen, and Lauren presented posters showcasing the research they do in the lab. Congratulations to Stephen, whose poster won second prize in the poster competition!
- As a collaborating investigative team, the Sattler lab was awarded an NIH SBIR award, submitted by Aquilus Pharma, who is investigating the neuroprotective mechanisms of matrix metalloproteinase 2/9 inhibitors in models of ALS.
August 2019
- In August, Rita, Lynette, and Lauren attended one of the Robert Packard ALS Center’s Monthly Meetings in Baltimore. At the meeting, we met with several of our collaborators and Rita presented data from our lab’s work on C9orf72 iPSC-derived microglia.
July 2019
- Congratulations to Stephen on the publication of his first author paper! Stephen’s paper, “ADAR2 mislocalization and widespread RNA editing aberrations in C9orf72-mediated ALS/FTD,” was published in print in the journal Acta Neuropathologica in July 2019. Well done for all your hard work, Stephen!
- In July, we welcomed post-doctoral fellow Lauren Gittings to the lab. Lauren recently received her PhD from University College London (U.K.) and will be working on a project to uncover underlying transcriptional differences between patients that clinically present with FTD, FTD-ALS, and ALS.
- As a thank you for all the hard work our summer students have done over the summer months, the lab spent an afternoon having fun doing a puzzle room and going out for ice cream! We are very grateful for all the hard work that our students have done throughout the year and over the summer. Thank you for all your hard work Megan, Junny Kim, Nancy Twishime, Shiv Shah and Divya Bhatia!
- At the end of July, Rita, Ileana, and Stephen traveled to Vermont to attend the Gordon Research Conference on ‘Mechanisms of Motor Neuron Degeneration and Therapeutic Intervention. Rita was a co-organizer and chair of this meeting, and Stephen was a co-organizer of the related Gordon Research Seminar for scientific trainees. At the meeting, both Ileana and Stephen gave oral presentations about their research. Stephen presented his recently published work on “ADAR2 Mislocalization and Widespread RNA Editing Aberrations in C9orf72-Mediated ALS/FTD” and Ileana presented data on her project “Human In Vitro Culture Systems of C9orf72-ALS/FTD Patient-Derived iPSC Cortical Neurons and Microglial Cells to Study Mechanisms of Synaptopathy.”
June 2019
- In June, we welcomed graduate student Lynette Bustos to the lab. Lynette is part of the Interdisciplinary Graduate Program in Neuroscience at Arizona State University and will be working on a project using iPSC-derived astrocytes to decipher their role in FTD/ALS. Welcome to the lab, Lynette!
- New publication alert! Ileana, Ben, Alex, and Rita recently contributed to a collaboration with Daniela Zarnescu’s lab at the University of Arizona. This work, titled “Glycolysis upregulation is neuroprotective as a compensatory mechanism in ALS,” has now been published in the journal ELife. Congratulations to all of those involved in this work!
- The Sattler lab received some funding from the Arizona Alzheimer’s Reseearch Consortium (AARC) and the Barrow Neurological Foundation to study the role of the ADAR2 editing protein in Alzheimer’s disease.
May 2019
- May is ALS Awareness Month so every day throughout May 2019 the Sattler lab have been participating in events to raise awareness for ALS. To make it interesting, we chose to do the selected activity for the duration/amount to match the day of the month. Some of the activities we did included Stephen hiking five miles, Ben taking images of 21 neurons, Rita running for 22 minutes, Lauren baking 24 cupcakes, and Mo kayaking for 26 minutes. We finished off the month by participating in the “In Your Face ALS” challenge, where we took great pleasure in putting pies in each other’s faces! The lab would like to say a big thank you to Mo Roberts for coordinating the efforts of the lab throughout ALS Awareness Month!
- New publication alert! Ileana and Rita recently contributed to a collaboration with Jin Xu’s lab. This work, titled “Reactivation of nonsense-mediated mRNA decay protects against C9orf72 dipeptide-repeat neurotoxicity,” has now been published in the journal Brain. Congratulations to all of those involved in this work!
- Congratulation to our post-doctoral fellow, Ileana Lorenzini, who was awarded a Barrow Neurological Foundation grant to study the role of microglial cells in C9orf72-linked ALS/FTD synaptic dysfunction. Good luck with the project, Ileana!
March 2019
What a busy year… here are a few highlights of what has been going on:
Career developments
- Both Layla and Alex have moved on to graduate school this fall. Layla joined the Neuroscience Graduate Program at Thomas Jefferson University, and Alex joined the graduate program in the Department of Pharmacology at University of Pennsylvania.
- Our former high school students, Devika and Nancy, are almost done with their first year in college—Devika at UCLA and Nancy at ASU Barrett Honors College. Time flies!!
- Stephen passed his oral exam and is now officially a PhD candidate. Congratulations!
- Shiv will start his undergraduate training at ASU this fall! Congratulations, Shiv, we are excited to have you stay close by.
- Latavya is also starting her college training but has not yet picked a school. Fingers crossed that her first choice will become available.
- Both Latavya and Shiv participated in the Arizona Regional Brain Bee competition, and finished 1st and 2nd place, respectively. Congratulations! Latavya is now competing in the upcoming USA Regional Brain Bee Championship in Hershey, PA. Good luck, Latavya!
AZ: Basis Chandler Student Wins Regional Brain Bee
New lab members
- Ben Rabichow and Mo Roberts joined the lab as a research technicians in June 2018. It feels like these two have been with us forever – so great to have them on board!
- Latavya Chintada, a high school student from Basis High School Chandler, joined us last winter to work on her senior project with us. Welcome to the lab, Latavya!
- Junny Kim, a high school student from Scottsdale Preparatory Academy, joined the lab in the fall of 2018 and has since been tracing endless golgi stained neurons! So great to have her in the lab!
- Lauren Gittings, from University College London, London, UK, will be joining the lab as a postdoctoral fellow in July. We are very excited about her moving to the desert!
Science updates
- Stephen’s first manuscript was accepted at Acta Neuropathologica! Congratulations to Stephen and all of our collaborators!
- In September last year, we hosted our Third Annual Arizona ALS Symposium, this time at Northern University in Flagstaff. The symposium was attended by close to 100 participants from all over the state of Arizona and was, once again, a great success.
- The lab had a strong presence with numerous poster presentations at SFN 2018 in San Diego, CA.
- Stephen was picked to present his abstract in form of an oral presentation at the RNA Metabolism meeting in San Diego last November, held right before SFN. A great exposure for him to the experts in the field — and he did a terrific job with his presentation. Congratulations!
- Sattler lectured again at the Wellcome Trust Neurodegeneration Course in Hinxton/Cambridge, UK.
- Some of the members from the lab attended the annual Robert Packard Center for ALS Research Symposium in Baltimore, MD in March! Always a great meeting with lots of new and exciting science!
Funding updates
- In December 2018, Stephen was awarded for the second year a BNI Neurobiology science award! Congratulations!
- In March 2019, the Sattler lab was awarded a Partnering PI DOD research grant on C9orf72 FTD together with our collaborators Drs. Van Keuren-Jensen (TGen) and Zarnescu (UA Tucson).
- In July 2018, the Sattler lab was awarded a grant from the Arizona Alzheimer’s Research Consortium (AARC) with matching funds from the Barrow Neurological foundation on molecular aspects of dementia in sporadic ALS patients.
Other noteworthy news
- In October last year, the lab participated for the fourth year in the AZ ALSA walk with team Fulton ALS Center and raised a record high number in funds towards the Arizona ALSA chapter! Congratulations everybody, and thanks to those who supported us.
- Stephen and Dr. Sattler participated in the Climb to Defeat ALS, which raises funds for the ALS Association. They successfully climbed Mt. Elbert in Colorado, the highest 14er of the Rocky Mountains in North America. Stephen did reach the summit 30 sec before Dr. Sattler, earning the winning two bags of Starbursts!
May 2018
The lab has been incredibly busy over the last six months. Here are a few highlights of what has been going on:
Career developments
- Both Layla and Alex are moving on to graduate school this fall. Layla will join the Neuroscience Graduate Program at Thomas Jefferson University, and Alex accepted an offer from the graduate program in the Department of Pharmacology at University of Pennsylvania. We wish them both the best of luck and will certainly miss them!
- Our high school students Devika and Nancy are heading to college! Devika will be joining UCLA, while Nancy will head to ASU Barrett Honors College with a Flinn Foundation Scholarship. Congratulations to both of them!!
New lab members
- In October last year, Divya Bhatia joined the Sattler Lab as an undergraduate student from ASU and has since been an incredibly hard-working member of the lab.
- In November last year, Kevin Ma joined the Sattler Lab as a research technician. Welcome on board, Kevin!
- Ashley Nelson, a MD/PhD student from UofA College of Medicine Phoenix, did a research lab rotation in the Sattler Lab from Jan-April, studying the role of TDP43 in ALS/FTD.
Science updates
- In September last year, we hosted our Second Annual Arizona ALS Symposium, this time at Biosphere 2. The symposium was attended by close to 60 participants from all over the state of Arizona and was a great success.
- In December, Dr. Sattler in collaboration with Dr. Salvatore Oddo (ASU) hosted the first Neuro Division meeting for the Arizona Wellbeing Commons.
- The Sattler Lab co-authored two papers in collaboration with Dr. Robert Bowser’s laboratory (BNI) and Dr. Wilfried Rossoll’s laboratory (Mayo Clinic Jacksonville, FL):
https://www.ncbi.nlm.nih.gov/pubmed/29134320
https://www.ncbi.nlm.nih.gov/pubmed/29311743
See also the following link: http://web.alsa.org/site/PageNavigator/blog_010918.html
- Two invited review articles were published, one in the Special Edition of Brain Research and one in a Special Issue of Frontiers in Neuroscience, first-authored by Alex and Layla, respectively. Congratulations!!
https://www.ncbi.nlm.nih.gov/pubmed/29453960
https://www.ncbi.nlm.nih.gov/pubmed/29479303
- The lab has a strong presence with numerous poster presentations at SFN 2017 in Washington, DC.
- Sattler lectured again at the Wellcome Trust Neurodegeneration Course in Hinxton/Cambridge, UK. This year, Stephen was one of the students attending the course!
- Some of the members from the lab attended the annual Robert Packard Center Research symposium in Baltimore, MD! Always a great meeting with lots of new and exciting science!
Funding updates
- Stephen was awarded a BNI Neurobiology science award
- The Sattler Lab, in collaboration with Dr. Daniela Zarnescu’s lab (UofA Tucson), was awarded a DOD research grant
- The Sattler Lab was awarded a grant from the Robert Packard Center for ALS Research
Other noteworthy news
- In October last year, the lab participated for the third year in the AZ ALSA walk with team “Fulton ALS Center” and raised a record high number in funds towards the Arizona ALSA chapter! Congratulations everybody, and thanks to those who supported us.
- Doug, our ALS patient volunteer, was featured on the Barrow Newsletter and Barrow Facebook page. We are all so proud of Doug and all he does to advocate for ALS research!!
- Stephen and Dr. Sattler are participating in the ‘Climb to defeat ALS’, which raises funds for the ALS Association. The two will climb Mt. Elbert in Colorado, the highest 14er of the Rocky Mountains in North America. Please support team “Barrow Brainiacs” for this incredible adventure by donating money towards their fundraising efforts here.
September 2017
- A collaborative study under the leadership of Dr. Daniela Zarnescu’s laboratory at University of Arizona was accepted for publication at Cell Press! Congratulations to everybody!
- The Sattler Lab welcomes yet another research volunteer to the lab: Doug Clough – ALS patient, advocate, and ambassador!!! We are extremely excited to give Doug an opportunity to, as he says, ‘own his disease’ by having him work alongside the researchers in the lab. We keep questioning who is more excited about this opportunity, him or us?! Welcome, Doug!
August 2017
- Abigail and Nancy participated at the Barrow summer intern research symposium. Abigail gave a brilliant slide presentation, and Nancy effortlessly mastered her poster presentation. We are very proud of these two incredibly talented young scientists! Congratulations!
- Welcome to our new high school student intern, Shiv Shah. Shiv is a junior at Mountain Ridge High School.
July 2017
- Andrew left the Sattler Lab and Arizona to start his PhD program in Neuroscience at Thomas Jefferson University! Congratulations again and good luck to his future studies!
- Stephen presented his thesis project at the Barrow Tuesday seminar series. This was his first big audience presentation, and he excelled easily!
- Some of the Sattler Lab members attended the inaugural Gordon Conference on “ALS and related motor neuron diseases,” co-chaired by Dr. Sattler. Great science discussions in beautiful Vermont.
June 2017
- The lab welcomes our summer student Abigail Byford from the University of Leeds, UK, and Michael Goldstein, an undergraduate student from ASU.
- With 19:26 minutes left, the lab escaped successfully from the Pearled Pachyderm at the Phoenix Puzzle Room. One of the fastest groups to make it out of the room! Great team effort!!
- Ileana was awarded a postdoctoral fellowship award by the Barrow Neurological Foundation to study mechanisms of synaptic dysfunction in ALS and related disorders. Congratulations, Ileana!
- Nancy made headlines in the news when she was included in an article about women in STEM.
May 2017
- Stephen won the ASU Graduate and Professional Student Association (GPSA) Individual Travel Grant to present a poster at this year’s Gordon Conference on ALS. Congratulations, Stephen!
- In collaboration with the Mufson Laboratory, the Sattler Laboratory was awarded a research grant from the Barrow Neurological Foundation/Arizona Alzheimer’s Research Consortium (AARC) to study mechanisms of Down syndrome neurodegeneration in patient-derived iNeurons
- The lab had its second annual Salt River Tubing adventure – without any sunburns this year!
- Dr. Sattler participated in her first Barrow Neurological Institute Grand Canyon Rim-to-Rim hike! She made it through without major problems and is planning on a Rim-to-Rim-to-Rim for next year!
April 2017
- Devika presented a poster and won third place at the 2017 Arizona Science and Engineering Fair (AzSEF). Congratulations, Devika!
- The lab did a weekend camping trip to Prescott, Arizona. Lots of relaxing, hiking, cooking, and eating!
- The lab was awarded a pilot grant from the Arizona Alzheimer’s Disease Core Center (ADCC) to study patient-derived iPSC microglial cells in C9orf72 ALS/FTD
March 2017
- On March 2, Camelia gave birth to a healthy baby boy, Stefan. Everybody is excited about our first lab baby!
- Andrew was accepted into the neuroscience graduate program at Thomas Jefferson University in Philadelphia. Congratulations, Andrew!
- Layla Ghaffari joined the laboratory as a research technician.
- Ileana was invited to give a presentation at the Synapses and Circuits: Formation, Function and Dysfunction Keystone Symposium in Santa Fe, New Mexico.
February 2017
- Devika won first place at the BASIS High School Science Fair and is moving on to compete at the 2017 Arizona Science and Engineering Fair (AzSEF) to be held in April in Phoenix. Congratulations, Devika!
January 2017
- Stephen received a graduate student spring fellowship from Arizona State University.
- Sarah was awarded a Bidstrup Undergraduate Fellowship to support her honor’s thesis research project.
- The laboratory was awarded a research grant by the Robert Packard Center for ALS Research.
December 2016
- Dr. Rita Sattler and Dr. Stella Dracheva of the Icahn School of Medicine at Mount Sinai were awarded a grant from the Department of Veterans Affairs to study the role of ADAR2-associated RNA editing in ALS.
- Congratulations to Camelia Burciu! She is expecting a baby boy, and we are very happy for her!
November 2016
- Members of the Sattler Laboratory presented at two scientific conferences. Dr. Ileana Lorenzini presented work regarding GluA2 misediting in ALS at the 4th RNA Metabolism in Neurological Disease Satellite Meeting of the 11th Brain Research Conference and at the 2016 Society for Neuroscience (SfN) Annual Meeting, both in San Diego, California. Alex Starr also presented work investigating the role of a copper transporter mutation in ALS at the 2016 SfN Meeting.
October 2016
- The Sattler Laboratory, along with colleagues from Barrow Neurological Institute and other institutions, participated in the ALS Association’s Walk to Defeat ALS in Scottsdale, Arizona. The annual walk supports the Arizona chapter of the ALS Association in its mission to fund ALS research and provide care for patients. Researchers, family, and friends filled Scottsdale Stadium and raised over $470,000!
September 2016
- Camelia Burciu joined the laboratory as a research technician.
- Nancy Twishime gave a poster presentation summarizing her summer research at the Arizona Biosciences event.
- The Downtown Devil reported on our collaboration with Dr. Kruer’s Laboratory.
- The lab gathered at Dr. Sattler’s house for an end-of-the-summer pool party.
- Dr. Rita Sattler and Dr. Robert Bowser organized and chaired the Inaugural Arizona ALS Symposium. It was attended by more than 35 scientists and clinicians representing universities, nonprofits, hospitals, and the biomedical industry across Arizona. Over the course of three days, participants shared their work, formed new collaborations, and brainstormed future projects to expand our understanding and treatment of ALS.
August 2016
- Andrew Nelson joined the laboratory as a research technician.
- Devika Shenoy, from BASIS High School in Scottsdale, joined the laboratory as an intern.
- The staff of the laboratory had a great time and grew as a team while tubing down the Salt River.
- Nancy gave an oral presentation of her summer research at the annual Barrow summer research student day.
- Sarah started her senior thesis project in our laboratory.
July 2016
- The laboratory was awarded a grant with the Zarnescu Laboratory at the University of Arizona from the Muscular Dystrophy Association (MDA) to study the role of metabolism in ALS.
- Ileana got engaged. Congratulations, Ileana!
June 2016
- Stephen Moore joined the lab as a graduate student in neuroscience.
- Sara Swinford, an undergraduate student at ASU, began a summer internship with the laboratory.
- Nancy Twishime, a student at Bioscience High School in Phoenix, joined the laboratory as a summer intern.
- The laboratory was awarded a grant with the Kruer Laboratory at Phoenix Children’s Hospital from the Research Development and Partnership Initiative (RDPI) to study genetic forms of cerebral palsy.
April 2016
- The first hiPSC neurons are differentiating in the new desert climate.
- All new microscopes are up and running.
March 2016
- Stephen joined the laboratory as a Neuroscience graduate student on June 1.
- Sarah joined the laboratory as an undergraduate research summer intern on June 1.
February 2016
- Ileana received an invitation to submit a postdoctoral fellowship application to the ALS association (ALSA).
- Ileana received a travel award for the American Society for Neurochemistry (ASN) Meeting in Denver, Colorado.
January 2016
- Isaiah Tesfay joined the laboratory as an undergraduate research assistant.
- The laboratory received renewed funding from the Robert Packard Center for ALS Research.
- Alex Starr joined the laboratory as a new research technician.
November 2015
- Ileana arrived in Phoenix and rejoined the Sattler Laboratory.
October 2015
- Dr. Sattler and Jenny started setting up the Sattler Laboratory at Barrow.
Sattler Laboratory Publications
Modelling TDP-43 proteinopathy in Drosophila uncovers shared and neuron-specific targets across ALS and FTD relevant circuits
Date: 10/2023
Authors: R Keating Godfrey, Eric Alsop, Reed T. Bjork, Brijesh S. Chauhan, Hillary C. Ruvalcaba, Jerry Antone, Lauren M. Gittings, Allison F. Michael, Christi Williams, Grace Hala’ufia, Alexander D. Blythe, Megan Hall, Rita Sattler, Kendall Van Keuren-Jensen, Daniela C. Zarnescu
Cryptic exon detection and transcriptomic changes revealed in single-nuclei RNA sequencing of C9ORF72 patients spanning the ALS-FTD spectrum
Date: 09/2023
Authors: Lauren M. Gittings, Eric B. Alsop, Jerry Antone, Mo Singer, Timothy G. Whitsett, Rita Sattler, Kendall Van Keuren-Jensen
Moderate intrinsic phenotypic alterations in ALS/FTD iPSC-microglia despite the presence of C9orf72 pathological features
Date: 01/2023
Authors: Ileana Lorenzini, Eric Alsop, Jennifer Levy, Lauren M. Gittings, Deepti Lall, Benjamin E. Rabichow, Stephen Moore, Ryan Pevey, Lynette M. Bustos, Camelia Burciu, Divya Bhatia, Mo Singer, Justin Saul, Amanda McQuade, Makis Tzioras, Thomas A. Mota, Amber Logemann, Jamie Rose, Sandra Almeida, Fen-Biao Gao, Michael Marks, Christopher J. Donnelly, Elizabeth Hutchins, Shu-Ting Hung, Justin Ichida, Robert Bowser, Tara Spires-Jones, Mathew Blurton-Jones, Tania F. Gendron, Robert H. Baloh, Kendall Van Keuren-Jensen, Rita Sattler
Editorial: Mechanisms of Action in Neurodegenerative Proteinopathies
Date: 01/2022
Authors: Sean J. Miller, Selina Wray, Rita Sattler, Can Zhang
C9orf72 deficiency promotes microglial-mediated synaptic loss in aging and amyloid accumulation
Date: 07/2021
Authors: Deepti Lall, Ileana Lorenzini, Thomas A. Mota, Shaughn Bell, Thomas E. Mahan, Jason D. Ulrich, Hayk Davtyan, Jessica E. Rexach, A K. Muhammad, Oksana Shelest, Jesse Landeros, Michael Vazquez, Junwon Kim, Layla Ghaffari, Jacqueline Gire O’Rourke, Daniel H. Geschwind, Mathew Blurton-Jones, David M. Holtzman, Rita Sattler, Robert H. Baloh
Sattler Laboratory Positions
At this time, we are actively recruiting postdoctoral fellows.
Join a highly enthusiastic research lab that will provide the opportunity to participate in cutting edge research involving the latest discoveries in ALS and FTD research. Our lab uses a combination of state of the art molecular, cellular, and imaging technologies coupled with hiPSCs, patient autopsy brain tissue, and novel animal models in the hopes of discovering future therapies for ALS and FTD patients. This research experience is ideal for someone interested in pursuing an advanced degree and/or a career as a researcher or practitioner, or for someone who loves science and research and aspires to help people.
Sattler Lab: Alumni
The following are a list of alumni of the Sattler Laboratory along with their first placement after leaving Barrow.
Former Research Technicians
Andrew Nelson — PhD Neuroscience Graduate Student, Thomas Jefferson University
Alex Starr — Pharmacology PhD Graduate Student, University of Pennsylvania
Layla Ghaffari — Neuroscience PhD Graduate Student, Thomas Jefferson University
Helen Magee — Clinical Translational Neuroscience Master’s Student, University of Arizona, College of Medicine Phoenix
Ileana Lorenzini – Research Scientist, Banner Sun Health Research Institute
Stephen Moore — Postdoctoral Fellow, University of California San Diego
Lynette Bustos — Postdoctoral Fellow, Ionis Pharmaceuticals
Mo Roberts — Research Technician, Thomas Jefferson University
Former Interns
Abigail Byford — Undergraduate Student, University of Leeds
Sarah Swinford — Research Technician, Norton Thoracic Institute
Isaiah Tesfay — Undergraduate Student, Arizona State University
Michael Goldstein — Undergraduate Student, Arizona State University
Jordyn Shevat — Undergraduate Student, Arizona State University
Devika Shenoy — Undergraduate Student, University of California-Los Angeles
Nancy Twishime — Undergraduate Barrett Honors College Student, Arizona State University
Latavya Chintada
Divya Bhatia – Medical Student
Junny Kim
Principal Investigator
Postdoctoral Fellows

Robert Culibrk, PhD
Postdoctoral Fellow
Graduate Students

Dominic Julian
Graduate Student
Research Technicians

Kim Preller
Research Technician
Interns
- Doug Clough
- Landon Dawson
- Anushka Kale