Stokes Laboratory
Laboratory Focus
Imaging-based biomarkers play a critical role in many neuropathologies, ideally giving insight into the structural and functional changes in the brain that lead to clinical manifestations of disease. Conventional magnetic resonance imaging (MRI) is sensitive to downstream indicators of neuropathology but often lacks specificity to the underlying causes of disease. More advanced imaging methods provide unique and complementary information that is directly related to pathophysiology and could have a significant clinical impact that crosscuts all aspects of patient management.
Our research focuses on developing, validating, and translating advanced MRI acquisition and analysis methods to noninvasively assess neurological diseases and disorders, including Alzheimer’s disease, Parkinson’s disease, and multiple sclerosis. These advanced imaging methods include methods to interrogate brain hemodynamics (perfusion MRI), microstructure (diffusion MRI), and brain function (functional MRI). The long-term goal of our research is to develop advanced imaging biomarkers that can inform on the underlying disease pathophysiology.
Advanced Imaging Biomarkers in Alzheimer’s Disease
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder and is the most common cause of dementia. AD symptoms generally begin with memory difficulties and evolve to include multiple domains of cognition, eventually affecting activities of daily living. Clinical outcomes in AD remain exceedingly poor, in part due to limited understanding of its neuropathological underpinnings.
Patients may also experience mild cognitive impairment (MCI), a related syndrome characterized by objective memory decline with relative preservation of other cognitive domains and functional activities. MCI is associated with an increased risk of conversion to AD, and the MCI phase represents an opportunity for potential early intervention.
Imaging-based biomarkers using MRI play a critical role in assessing Alzheimer’s-related pathological changes, but current biomarkers are limited in their diagnostic and prognostic ability, particularly in early disease stages when intervention would be most beneficial. Work in our lab aims to overcome these challenges by developing novel MRI biomarkers with high biological and pathological specificity to better understand both aging and AD pathology. These biomarkers relate to microstructural integrity, brain network connectivity, and cerebrovascular health.
Advanced Imaging Biomarkers in Parkinson’s Disease
Parkinson’s disease (PD) is a progressive neurodegenerative disorder, and patients with PD experience significant motor and non-motor symptoms. Treatment for PD has historically focused on ameliorating the classic motor symptoms; however, patients also experience a wide range of non-motor symptoms, some of which are found to be more debilitating than the motor symptoms themselves, severely burdening patients and their families.
Imaging-based biomarkers play a critical role in assessing Parkinson’s-related pathological changes, but current biomarkers are limited in their diagnostic and prognostic ability, particularly in early disease stages when intervention would be most beneficial. Functional magnetic resonance imaging (fMRI) enables the study of brain activation and has been widely used to study global functional network changes in Parkinson’s disease. However, standard fMRI is limited in its ability to robustly measure subtle changes with disease, in part due to low sensitivity and specificity; furthermore, interpretation of standard fMRI is challenging due to the indirect link between neuronal function and MRI signal change.
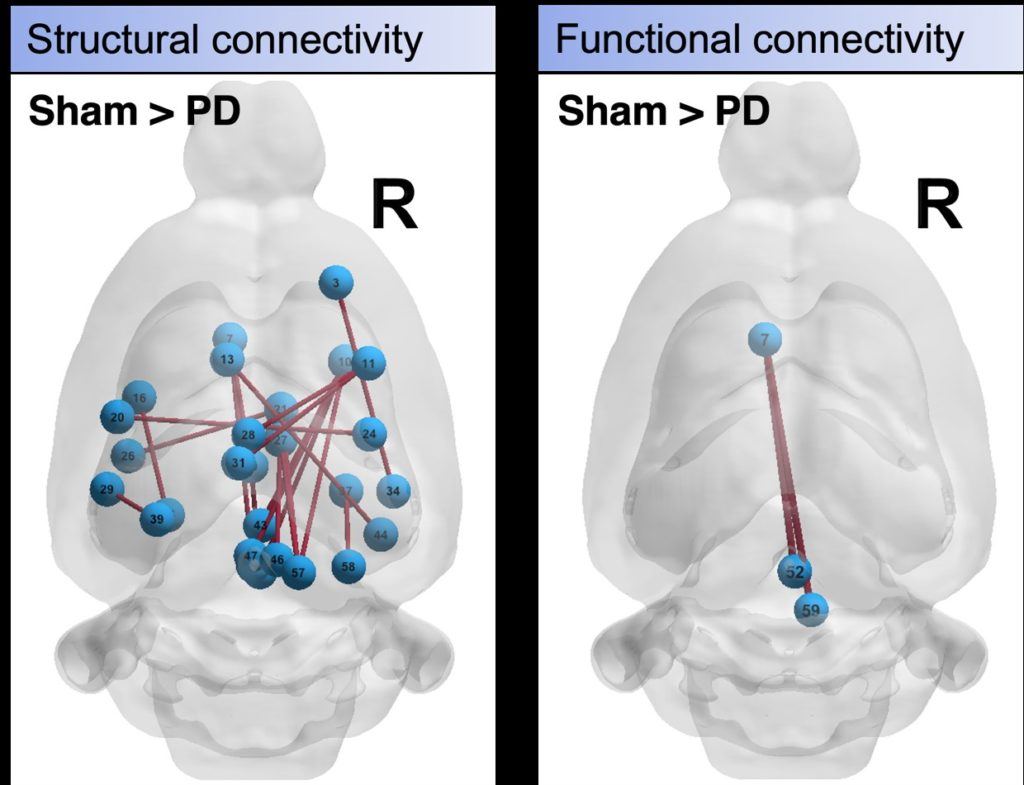
This lack of robust direct biomarkers is a critical gap that ultimately limits our ability to understand the underlying pathological changes, as well as evaluate emerging therapies. To overcome these limitations, our lab has developed and optimized an advanced multi-contrast fMRI method that provides high contrast sensitivity, as well as distinct microvascular sensitivity. By coupling this method with pharmacological and chemogenetic manipulations, a direct link between fMRI-based functional networks and underlying neuronal function can be inferred.
These studies provide critical insight into functional network changes that occur over different vascular scales and via different neurotransmitter populations, which are implicated in the multifaceted nature of Parkinson’s disease that contributes to both motor and non-motor symptoms. As functional brain network dysfunction is widely observed in Parkinson’s disease, this integrative approach will enable the development of robust biomarkers of Parkinson’s disease with well-characterized pathophysiological origins, which is a critical shortcoming of current technologies.
Advanced Imaging Biomarkers in Multiple Sclerosis
Multiple sclerosis (MS) is a chronic, debilitating disease of the central nervous system. Patients with MS experience a wide range of clinical and pathological symptoms over an unpredictable time-course often spanning decades. Relapsing-remitting MS is characterized by periods of new or worsening neurologic symptoms with a relative return to baseline between episodes.
Pathologically, MS is characterized by neuroinflammation, focal demyelination, gliosis, axonal degeneration, and neuronal loss. Remyelination of MS lesions is associated with improvement of symptoms but is highly variable; thus, therapies that foster remyelination represent an opportunity for repair before irreversible damage and decline.
Neuroimaging plays a critical role in the clinical management of MS, but conventional MRI lacks pathological specificity to the factors that exacerbate demyelination or promote remyelination. The development of MS-specific biomarkers remains a highly relevant target and may impact patient diagnosis, prognosis, disease management, therapeutic planning, and clinical trial outcomes.
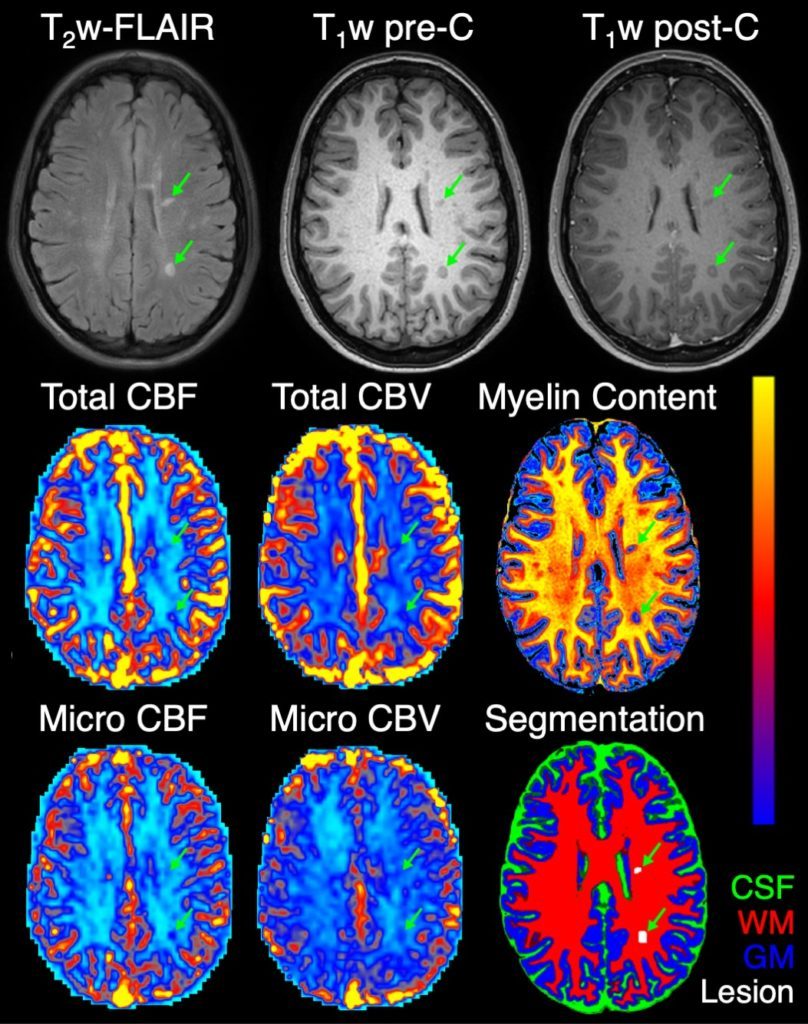
Given the importance of myelination, MRI biomarkers of myelin integrity have been developed for use in clinical trials. Unfortunately, these biomarkers reflect static levels of myelin and cannot predict demyelination or remyelination processes. Recent studies have suggested that remyelination relies on adequate tissue perfusion. While altered perfusion has been reported in MS, the relationship between perfusion and myelin has not been fully characterized in vivo.
To robustly assess cerebral perfusion across the brain, our lab has developed and validated an advanced MRI method to measure perfusion across varying vascular scales, including at the microvascular level. Using this method, we recently showed that microvascular perfusion in MS lesions is associated with reduced microstructural integrity, a key feature of demyelination. Ongoing work in our lab—in collaboration with the Dortch Laboratory—focuses on investigating the role of perfusion in demyelination and remyelination using advanced MRI biomarkers.

Contact Information
Ashley Stokes, PhD
Associate Professor, Neuroimaging Research
Barrow Neurological Institute
350 West Thomas Road
Phoenix, Arizona 85013
Ashley.Stokes@CommonSpirit.org
We are currently recruiting both graduate students and postdoctoral fellows. Our research focuses on the development, validation, and translation of advanced MRI methods in Parkinson’s disease, multiple sclerosis, and Alzheimer’s disease. These advanced methods involve acquisition and analysis of perfusion, diffusion, and functional MRI data in both preclinical disease models and clinical applications. Graduate students and fellows will work in a highly collaborative environment, with imaging-focused scientists in the Barrow Neuroimaging Innovation Center and with neuroscientists and neurologists at Barrow.
We are also accepting applications from motivated individuals who can commit to volunteering in the laboratory, but these spaces are limited. Volunteers in the Stokes Laboratory can expect to participate in a variety of research-related activities, including contribution to a research project.
Repeatability and reliability of cerebrovascular reactivity in young adults using multi-echo, multi-contrast MRI
Date: 10/2025
Authors: Elizabeth G. Keeling, Maurizio Bergamino, Lauren R. Ott, Molly M. McElvogue, Ashley M. Stokes
Motor correlates of finger tapping variability in subjective memory complaints, mild cognitive impairment and probable Alzheimer’s disease
Date: 02/2025
Authors: George P. Prigatano, Lucia W. Braga, Molly McElvogue, Katelyn Coddaire, Lori Steffes, Anna Burke, Ashley M. Stokes
Distinguishing Early from Late Mild Cognitive Impairment Using Magnetic Resonance Free-Water Diffusion Tensor Imaging
Date: 01/2025
Authors: Maurizio Bergamino, Molly M. McElvogue, Ashley M. Stokes, Alzheimer’s Disease Neuroimaging Initiative
Assessment of microstructural changes in white matter hyperintensities in aging and mild cognitive impairment revealed by advanced diffusion MRI
Date: 01/2025
Authors: Maurizio Bergamino, Megan R. Nelson, Elizabeth Keeling, Ashley M. Stokes
Investigating white matter alterations in Parkinson’s disease using multi-shell free-water DTI and NODDI: insights into neurodegeneration and levodopa effects
Date: 01/2025
Authors: Maurizio Bergamino, Shuyi Zhu, Holly A. Shill, Ashley M. Stokes
Principal Investigator
Investigators

Maurizio Bergamino, PhD
Research Assistant Professor
Postdoctoral Fellows

Mohammadreza Soltany, PhD
Postdoctoral Fellow
Graduate Students
Research Staff
- Molly McElvogue, MS – Study Coordinator
- Cindy Moreno – Study Coordinator
- Lauren Ott – Project Manager
- Jennifer White – Research Technician